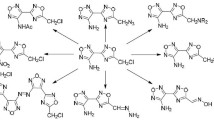
Abstract
Using KF as base and THF as solvent, 5-alkoxy-3,4-dihalo-2(5H)-furanones were reacted with secondary amines. The normal products, β-amino-2(5H)-furanones, were obtained via the tandem Michael addition-elimination reaction as expected in most cases. However, the reaction between diisopropylamine and 3,4-dihalo-5-methoxy-2(5H)-furanones yielded unanticipated products, methyl (E)-2-halo-4-(diisopropylamino)-4-oxobut-2-enoates. The possible synthetic mechanism involving a rearrangement reaction was proposed. The biological activity assay of products derived from the reactions of 5-alkoxy-3,4-dihalo-2(5H)-furanones with different amino compounds was preliminarily investigated by the MTT method on A549 cells in vitro, which revealed that some derivatives of amino acid esters with a benzene ring exhibited the best anticancer bioactivity.
Graphical Abstract





Similar content being viewed by others
Notes
Some literature on alkoxy migration (especially involving in the 1,4-methoxy migration and the migration on five-membered rings).
Some literature involving the formation of different propadienones.
References
Rahim MA, Rao PNP, Knaus EE (2002) Bioorg Med Chem Lett 12:2753
Pal M, Veeramaneni VR, Nagabelli M, Kalleda SR, Misra P, Casturi SR, Yeleswarapu KR (2003) Bioorg Med Chem Lett 13:1639
Ghosh AK, Leshchenko S, Noetzel M (2004) J Org Chem 69:7822
Guerrero MD, Aquino M, Bruno I, Terencio MC, Paya M, Riccio R, Gomez-Paloma L (2007) J Med Chem 50:2176
Chen QH, Geng Z, Huang B (1995) Tetrahedron Asymmetr 6:401
Bertrand S, Hoffmann N, Pete JP (1999) Tetrahedron Lett 40:3173
Carter NB, Nadany AE, Sweeney JB (2002) J Chem Soc Perkin Trans 1 2324
Bellina F, Falchi E, Rossi R (2003) Tetrahedron 59:9091
Ma S, Wu B, Shi Z (2004) J Org Chem 69:1429
Kabalka GW, Dong G, Venkataiah B (2004) Tetrahedron Lett 45:5139
Song YS, Lee YJ, Kim BT, Heo JN (2006) Tetrahedron Lett 47:7427
Sarma DK, Zhang J, Curran TT (2007) J Org Chem 72:3311
Boukouvalas J, Côté S, Ndzi B (2007) Tetrahedron Lett 48:105
Garzelli R, Samaritani S, Malanga C (2008) Tetrahedron 64:4183
Iskander G, Zhang R, Chan D, Black DS, Alamgir M, Kumar N (2009) Tetrahedron Lett 50:4613
Murthy SN, Madhav B, Kumar AV, Rao KR, Nageswar YVD (2009) Tetrahedron 65:5251
Kataoka T, Kinoshita H, Kinoshita S, Iwamura T (2002) Tetrahedron Lett 43:7039
Rama-Rao VVVNS, Venkat-Reddy G, Maitraie D, Ravikanth S, Yadla R, Narsaiah B, Shanthan-Rao P (2004) Tetrahedron 60:12231
Li XW, Wang ZY, Zheng LY (2006) Chin J Org Chem 26:1144
Zheng JC, Liao WW, Tang Y, Sun XL, Dai LX (2005) J Am Chem Soc 127:12222
Guo JB, Zhang XY, Chen QH (2006) Acta Chim Sin 64:2008
Chen CG, Hou XL, Pu L (2009) Org Lett 11:2073
Xia XF, Shu XZ, Ji KG, Yang YF, Shaukat A, Liu XY, Liang YM (2010) J Org Chem 75:2893
Kang S, Shi QH, Ha MW, Ku JM, Cheng MS, Jeong BS, Park HG, Jew SS (2010) Tetrahedron 66:4326
Zikan V, Vrba L, Kakac B, Semosky M (1973) Collect Czech Chem Commun 38:1091
Chen QH, Geng Z (1993) Chin J Org Chem 13:299
Li XQ, Gong BL, Wang FR, Chen QH, Wang ZD (2001) Chem J Chin Univ 22:78
Lattmann E, Sattayasai N, Schwalbe CS, Niamsanit S, Billington DC, Lattmann P, Langley CA, Singh H, Dunn S (2006) Curr Drug Disc Technol 3:125
Song XM, Wang ZY, Fu JH, Li JX (2009) J South China Normal Univ (Natural Sci) 75
Song XM, Wang ZY, Li JX, Fu JH (2009) Chin J Org Chem 29:1804
Mo YQ, Wang ZY, Li JX, Hong WK (2010) Chin J Org Chem 30:1051
Song XM, Tan YH, Li JX, Wang ZY (2010) Chin J Org Chem 30:1890
Kurbangalieva AR, Devyatova NF, Bogdanov AV, Berdnikov EA, Mannafov TG, Krivolapov DB, Litvinov IA, Chmutova GA (2007) Phosphorus, Sulfur Silicon Relat Elem 182:607
Devyatova NF, Kosolapova LS, Kurbangalieva AR, Berdnikov EA, Lodochnikova OA, Litvinov IA, Chmutova GA (2008) Russ J Org Chem 44:1225
Kurbangalieva AR, Devyatova NF, Kosolapova LS, Lodochnikova OA, Berdnikov EA, Litvinov IA, Chmutova GA (2009) Russ Chem Bull 58:126
Wei MX, Feng L, Li XQ, Zhou XZ, Shao ZH (2009) Eur J Med Chem 44:3340
Ratts KW, Phillips WG (1974) J Org Chem 39:3300
Polezhaeva NA, Volodina YM, Kalinina IV, Sakhibullina VG, Chmutova GA, Galkin VI, Cherkasov RA (2002) Russ J Gen Chem 72:1283
Kurbangalieva AR, Bogdanov AV, Movchan AI, Chmutova GA (2004) Russ J Org Chem 40:1216
Teitei T (1986) Aust J Chem 39:503
Mellor JM, Wagland AM (1989) J Chem Soc Perkin Trans 1 997
Dawson JR, Mellor JM (1995) Tetrahedron Lett 36:9043
Beccalli EM, Marchesini A (1995) Tetrahedron 51:2353
Hashmi ASK, Grundl MA, Nass AR, Naumann F, Bats JW, Bolte M (2001) Eur J Org Chem 4705
Frisch MJ, Trucks GW, Schlegel HB, Scuseria GE, Robb MA, Cheeseman JR, Montgomery JA Jr, Vreven T, Kudin KN, Burant JC, Millam JM, Iyengar SS, Tomasi J, Barone V, Mennucci B, Cossi M, Scalmani G, Rega N, Petersson GA, Nakatsuji H, Hada M, Ehara M, Toyota K, Fukuda R, Hasegawa J, Ishida M, Nakajima T, Honda Y, Kitao O, Nakai H, Klene M, Li X, Knox JE, Hratchian HP, Cross JB, Bakken V, Adamo C, Jaramillo J, Gomperts R, Stratmann RE, Yazyev O, Austin AJ, Cammi R, Pomelli C, Ochterski JW, Ayala PY, Morokuma K, Voth GA, Salvador P, Dannenberg JJ, Zakrzewski VG, Dapprich S, Daniels AD, Strain MC, Farkas O, Malick DK, Rabuck AD, Raghavachari K, Foresman JB, Ortiz JV, Cui Q, Baboul AG, Clifford S, Cioslowski J, Stefanov BB, Liu G, Liashenko A, Piskorz P, Komaromi I, Martin RL, Fox DJ, Keith T, Al-Laham MA, Peng CY, Nanayakkara A, Challacombe M, Gill PMW, Johnson B, Chen W, Wong MW, Gonzalez C, Pople JA (2003) Gaussian 03, revision C.01. Gaussian Inc, Pittsburgh, PA
Somnitz H, Zellner R (2000) Phys Chem Chem Phys 2:1899
Plützer C, Kleinermanns K (2002) Phys Chem Chem Phys 4:4877
Finnerty JJ, Wentrup C (2004) J Org Chem 69:1909
Iyer VK, Horwitz JP (1982) J Org Chem 47:644
Morlender-Vais N, Mandelbaum A (1999) J Mass Spectrom 34:637
Xu Y, Zhu SZ (2001) Tetrahedron 57:3909
Abe M, Hattori M, Takegami A, Masuyama A, Hayashi T, Seki S, Tagawa S (2006) J Am Chem Soc 128:8008
Briehl H, Lukosch A, Wentrup C (1984) J Org Chem 49:2772
Rickborn B (1998) Org React 52
Bibas H, Moloney DWJ, Neumann R, Shtaiwi M, Bernhardt PV, Wentrup C (2002) J Org Chem 67:2619
Chiang Y, Gaplovsky M, Kresge AJ, Leung KH, Ley C, Mac M, Persy G, Phillips DL, Popik VV, Roedig C, Wirz J, Zhu Y (2003) J Am Chem Soc 125:12872
Halton B, Dixon GM, Jones CS, Parkin CT, Veedu RN, Bornemann H, Wentrup C (2005) Org Lett 7:949
Veedu RN, Kokas OJ, Couturier-Tamburelli I, Koch R, Aycard JP, Borget F, Wentrup C (2008) J Phys Chem A 112:9742
Fulloon BE, Wentrup C (2009) Aust J Chem 62:115
Alvarez MA, Garcia ME, Martinez ME, Menendez S, Ruiz MA (2010) Organometallics 29:710
Wang JP, Zhang XY, Chen QH (2001) Chin Sci Bull 46:1427
Shi DQ, Chen Q, Li ZH (2005) Chin J Org Chem 25:549
Isidro-Llobet A, Alvarez M, Albericio F (2009) Chem Rev 109:2455
Mao ZZ, Wang ZY, Mei WJ, Yang K (2010) Chin J Chem 28:818
Flack HD (1983) Acta Cryst A 39:876
Farrugia LJ (1997) J Appl Cryst 30:565
Sheldrick GM (1996) SADABS. University of Göttingen, Germany
Sheldrick GM (2008) Acta Cryst A 64:112
Acknowledgements
The authors are grateful to the National Natural Science Foundation of China (no. 20772035) and the Natural Science Foundation of Guangdong Province (no. 5300082) for the financial support of this work. We also thank Dr. Guo-Liang Li and Dr. Li-Ming Fang for helpful discussions.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Mo, YQ., Wang, ZY., Mei, WJ. et al. Reaction of 5-alkoxy-3,4-dihalo-2(5H)-furanones with secondary amines: expected versus unanticipated products and their preliminary bioactivity investigations. Monatsh Chem 143, 443–453 (2012). https://doi.org/10.1007/s00706-011-0594-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00706-011-0594-3