Abstract
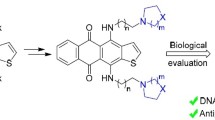
ω,ω′-Disubstituted hypericin derivatives with the nucleo-bases thymine, cytosine, and adenine in these positions were prepared starting from tri-O-methyl-ω-bromoemodin. The most promising derivative proved to be that with a thymine moiety. It displayed the best solubility of the three products together with a potency to produce singlet oxygen and/or reactive oxygen species comparable to the parent compound hypericin. In addition, although no specific interaction with DNA or poly(2′-deoxyadenylic acid) could be detected, it proved to be significantly better accumulating in the nucleus of prostatic cancer LNCaP cells than hypericin making it a promising candidate for a second-generation photodynamic hypericin agent.
Similar content being viewed by others
References
H Falk (1999) Angew Chem 111 3306 Occurrence Handle10.1002/(SICI)1521-3757(19991102)111:21<3306::AID-ANGE3306>3.0.CO;2-E
H Falk (1999) Angew Chem Int Ed Engl 38 3116 Occurrence Handle10.1002/(SICI)1521-3773(19991102)38:21<3116::AID-ANIE3116>3.0.CO;2-S
M Waser H Falk (2007) Curr Org Chem 11 547 Occurrence Handle10.2174/138527207780368229 Occurrence Handle1:CAS:528:DC%2BD2sXkt1Ojsrg%3D
M Frieden M Giraud CB Reese Q Song (1998) J Chem Soc Perkin Trans 1 2827 Occurrence Handle10.1039/a803878c
RA Obermüller H Falk (2001) Monatsh Chem 132 1519 Occurrence Handle10.1007/s007060170009
H Falk J Meyer M Oberreiter (1993) Monatsh Chem 124 339 Occurrence Handle10.1007/BF00810594 Occurrence Handle1:CAS:528:DyaK3sXltlyqtLw%3D
IM Lagoja S Pochet V Boudou R Little E Lescrinier J Rozenski P Herdewijn (2003) J Org Chem 68 1867 Occurrence Handle10.1021/jo0205098 Occurrence Handle1:CAS:528:DC%2BD3sXnsVyrsw%3D%3D
MW Bullock JJ Hand ELR Stokstad (1957) J Org Chem 22 568 Occurrence Handle10.1021/jo01356a607 Occurrence Handle1:CAS:528:DyaG1cXmsVam
Zikan V, Radl S (1988) CS 252622
K Hagenbuchner H Falk (1999) Monatsh Chem 130 1075 Occurrence Handle1:CAS:528:DyaK1MXmtlKls70%3D
Z Zeng R Qiao J Zhou S Xia Y Zhang Y Liu J Chen X Wang B Zhang (2007) J Phys Chem B 111 3742 Occurrence Handle10.1021/jp066301y Occurrence Handle1:CAS:528:DC%2BD2sXktVygtr4%3D
RF Pasternack RA Brigandi MJ Abrams AP Williams EJ Gibbs (1990) Inorg Chem 29 4483 Occurrence Handle10.1021/ic00347a030 Occurrence Handle1:CAS:528:DyaK3cXmtVKrtb0%3D
P Miskovsky F Sureau L Chinsky PY Turpin (1995) Photochem Photobiol 62 546 Occurrence Handle10.1111/j.1751-1097.1995.tb02382.x Occurrence Handle1:CAS:528:DyaK2MXotVWjtLg%3D
A Colasanti A Kisslinger R Liuzzi M Quarto P Riccio G Roberti D Tramontano F Villani (2000) J Photochem Photbiol B 54 103 Occurrence Handle10.1016/S1011-1344(99)00149-9 Occurrence Handle1:CAS:528:DC%2BD3cXjsVCqtb4%3D
ATR Williams SA Winfield JN Miller (1983) Analyst 108 1067 Occurrence Handle10.1039/an9830801067 Occurrence Handle1:CAS:528:DyaL2cXislKquw%3D%3D
TNH Tran H Falk (1996) Monatsh Chem 127 717 Occurrence Handle10.1007/BF00817262
Author information
Authors and Affiliations
Corresponding author
Additional information
Correspondence: Heinz Falk, Institute of Organic Chemistry, Johannes Kepler University, Altenbergerstraße 69, 4040 Linz, Austria, Europe.
Rights and permissions
About this article
Cite this article
Geißlmeir, D., Falk, H. ω,ω′-Appended nucleo-base derivatives of hypericin. Monatsh Chem 139, 1127–1136 (2008). https://doi.org/10.1007/s00706-008-0898-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00706-008-0898-0