Abstract
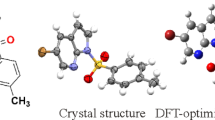
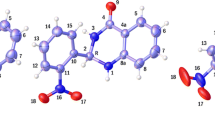
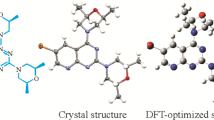
Density functional theory at B3LYP/6-31++G(d,p) level was applied to study structural, electronic, and bonding characteristics of some 3,4-dihydropyrimidin-2(1H)-ones substituted at C4 position (DHPMs) of pharmaceutical interest. Results of this study show that in these DHPMs the six-membered ring adopts a pseudo-boat conformation with a pseudo-axial orientation of the C4 substituent, and the heights of the C4 and N4 atoms from the boat plane depend on the type and position of the substituent on the aryl ring. Thermochemical analysis of these DHPMs and their corresponding oxidation products shows that the enthalpy of the oxidation reaction depends on the nature of the substituent at the C4 position, falling in a range of 25 kJ/mol for all compounds.
Graphical Abstract








Similar content being viewed by others
References
Nakamichi N, Kawashita Y, Hayashi M (2002) Org Lett 4:3955
Kappe CO (1993) Tetrahedron 49:6937
Kappe CO, Fabian WMF, Semones MA (1997) Tetrahedron 53:2803
Kappe CO (2000) Eur J Med Chem 35:1043
Atwal KS, Rovnyak GC, Kimball SD, Floyd DM, Moreland S, Swanson BN, Gougoutas JZ, Schwartz J, Smillie KM (1990) J Med Chem 33:2629
Blacquiere JM, Sicora O, Vogles CM, Čuperlović-Culf M, Decken A, Ouellelte RJ, Westcott SA (2005) Can J Chem 83:2052
Brands M, Endermann R, Gahlmann R, Krüger J, Raddatz S (2003) Bioorg Med Chem Lett 13:241
Memarian HR, Sadeghi MM, Aliyan H (1998) Indian J Chem 37B:219
Sadeghi MM, Memarian HR, Khosropour AR (1998) J Sci Islamic Repub Iran 9:240
Memarian HR, Sadeghi MM, Momeni AR (1999) Indian J Chem 38B:800
Memarian HR, Sadeghi MM, Momeni AR, Döpp D (2002) Monatsh Chem 133:661
Memarian HR, Bagheri M, Döpp D (2004) Monatsh Chem 135:833
Memarian HR, Abdoli-Senejani M, Döpp D (2006) Z Naturforsch 61B:50
Memarian HR, Ghazaie M, Khakhki Mehneh S (2009) Z Naturforsch 64B:1187
Shobha D, Adharvana Chari M, Ahn KH (2009) Chin Chem Lett 20:1059
Kamaljit S, Sukhdeep S (2009) Tetrahedron 65:4106
Ahmed B, Khan RA, Habibullah, Keshari M (2009) Tetrahedron Lett 50:2889
Debache A, Amimour M, Belfaitah A, Rhouati S, Carboni B (2008) Tetrahedron Lett 49:6119
Goldman S, Stoltefuss J (1991) Angew Chem 103:1587
Goldman S, Stoltefuss J (1991) Angew Chem Int Ed 30:1559
Memarian HR, Sabzyan H, Abdoli-Senejani M (2007) J Mol Struct (Theochem) 813:39
Triggle DJ, Padmanabhan S (1995) Chemtract Org Chem 8:191
Rovnyak GC, Kimball SD, Beyer B, Cucinotta G, Dimarco JD, Gougoutas J, Berg AH, Malley M, Mc Carthy JP, Zhang R, Moreland S (1995) J Med Chem 38:119
Bikker JA, Weaver DF (1993) J Mol Struct (Theochem) 281:173
Palmer RB, Andersen NH (1996) Tetrahedron 52:9665
Langs DA, Kwon YW, Strong PD, Triggle DJ (1991) J Comput Aided Mol Des 5:95
Gaudio AC, Korolkovas A, Takahata Y (1994) J Mol Struct (Theochem) 303:255
Schnell B, Strauss UT, Verdino P, Faber K, Kappe CO (2000) Tetrahedron Asymmetry 11:1449
Kappe CO, Shishkin O, Uray G, Verdino P (2000) Tetrahedron 56:1859
Atwal KS, Swanson BN, Unger SE, Floyd DM, Moreland S, Hedberg A, ÓReilly BC (1991) J Med Chem 34:806
Krenn W, Verdino P, Uray G, Faber K, Kappe CO (1991) Chirality 11:659
Uray G, Verdino P, Belaj F, Kappe CO, Fabian WMF (2001) J Org Chem 66:6685
Fabian WMF, Semones MA, Kappe CO (1998) J Mol Struct (Theochem) 432:219
Memarian HR, Farhadi A (2009) J Iran Chem Soc 6:638
Memarian HR, Sabzyan H, Farhadi A (2009) Z Naturforsch 64B:532
Memarian HR, Farhadi A (2008) Ultrason Sonochem 15:1015
Kappe CO (1997) First international electronic conference on synthetic organic chemistry (ECSOC-1), September 1–30 (1997). http://www.mdpi.org/ecsoc/posters/postersn.htm. Accessed 25 Aug 2010
Hyperchem Release 7.0, Windows Molecular Modeling System, Hypercube, Inc. http://www.hyper.com. Accessed 25 Aug 2010
Frisch MJ, Trucks GW, Schlegel HB, Scuseria GE, Robb MA, Cheeseman JR, Zakrzewski VG, Montgomery JA, Stratmann RE, Burant JC, Dapprich S, Millam JM, Daniels AD, Kudin KN, Strain MC, Farkas O, Tomasi J, Barone V, Cossi M, Cammi R, Mennucci B, Pomelli C, Adamo C, Clifford S, Ochterski J, Petersson GA, Ayala PY, Cui Q, Morokuma K, Malick DK, Rabuck AD, Raghavachari K, Foresman JB, Ciolowski J, Ortiz JV, Stefanov BB, Liu G, Liashenko A, Piskorz P, Komaromi I, Gomperts R, Martin RL, Fox DJ, Keith T, Al-Laham MA, Peng CY, Nanayakkara A, Gonzalez C, Challacombe M, Gill PMW, Johnson B, Chen W, Wong MW, Andres JL, Head-Gordon M, Replogle ES, Pople JA (1998) Gaussian 98. Gaussian Inc, Pittsburg
Carpenter JE, Weinhold F (1988) J Mol Struct (Theochem) 169:41
Reed AE, Weinstock RB, Weinhold F (1985) J Chem Phys 83:735
Reed AE, Curtiss LA, Weinhold F (1988) Chem Rev 88:899
Xiao-Hong L, Zheng-Xin T, Xian-Zhou Z (2009) J Mol Struct (Theochem) 900:50
Acknowledgments
We are thankful to the Center of Excellence (Chemistry), Research Council, and Office of Graduate Studies of the University of Isfahan for financial support.
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Memarian, H.R., Sabzyan, H. & Farhadi, A. DFT study of the molecular structure of 3,4-dihydropyrimidin-2(1H)-ones. Monatsh Chem 141, 1203–1212 (2010). https://doi.org/10.1007/s00706-010-0389-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00706-010-0389-y