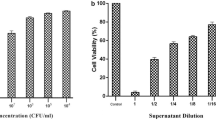
Abstract
Background
The excessive usage of chlorhexidine gluconate (CHG) as a broad-spectrum antimicrobial reagent can have a negative impact on the environment and on human health. The aim of this study was to investigate the effectiveness of some plant-derived compounds in reducing the CHG concentration required to exert its antiviral activity.
Methods
Antiviral assays were conducted according to EN 14476 (2019) against herpes simplex virus type 1 (HSV-1), H1N1 influenza A virus, and adenovirus type 5 (Ad-5) as enveloped and non-enveloped viral models to assess the synergistic interaction of CHG and natural additive compounds.
Results
The effective concentration of 0.247 mM CHG against HSV-1 was decreased tenfold in combination with 0.0125 mM salicylic acid, with a titer reduction of 1.47 ⋅ 104 CCID50/ml. The time required for complete inactivation of HSV-1 particles was reduced to 15 min when the virus was exposed to 0.061 mM CHG and 0.0125 mM salicylic acid. Additionally, the presence of salicylic acid protected the CHG activity against interfering substances.
Conclusion
Our supplemented CHG formulation showed immediate antiviral effectiveness, which is important for management of the infections. CHG can be combined with salicylic acid to exhibit synergistic antiviral activity at lower concentrations and reduce the time required for inactivation. Furthermore, in the presence of interfering substances, the combination has higher antiviral activity than CHG alone.









Similar content being viewed by others
References
Kampf G (2018) Antiseptic stewardship: Biocide resistance and clinical implications.
Babich H, Wurzburger BJ, Rubin YL (1995) An in vitro study on the cytotoxicity of chlorhexidine digluconate to human gingival cells. Cell Biol Toxicol 11:79–88. https://doi.org/10.1016/j.fct.2015.02.018
McDonnell G, Russell D (1999) Antiseptics and Disinfectants: Activity, Action, and Resistance. Am Soc Microbiol 12(1):147–179
Karpiński TM, Szkaradkiewicz AK (2015) Chlorhexidine - Pharmaco-biological activity and application. Eur Rev Med Pharmacol Sci 19(7):1321–1326
Lachenmeier DW (2019) Antiseptic drugs and disinfectants, vol 41, 1st edn. Elsevier B.V.
Wyganowska-Swiatkowska M, Kotwicka M, Urbaniak P, Nowak A, Skrzypczak-Jankun E, Jankun J (2016) Clinical implications of the growth-suppressive effects of chlorhexidine at low and high concentrations on human gingival fibroblasts and changes in morphology. Int J Mol Med 37(6):1594–1600. https://doi.org/10.3892/ijmm.2016.2550
Saadatpour F, Mohammadipanah F (2021) Enhancement of bactericidal effect of Chlorhexidine using Choline augmentation as a natural additive. Am J Infect Control. https://doi.org/10.1016/j.ajic.2021.05.012
Hayat Q, Hayat S, Irfan M, Ahmad A (2010) Effect of exogenous salicylic acid under changing environment: A review. Environ Exp Bot 68(1):14–25. https://doi.org/10.1016/j.envexpbot.2009.08.005
Alyemeni MN, Ahmad A, Hayat S (2013) SALICYLIC ACID Plant Growth and Development.
“National Center for Biotechnology Information (2021) PubChem Compound Summary for CID 338, Salicylic acid. https://pubchem.ncbi.nlm.nih.gov/compound/Salicylic-acid. Accessed June 7, ”
Raskin I (1992) Role of salicylic acid in plants. Annu Rev Plant Physiol Plant Mol Biol 43(1):439–463. https://doi.org/10.1146/annurev.pp.43.060192.002255
Kukhanova MK, Korovina AN, Kochetkov SN (2014) Human herpes simplex virus: Life cycle and development of inhibitors. Biochem 79(13):1635–1652. https://doi.org/10.1134/S0006297914130124
Moscatto JA et al (2013) Antiviral Agents for Herpes Simplex Virus, 31(2).
Lynch JP, Fishbein M, Echavarria M, Ph D (2011) Adenovirus. InSeminars Respir Crit care Med 32(04):494–511
Boëlle PY, Ansart S, Cori A, Valleron AJ, /H1N1 (2009) “Transmission parameters of the A influenza virus pandemic: A review,” Influenza Other Respi. Viruses, 5(5):306–316, 2011, https://doi.org/10.1111/j.1750-2659.2011.00234.x
Sugiyono PD (2016) EN 14476. J Chem Inf Model 53(9):1689–1699. https://doi.org/10.1017/CBO9781107415324.004
Rabenau HF, Schwebke I, Blumel J (2020) Guideline for testing chemical disinfectants regarding their virucidal activity within the field of human medicine. Bundesgesundheitsbl 645–655. https://doi.org/10.1007/s00103-020-03115-w
“ISO 10993- (2009) 5 Biological evaluation of medical devices - Part 5: Tests for in vitro cytotoxicity - Third Edition, ISO,
Williamson DA, Carter GP, Howdena BP (2017) Current and Emerging Topical Antibacterials and Antiseptics: Agents, Action, and Resistance Patterns. Am Soc Microbiol 30(3):827–860
Giannelli M (2008) Effect of chlorhexidine digluconate on different cell types: A molecular and ultrastructural investigation. Toxicol Vitr 22:308–317. https://doi.org/10.1016/j.tiv.2007.09.012
Lessa FCR, Aranha AMF, Nogueira I, Giro EMA, Hebling J, de Costa CA (2010) Toxicity of chlorhexidine on odontoblast-like cells. J Appl Oral Sci 18(1):50–58. https://doi.org/10.1590/S1678-77572010000100010
Cabral CT, Fernandes MH (2007) In vitro comparison of chlorhexidine and povidone – iodine on the long-term proliferation and functional activity of human alveolar bone cells. Clin Oral Investig 11:155–164. https://doi.org/10.1007/s00784-006-0094-8
Brauner A, Fridman O, Gefen O, Balaban NQ (2016) Distinguishing between resistance, tolerance and persistence to antibiotic treatment. Nat Rev Microbiol 14(5):320–330. https://doi.org/10.1038/nrmicro.2016.34
Webster RG, Govorkova EA (2015) Continuing challenges in influenza. Ann N Y Acad Sci 1323(1):115–139. https://doi.org/10.1111/nyas.12462.Continuing
Fernandez MdosS, Guedes MIF, Langa GPJ, Rösing CK, Cavagni J, Muniz FWMG (2022) Virucidal efficacy of chlorhexidine: a systematic review. Odontology 110(2):376–392. https://doi.org/10.1007/s10266-021-00660-x
Park JB, Park NH (1989) Effect of chlorhexidine on the in vitro and in vivo herpes simplex virus infection. Oral Surg Oral Med Oral Pathol 67(2):149–153. https://doi.org/10.1016/0030-4220(89)90320-4
Singh S, Sharma P, Kumar M (2020) Evaluation of the effects of 0.05% sodium hypochlorite and 0.12% chlorhexidine gluconate twice daily rinse on periodontal parameters and gingival crevicular fluid HSV1 and CMV levels in patients with chronic periodontitis: a multicentric study. Med J Armed Forces India xxxx:1–7. https://doi.org/10.1016/j.mjafi.2020.08.002
Bernstein D, Schiff G, Echler G (1989) In vitro Virucidal Effectiveness of a 0. 12%-Chlorhexidine Gluconate Mouthrinse, pp 874–876
Fernandez MdosS, Guedes MIF, Langa GPJ, Rösing CK, Cavagni J, Muniz FWMG (2021) Virucidal efficacy of chlorhexidine: a systematic review. Odontology no 012345678910.1007/s10266-021-00660-x
Park NH, Park JB, Min BM, Cherrick HM (1991) Combined synergistic antiherpetic effect of acyclovir and chlorhexidine in vitro. Oral Surg Oral Med Oral Pathol 71(2):193–196. https://doi.org/10.1016/0030-4220(91)90467-Q
Acknowledgments
This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that this research was conducted in the absence of any financial relationships that could become a potential conflict of interest.
Additional information
Communicated by Graciela Andrei
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Jamshidinia, N., Saadatpour, F., Arefian, E. et al. Augmented antiviral activity of chlorhexidine gluconate on herpes simplex virus type 1, H1N1 influenza A virus, and adenovirus in combination with salicylic acid. Arch Virol 168, 302 (2023). https://doi.org/10.1007/s00705-023-05932-1
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00705-023-05932-1