Abstract
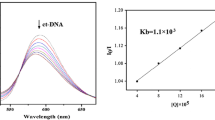
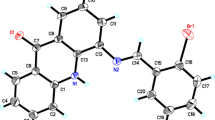
A series of novel acenaphthopyrazine derivatives was synthesized from acenaphthylene-1,2-dione via three steps, including bromination, cyclization, and SNArH reaction. These new compounds exhibited potential antiproliferative activity against MCF-7 cells in vitro, and 3-[2-(dimethylamino)ethylamino]acenaphtho[1,2-b]pyrazine-8,9-dicarbonitrile exhibited the highest activity (IC 50 = 4.60 μM). DNA-binding experiments suggested that these derivatives bind to DNA through intercalation with intrinsic binding constants K all above 105 M−1. Optical property studies indicated that these compounds have long emission wavelength (λ em > 560 nm), high quantum yields in toluene (Φf = 0.59 for 3-(morpholin-4-yl)acenaphtho[1,2-b]pyrazine-8,9-dicarbonitrile), and large Stokes shift (ΔS > 130 nm).
Graphical Abstract









Similar content being viewed by others
References
Coleman RE (2001) Cancer Treat Reviews 27:165
Jemal A, Siegel R, Ward E, Hao Y, Xu J, Murray T, Thun MJ (2008) Ca-Cancer J Clin 58:71
Coleman RE (1997) Cancer 80:1588
Corbett AH, Osheroff N (1993) Chem Res Toxicol 6:585
Braña MF, Berlanga JMC, Roldan CM (1973) DE Patent 2318136
Sami SM, Dorr RT, Alberts DS, Remers WA (1993) J Med Chem 36:765
Sami SM, Dorr RT, Alberts DS, Remers WA (1995) J Med Chem 38:983
Sami SM, Dorr RT, Alberts DS, Remers WA, Bhashyam SI (1996) J Med Chem 39:1609
Sami SM, Dorr RT, Alberts DS, Solyom AM (1996) J Med Chem 39:4978
Sami SM, Dorr RT, Alberts DS, Solyom AM, Remers WA (2000) J Med Chem 43:3067
Braña MF, Cacho M, Garcia MA, PascualTeresa B, Ramos A, Acero N, Llinares F, Munoz Mingarro D, Abradelo C, Rey-Stolle MF, Yuste M (2002) J Med Chem 45:5813
Braña MF, Cacho M, Ramos A, Dominguez MT, Pozuelo JM, Abradelo C, Rey-Stolle MF, Yuste M, Carrasco C, Bailly C (2003) Org Biomol Chem 1:648
Braña MF, Cacho M, Garcia MA, Pascual-Teresa B, Ramos AM, Dominguez T, Pozuelo JM, Abradelo C, Rey-Stolle MF, Yuste M, Banez-Coronel M, Lacal JC (2004) J Med Chem 47:1391
Braña MF, Gradillas A, Gomez A, Acero N, Llinares F, Munoz-Mingarro D, Abradelo C, Rey-Stolle MF, Yuste M, Campos J, Gallo MA, Espinosa A (2004) J Med Chem 47:2236
Li YG, Xu YF, Qian XH, Qu B (2004) Tetrahedron Lett 45:1247
Li ZG, Yang Q, Qian XH (2005) Bioorg Med Chem Lett 15:3143
Wu AB, Liu JD, Qin SX, Mei P (2010) Monatsh Chem 141:95
Xie LJ, Qian XH, Cui JN (2009) Bioorg Med Chem 17:7615
Braña MF, Cacho M, Gradillas A, Pascual-Teresa B, Ramos A (2001) Curr Pharm Des 7:1745
Erkkila KE, Odom DT, Barton JK (1999) Chem Rev 99:2777
Gibson D (2002) Pharmacogenomics J 2:275
Ihmels H, Otto D (2005) Top Curr Chem 258:161
Martínez R, Chacón-García L (2005) Curr Med Chem 12:127
Palchaudhuri R, Hergenrother PJ (2007) Curr Opin Biotech 18:497
Wheate NJ, Brodie CR, Collins JG, Kemp S, Aldrich-Wright JR (2007) Mini-Rev Med Chem 7:627
Graves DE, Velea LM (2000) Curr Org Chem 4:915
Li ZG, Yang Q, Qian XH (2005) Bioorg Med Chem 13:4864
Zhang ZC, Yang YY, Zhang DN, Wang YY, Qian XH, Liu FY (2006) Bioorg Med Chem 14:6962
Yin H, Xu YF, Qian XH (2007) Bioorg Med Chem 15:1356
Xu ZC, Xiao Y, Qian XH (2005) Org Lett 7:889
Long EC, Barton JK (1990) Acc Chem Res 23:271
Shi S, Liu J, Li J, Zheng KC, Huang XM, Tan CP, Chen LM, Ji LN (2006) J Inorg Biochem 100:385
Chan HL, Ma DL, Yang M, Che CM (2003) Chem BioChem 4:62
Yang X, Liu WH, Jin WJ, Shen GL, Yu RQ (1999) Spectrochim Acta A 55:2719
Basili S, Bergen A, Dall’Acqua F, Faccio A, Ranzhan A, Ihmels H, Moro S, Viola G (2007) Biochemistry 46:12721
Qian XH, Xiao Y (2002) Tetrahedron Lett 43:2991
Li F, Cui JN, Guo LY, Qian XH, Ren WM, Wang KW, Liu FY (2007) Bioorg Med Chem 15:5114
Acknowledgments
We are grateful to the Natural Science Foundation of China (90713030; 20876025) and the National Basic Research Program of China (2009CB724706).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Xing, J., Cui, J., Wang, S. et al. Novel acenaphthopyrazine derivatives as antitumor agents against MCF-7 cells: molecular synthesis, optical properties, and DNA-binding studies. Monatsh Chem 143, 243–250 (2012). https://doi.org/10.1007/s00706-011-0653-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00706-011-0653-9