Abstract
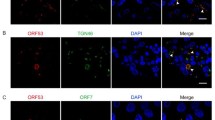
Kaposi’s sarcoma-associated herpesvirus (KSHV) causes Kaposi’s sarcoma, primary effusion lymphoma, and multicentric Castleman disease. The tegument is a structure that is unique to herpesviruses that includes host and viral proteins, including the viral ORF42 and ORF55 proteins. Alphaherpesvirus tegument proteins have been well studied, but much is unknown regarding KSHV. Here, we report an interaction between the ORF42 and ORF55 proteins. ORF55 interacted with and recruited ORF42 from the nucleus to the cytoplasm. When ORF42 and ORF55 were expressed simultaneously in cultured cells, the expression level of these two viral proteins was higher than when either was expressed independently. ORF55, but not ORF42, was polyubiquitinated, suggesting that an unidentified regulatory mechanism may be present. A recombinant virus with an ectopic stop codon in ORF42 exhibited normal replication of genomic DNA, but fewer virus particles were released with the recombinant than with the wild-type virus. A unique R136Q mutation in ORF42, which is found in a KSHV strain that is prevalent on Miyako Island, Okinawa Prefecture, Japan, further increased the expression of ORF42 and ORF55 when these proteins were expressed simultaneously. However, the ORF42 R136Q mutation did not affect the localization pattern of ORF42 itself or of ORF55. In addition, experiments with a recombinant virus possessing the ORF42 R136Q mutation showed lower levels of production of the mutant virus than of the wild-type virus, despite similar levels of genome replication. We suggest that the R136Q mutation in ORF42 plays an important role in ORF55 protein expression and virus production.








Similar content being viewed by others
Data availability
No publicly available datasets were included in this study.
References
Krug LT, Pellet PE (2022) The family Herpesviridae: A Brief introduction. In: Knipe DM, Howley PM, Damania BA, Cohen JI (eds) Fields Virology, vol 2, 7th edn. Lippincott Williams & Wilkins, pp 212–234
Chang Y, Cesarman E, Pessin MS, Lee F, Culpepper J, Knowles DM, Moore PS (1994) Identification of herpesvirus-like DNA sequences in AIDS-associated Kaposi’s sarcoma. Science 266:1865–1869. https://doi.org/10.1126/science.7997879
Soulier J, Grollet L, Oksenhendler E, Cacoub P, Cazals-Hatem D, Babinet P, d’Agay MF, Clauvel JP, Raphael M, Degos L et al (1995) Kaposi’s sarcoma-associated herpesvirus-like DNA sequences in multicentric Castleman’s disease. Blood 86:1276–80. https://doi.org/10.1182/blood.V86.4.1276.bloodjournal8641276
Nador RG, Cesarman E, Chadburn A, Dawson DB, Ansari MQ, Sald J, Knowles DM (1996) Primary effusion lymphoma: a distinct clinicopathologic entity associated with the Kaposi’s sarcoma-associated herpes virus. Blood 88:645–56. https://doi.org/10.1182/blood.V88.2.645.bloodjournal882645
Damania BA, Cesarman E (2022) Kaposi’s Sarcoma Herpesvirus. In: Knipe DM, Howley PM, Damania BA, Cohen JI (eds) Fields Virology, vol 2, 7th edn. Lippincott Williams & Wilkins, pp 513–572
Decker LL, Shankar P, Khan G, Freeman RB, Dezube BJ, Lieberman J, Thorley-Lawson DA (1996) The Kaposi sarcoma-associated herpesvirus (KSHV) is present as an intact latent genome in KS tissue but replicates in the peripheral blood mononuclear cells of KS patients. J Exp Med 184:283–288. https://doi.org/10.1084/jem.184.1.283
Sun R, Lin SF, Staskus K, Gradoville L, Grogan E, Haase A, Miller G (1999) Kinetics of Kaposi’s sarcoma-associated herpesvirus gene expression. J Virol 73:2232–42. https://doi.org/10.1128/jvi.73.3.2232-2242.1999
Jenner RG, Alba MM, Boshoff C, Kellam P (2001) Kaposi’s sarcoma-associated herpesvirus latent and lytic gene expression as revealed by DNA arrays. J Virol 75:891–902. https://doi.org/10.1128/jvi.75.2.891-902.2001
Arias C, Weisburd B, Stern-Ginossar N, Mercier A, Madrid AS, Bellare P, et al. KSHV 2.0: A Comprehensive Annotation of the Kaposi’s Sarcoma-Associated Herpesvirus Genome Using Next-Generation Sequencing Reveals Novel Genomic and Functional Features. PLoS Pathog 2014;10:1003847. https://doi.org/10.1371/journal.ppat.1003847.
Nabiee R, Syed B, Castano JR, Lalani R, Totonchy JE. An update of the virion proteome of kaposi sarcoma-associated herpesvirus. Viruses 2020;12. https://doi.org/10.3390/v12121382.
Zhu FX, Chong JM, Wu L, Yuan Y. Virion Proteins of Kaposi’s Sarcoma-Associated Herpesvirus. J Virol 2005;79:800. https://doi.org/10.1128/JVI.79.2.800-811.2005.
Butnaru M, Gaglia MM. The kaposi’s sarcoma-associated herpesvirus protein orf42 is required for efficient virion production and expression of viral proteins. Viruses 2019;11. https://doi.org/10.3390/v11080711.
Liang Q, Wei D, Chung B, Brulois KF, Guo C, Dong S, et al. Novel Role of vBcl2 in the Virion Assembly of Kaposi’s Sarcoma-Associated Herpesvirus. J Virol 2018;92. https://doi.org/10.1128/JVI.00914-17.
McGeoch DuncanJ, Davison AJ. The Molecular Evolutionary History of the Herpesviruses. Origin and Evolution of Viruses 1999:441–65. https://doi.org/10.1016/B978-012220360-2/50018-0.
Longnecker R, Neipel F. Introduction to the human γ-herpesviruses. Human Herpesviruses: Biology, Therapy, and Immunoprophylaxis 2007:341–59. https://doi.org/10.1017/CBO9780511545313.023.
Yanagi Y, Abdullah Al Masud HM, Watanabe T, Sato Y, Goshima F, Kimura H, et al. Initial Characterization of the Epstein–Barr Virus BSRF1 Gene Product. Viruses 2019;11. https://doi.org/10.3390/V11030285.
Nozawa N, Daikoku T, Koshizuka T, Yamauchi Y, Yoshikawa T, Nishiyama Y. Subcellular Localization of Herpes Simplex Virus Type 1 UL51 Protein and Role of Palmitoylation in Golgi Apparatus Targeting. J Virol 2003;77:3204–16. https://doi.org/10.1128/JVI.77.5.3204-3216.2003.
Albecka A, Owen DJ, Ivanova L, Brun J, Liman R, Davies L, et al. Dual Function of the pUL7-pUL51 Tegument Protein Complex in Herpes Simplex Virus 1 Infection. J Virol 2017;91. https://doi.org/10.1128/JVI.02196-16.
Masud HMA Al, Yanagi Y, Watanabe T, Sato Y, Kimura H, Murata T. Epstein-Barr Virus BBRF2 Is Required for Maximum Infectivity. Microorganisms 2019;7. https://doi.org/10.3390/MICROORGANISMS7120705.
Watanabe T, Nishimura M, Izumi T, Kuriyama K, Iwaisako Y, Hosokawa K, et al. Kaposi’s Sarcoma-Associated Herpesvirus ORF66 Is Essential for Late Gene Expression and Virus Production via Interaction with ORF34. J Virol 2020;94. https://doi.org/10.1128/jvi.01300-19.
Brulois KF, Chang H, Lee AS-Y, Ensser A, Wong L-Y, Toth Z, et al. Construction and Manipulation of a New Kaposi’s Sarcoma-Associated Herpesvirus Bacterial Artificial Chromosome Clone. J Virol 2012;86:9708–20. https://doi.org/10.1128/JVI.01019-12/SUPPL_FILE/ZJV999096466SO1.PDF.
Nishimura M, Watanabe T, Yagi S, Yamanaka T, Fujimuro M. Kaposi’s sarcoma-associated herpesvirus ORF34 is essential for late gene expression and virus production. Sci Rep 2017;7. https://doi.org/10.1038/S41598-017-00401-7.
Wakao K, Watanabe T, Takadama T, Ui S, Shigemi Z, Kagawa H, et al. Sangivamycin induces apoptosis by suppressing Erk signaling in primary effusion lymphoma cells. Biochem Biophys Res Commun 2014;444:135–40. https://doi.org/10.1016/J.BBRC.2014.01.017.
Watanabe T, Nakamura S, Ono T, Ui S, Yagi S, Kagawa H, et al. Pyrrolidinium fullerene induces apoptosis by activation of procaspase-9 via suppression of Akt in primary effusion lymphoma. Biochem Biophys Res Commun 2014;451:93–100. https://doi.org/10.1016/J.BBRC.2014.07.068.
Roller RJ, Fetters R. The Herpes Simplex Virus 1 UL51 Protein Interacts with the UL7 Protein and Plays a Role in Its Recruitment into the Virion. J Virol 2015;89:3112–22. https://doi.org/10.1128/JVI.02799-14.
Erales J, Coffino P. Ubiquitin-independent proteasomal degradation. Biochimica et Biophysica Acta (BBA) - Molecular Cell Research 2014;1843:216–21. https://doi.org/10.1016/J.BBAMCR.2013.05.008.
Sander G, Konrad A, Thurau M, Wies E, Leubert R, Kremmer E, et al. Intracellular Localization Map of Human Herpesvirus 8 Proteins. J Virol 2008;82:1908–22. https://doi.org/10.1128/jvi.01716-07.
Awazawa R, Utsumi D, Katano H, Awazawa T, Miyagi T, Hayashi K, et al. High Prevalence of Distinct Human Herpesvirus 8 Contributes to the High Incidence of Non-acquired Immune Deficiency Syndrome-Associated Kaposi’s Sarcoma in Isolated Japanese Islands. Journal of Infectious Diseases 2017;216:850–8. https://doi.org/10.1093/infdis/jix424.
Karsten Tischer B, Smith GA, Osterrieder N. En passant mutagenesis: A Two Markerless red recombination system. Methods in Molecular Biology 2010;634:421–30. https://doi.org/10.1007/978-1-60761-652-8_30.
Xu X, Guo Y, Fan S, Cui P, Feng M, Wang L, et al. Attenuated phenotypes and analysis of a herpes simplex virus 1 strain with partial deletion of the UL7, UL41 and LAT genes. Virol Sin 2017;32:404–14. https://doi.org/10.1007/S12250-017-3947-1/METRICS.
Roller RJ, Haugo AC, Yang K, Baines JD. The Herpes Simplex Virus 1 UL51 Gene Product Has Cell Type-Specific Functions in Cell-to-Cell Spread. J Virol 2014;88:4058–68. https://doi.org/10.1128/jvi.03707-13.
Oda S, Arii J, Koyanagi N, Kato A, Kawaguchi Y. The Interaction between Herpes Simplex Virus 1 Tegument Proteins UL51 and UL14 and Its Role in Virion Morphogenesis. J Virol 2016;90:8754. https://doi.org/10.1128/JVI.01258-16.
Acknowledgements
The BAC16 KSHV clone was a gift from Kevin Brulois and Jae U. Jung (USC, USA). We thank Gregory A. Smith (Northwestern University, USA) for Escherichia coli strain GS1783, and Nikolaus Osterrieder (Cornell University, USA) for the plasmid pEP-KanS. K.K. was supported by a Nagai Memorial Research Scholarship from the Pharmaceutical Society of Japan. We would like to thank Editage (www.editage.jp) for English language editing.
Funding
This study was supported by a research grant from the Okinawa Medical Science Research Foundation (to Kazushi Kuriyama).
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conception and design. Experiments were conducted by Kazushi Kuriyama and Tadashi Watanabe. All authors interpreted the data. Kazushi Kuriyama wrote, and all authors commented on, the first draft of the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Ethical approval
Experiments using human or animal subjects were not included.
Conflict of interest
The authors declare no conflict of interest.
Additional information
Handling Editor: Akbar Dastjerdi
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Kuriyama, K., Watanabe, T. & Ohno, S. Analysis of the interaction between the ORF42 and ORF55 proteins encoded by Kaposi’s sarcoma-associated herpesvirus. Arch Virol 169, 98 (2024). https://doi.org/10.1007/s00705-024-06021-7
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00705-024-06021-7