Abstract
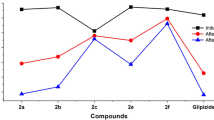
Spiroimidazolidine-2,4-diones were prepared from methylcyclohexanones by the Bucherer–Bergs reaction. Synthesis of the target 3-(arylsulfonyl)spiroimidazolidine-2,4-diones was achieved by reaction of arylsulfonyl chlorides with corresponding spiroimidazolidine-2,4-diones. The synthesis was confirmed by spectroanalytical techniques and the crystal structure of 3-(4-methoxyphenylsulfonyl)-6-methyl-1,3-diazaspiro[4.5]decane-2,4-dione, and the purity was checked by GC–MS analysis. The in-vivo hypoglycemic potential of 6-methyl-, 7-methyl-, and 8-methyl-3-(4-methylphenylsulfonyl)-1,3-diazaspiro[4.5]decane-2,4-dione was investigated on male albino rats. The screened compounds were found to have excellent hypoglycemic activity. 6-Methyl-3-(4-methylphenylsulfonyl)-1,3-diazaspiro[4.5]decane-2,4-dione was found highly active, reducing the blood glucose level by 60.79% compared with 41.60% by the standard (glipizide) at a dose level of 100 mg/kg of the mice body weight. The 8-methyl isomer was also more potent than the standard, with 48.56% reduction in blood glucose level.
Graphical Abstract


Similar content being viewed by others
References
Kashif MK, Ahmad I, Hameed S (2008) Arkivoc xvi:311
Wild S, Roglic G, Green A, Sicree R, King H (2004) Diabetes Care 27:1047
Hamdan II, Afifi FU (2004) J Ethnopharmacol 93:117
Zhang Q, Tang N, Schepmoes AA, Phillips LS, Smith RD, Metz TO (2008) J Proteome Res 7:2025
Phung OJ, Scholle JM, Talwar M, Coleman CI (2010) J Am Med Assoc 303:1410
Waqar MA, Aijaz S, Shaukat S, Waqar A (2009) J Chem Soc Pak 31:677
Shaukat S, Waqar A, Waqar MA (2009) J Chem Soc Pak 31:480 and references cited therein
Ono H, Nozawa Y, Hayano S (1982) Nippon Ganka Gakkai Zasshi 86:1343
Varma SD, Kinoshita JH (1976) Biochem Pharmacol 25:2505
Murcia-Soler M, Pérez-Giménez F, Nalda-Molina R, Salabert-Salvador MT, García-March FJ, Cercós-del-Pozo RA, Garrigues TM (2001) J Chem Inf Comput Sci 41:1345
Pandeya SN, Raja AS, Stables JP (2002) J Pharm Pharmaceut Sci 5:266
Kashif MK, Hussain A, Rauf MK, Ebihara M, Hameed S (2008) Acta Cryst E64:o444
Ahmad I, Hameed S, Duddeck H, Lenzen S, Rustenbeck I, Ahmad R (2002) Z Naturforsch 57B:349
Mahmoodi NO, Khodaee Z (2007) Arkivoc iii:29
Brimelow HC, Carrington HC, Vasey CH, Waring WS (1962) J Chem Soc 2789
Gauthier TJ, Yokum TS, Morales GA, McLaughlin ML, Liu YH (1997) Acta Cryst C53:1659
Kashif MK, Rauf MK, Bolte M, Hameed S (2009) Acta Cryst E65:o1893
Meanwell NA, Sing YS, Gao J, Wong HS, Gao Q, Denis R, Laurent S, Balasubramanian N (1995) J Org Chem 60:1565
Feng F, Uno B, Goto M, Zhang Z, An D (2002) Talanta 57:481
Henze HR, Speer RJ (1942) J Am Chem Soc 64:522
Kashif MK, Rauf MK, Bolte M, Hameed S (2009) Acta Cryst E65:o1892
APEX2 Ver. 2008.5 (2008) Bruker AXS, Inc, Madison, Wisconsin, USA
Sheldrick GM (2008) Acta Cryst A64:112
Farrugia LJ (1997) J Appl Cryst 30:565
Brown GR, Foubister AJ (1984) J Med Chem 27:79
Shukla R, Barve V, Padhye S, Bhonde R (2004) Bioorg Med Chem Lett 14:4961
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Iqbal, Z., Akhtar, T., Hendsbee, A.D. et al. Synthesis, characterization, and hypoglycemic activity of 3-(arylsulfonyl)spiroimidazolidine-2,4-diones. Monatsh Chem 143, 497–504 (2012). https://doi.org/10.1007/s00706-011-0600-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00706-011-0600-9