Summary.
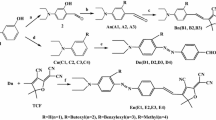
The benzoate 1B region exciton Cotton effects, hitherto unexplored, were analyzed for their use in stereochemical assignments in both acyclic (conformationally flexible) and cyclic molecules. It was found that a strong, allowed 1Ba transition, polarized longitudinally, dominates the 1B region (185–210 nm) both in the UV and the CD spectra. The exciton Cotton effects due to this transition have the same sign (but differing magnitude) as those due to the 1La (CT) band. The other component of the nearly degenerate 1B system, i.e. 1Bb transition, polarized orthogonally to the 1Ba transition, gives a Cotton effect in the case of di- and poly(4-chlorobenzoate) chromophoric system, the sign of which is sensitive to the configuration of di- or polyol. In rigid 5β-steroid skeleton 1Bb transition couplings appear responsible for strong exciton Cotton effects due to nearly parallelly oriented benzoate chromophores. Whereas 1Ba transition excitation energy appears insensitive to the nature of a substituent in 4-position of the benzoate chromophore, substitution with a donor group (methoxy, dimethylamino) brings about a red shift of the 1Bb band, although less pronounced than the red shift of the 1La (CT) band.
Similar content being viewed by others
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Grajewski, J., Gawronska, K. & Gawronski, J. Exciton Cotton Effects of Benzoates in the 1B Transition Region. Demonstration and Applications. Monatshefte für Chemie 136, 447–459 (2005). https://doi.org/10.1007/s00706-004-0272-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00706-004-0272-9