Summary.
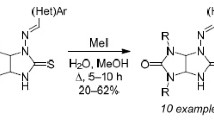
N-Acyl-α-triphenylphosphonioglycinates when treated with triethylamine are transformed to an equilibrium mixture of the corresponding N-acyliminoacetates and N-acyl-α-triphenylphosphoranylideneglycinates. Wittig reaction of the latter ylides with aromatic and aliphatic aldehydes or ketones enables a new easy entry to N-acyl-α,β-dehydro-α-amino acid esters.
Similar content being viewed by others
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Mazurkiewicz, R., Kuźnik, A., Grymel, M. et al. N-Acyl-α-triphenylphosphonioglycinates in the Synthesis of α,β-Dehydro-α-amino Acid Derivatives. Monatshefte für Chemie 135, 807–815 (2004). https://doi.org/10.1007/s00706-003-0167-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00706-003-0167-1