Summary.
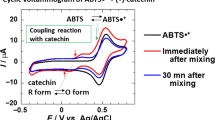
Comparative studies of redox behaviour and antioxidative activity (AOA) were carried out with two series of arylnaphthoquinone derivatives. For the electrochemical investigations, a voltammetric procedure combined with a glassy carbon electrode was applied. The AOA was examined using the photo-chemiluminometric method with the Photochem® measuring device according to Popov et al. and the chemical procedure with the radical quencher DPPH (diphenylpicrylhydrazyl). Both the redox potential and the antioxidative activity of the investigated compounds are influenced by their substituents. Compounds with the OH-group at position 3 are easiest to oxidize (E p = 0.18 V; pH 7.4). The lag phases (AOA) of these compounds are five to ten times greater than those of the strong antioxidants ascorbic acid and Trolox®. As suggested for other natural compounds, we also found a good correlation between the oxidation potential and the AOA. Therefore, it seems that a good antioxidative activity requires a low redox potential.
Similar content being viewed by others
Author information
Authors and Affiliations
Additional information
Corresponding authors. E-mail: wurglics@pharmchem.uni-frankfurt.de
Received March 1, 2002; accepted March 28, 2002
Rights and permissions
About this article
Cite this article
Wurglics, M., Michelitsch, A. & Wurm, G. Comparative Studies of the Redox Behaviour and the Antioxidative Activity of Arylnaphthoquinones. Monatshefte für Chemie 133, 1225–1230 (2002). https://doi.org/10.1007/s00706-002-0479-6
Issue Date:
DOI: https://doi.org/10.1007/s00706-002-0479-6