Summary.
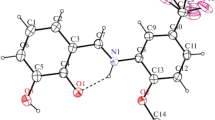
All-endo-3-amino-5-hydroxybicyclo[2.2.1]heptane-2-carboxylic acid and two epimers of 3-amino-6-hydroxybicyclo[2.2.1]heptane-2-carboxylic acid were prepared via 1,3-oxazine or γ-lactone intermediates by the stereoselective functionalization of endo-3-aminobicyclo[2.2.1]hept-5-ene-2-carboxylic acid derivatives. Their structures were proved by IR and NMR spectroscopy, with the use of HMQC, HMBC, DEPT, and DIFFNOE techniques.
Similar content being viewed by others
References
Juaristi E (1997) Enantioselective Synthesis of β-Amino Acids. Wiley-VCH, New York
F Fringuelli F Pizzo (2003) J Org Chem 68 7041 Occurrence Handle1:CAS:528:DC%2BD3sXmtFOltro%3D
M Ciclosi C Fava R Galeazzi M Orena J Sepulveda-Arques (2002) Tetrahedron Lett 43 2199 Occurrence Handle10.1016/S0040-4039(02)00233-2 Occurrence Handle1:CAS:528:DC%2BD38XhslGksr8%3D
R Roers GL Verdine (2001) Tetrahedron Lett 42 3563 Occurrence Handle1:CAS:528:DC%2BD3MXjtlSksLw%3D
AM Nocioni C Papa C Tomasini (1999) Tetrahedron Lett 40 8453 Occurrence Handle10.1016/S0040-4039(99)01772-4 Occurrence Handle1:CAS:528:DyaK1MXntlKiurc%3D
EGJC Warmerdam RD Van Rijn J Brussee CG Kruse A Van der Gen (1996) Tetrahedron Asymm 7 1723 Occurrence Handle1:CAS:528:DyaK28XjvVKnt7k%3D
O Roy G Pattenden DC Pryde C Wilson (2003) Tetrahedron 59 5115 Occurrence Handle1:CAS:528:DC%2BD3sXkvVKhtL4%3D
HJ Ha GS Park YG Ahn GS Lee (1998) Bioorg Med Chem Lett 8 1619 Occurrence Handle10.1016/S0960-894X(98)00271-6 Occurrence Handle1:CAS:528:DyaK1cXkvValur8%3D
RA Tromp M Van der Hoeven A Amore J Brussee M Overhand GA Van der Marel A Van der Gen (2001) Tetrahedron Asymm 12 1109 Occurrence Handle10.1016/S0957-4166(01)00193-8 Occurrence Handle1:CAS:528:DC%2BD3MXksFOisLs%3D
MA Cuifolini T Shimizu S Swaminathan N Xi (1997) Tetrahedron Lett 38 4947
F Fülöp (2001) Chem Rev 101 2181
A Avenoza C Cativiela M Paris JM Peregrina B Saenz-Torre (1997) Tetrahedron Asymm 8 1123 Occurrence Handle1:CAS:528:DyaK2sXivVeitbg%3D
C Cinquin M Bortolussi R Bloch (1996) Tetrahedron Asymm 7 3327 Occurrence Handle10.1016/0957-4166(96)00434-X Occurrence Handle1:CAS:528:DyaK28XntlGisro%3D
H Suga N Tanimoto AJ Sinskey S Masamune (1994) J Am Chem Soc 116 11197 Occurrence Handle1:CAS:528:DyaK2MXhvFyqt7c%3D
S Kobayashi K Kamiyama M Ohno (1990) J Org Chem 55 1169 Occurrence Handle1:CAS:528:DyaK3cXpvFSlug%3D%3D
Avenoza A, Cativiela C, Fernandez-Recio MA, Peregrina MJ (1995) Synlett 891
P Sohár G Stájer G Bernáth (1983) Org Magn Resonance 21 512
P Sohár I Pelczer G Stájer G Bernáth (1987) Magn Reson Chem 25 584
F Miklós P Sohár A Csámpai R Sillanpää M Péter G Stájer (2002) Heterocycles 57 2309
Sohár P (1983) Nuclear Magnetic Resonance Spectroscopy, vol 2. CRC Press, Boca Raton, Florida
Holly S, Sohár P (1975) In: Láng L, Prichard WH (eds) Theoretical and Technical Introduction to the Series “Absorption Spectra in the Infrared Region”. Akadémiai Kiadó, Budapest, p 95
The compounds discussed in this paper are racemates. The Schemes show only the enantiomers of the starting compounds 1 and 5 in which C–1 and C–3 have (S), while C–2 and C–4 have (R) configurations. IUPAC Nomenclature of Organic Chemistry, Section F, Stereochemistry, Pure Appl Chem (1976) 45: 11
G Stájer EA Szabó F Fülöp G Bernáth (1983) J Heterocyclic Chem 20 1181 Occurrence Handle10.1002/jhet.5570200509
P Canonne M Akssira A Dahdouh H Kasmi M Boumzebra (1993) Tetrahedron 49 1985 Occurrence Handle10.1016/S0040-4020(01)86298-0 Occurrence Handle1:CAS:528:DyaK3sXkt1yjtbg%3D
G Stájer EA Szabó F Fülöp G Bernáth P Sohár (1984) J Heterocyclic Chem 21 1373
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Palkó, M., Sándor, E., Sohár, P. et al. Synthesis and Stereostructure of 3-Amino-5- and -6-hydroxybicyclo[2.2.1]heptane-2-carboxylic Acid Diastereomers. Monatsh. Chem. 136, 2051–2058 (2005). https://doi.org/10.1007/s00706-005-0369-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00706-005-0369-9