Abstract
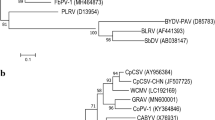
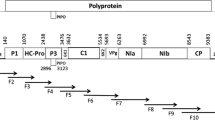
This study examined the natural and experimental host range and aphid and graft transmission of the tentative polerovirus phasey bean mild yellows virus (PBMYV). Eleven complete coding sequences from PBMYV isolates were determined from a range of hosts and locations. We found two genetically distinct variants of PBMYV. PBMYV-1 was the originally described variant, and PBMYV-2 had a large putative recombination in open reading frame 5 such that PBMYV-1 and PBMYV-2 shared only 65-66% amino acid sequence identity in the P5 protein. The virus was transmitted by a clonal colony of cowpea aphids (Aphis craccivora) and by grafting with infected scions but was not transmitted by a clonal colony of green peach aphids (Myzus persicae). PBMYV was found in natural infections in 11 host species with a range of symptoms and severity, including seven important grain legume crops from across a wide geographic area in Australia. PBMYV was common and widespread in the tropical weed phasey bean (Macroptilium lathyroides), but it is likely that there are other major alternative hosts for the virus in temperate regions of Australia. The experimental host range of PBMYV included the Fabaceae hosts chickpea (Cicer arietinum), faba bean (Vicia faba), pea (Pisum sativum), and phasey bean, but transmissions failed to infect several other members of the families Asteraceae, Cucurbitaceae, Fabaceae and Solanaceae. PBMYV was commonly found in grain legume crops in eastern and western Australia, sometimes at greater than 90% incidence. This new knowledge about PBMYV warrants further assessments of its economic impact on important grain legume crops.




Similar content being viewed by others
Availability of data and materials
Genome sequence data have been deposited in the GenBank database. Reference virus isolates are lodged in the Plant Virus Collection, Department of Agriculture and Fisheries, Queensland.
References
Abraham AD, Menzel W, Lesemann DE, Varrelmann M, Vetten HJ (2006) Chickpea chlorotic stunt virus: a new polerovirus infecting cool-season food legumes in ethiopia. Phytopathology 96:437–446
Altschul SF, Gish W, Miller W, Myers EW, Lipman DJ (1990) Basic local alignment search tool. J Mol Biol 215:403–410
Anonymous (2020) Agricultural commodities and trade data. Department of Agriculture, Water and the Environment
Bawden FC, Roberts FM (1947) The influence of light intensity on the susceptibility of plants to certain viruses. Ann Appl Biol 34:286–296
Brault V, Bergdoll M, Mutterer J, Prasad V, Pfeffer S, Erdinger M, Richards KE, Ziegler-Graff V (2003) Effects of point mutations in the major capsid protein of beet western yellows virus on capsid formation, virus accumulation, and aphid transmission. J Virol 77:3247–3256
Brault V, Périgon S, Reinbold C, Erdinger M, Scheidecker D, Herrbach E, Richards K, Ziegler-Graff V (2005) The polerovirus minor capsid protein determines vector specificity and intestinal tropism in the aphid. J Virol 79:9685–9693
Correa R, Silva T, Simoes-Araujo J, Barroso P, Vidal M, Vaslin M (2005) Molecular characterization of a virus from the family Luteoviridae associated with cotton blue disease. Arch Virol 150:1357–1367
D’Arcy CJ, Mayo M (1997) Proposals for changes in luteovirus taxonomy and nomenclature. Adv Virol 142:1285–1287
Edgar RC (2004) MUSCLE: multiple sequence alignment with high accuracy and high throughput. Nucleic Acids Res 32(5):1792–1797
Ellis MH, Silva TF, Stiller WN, Wilson LJ, Vaslin MFS, Sharman M, Llewellyn DJ (2013) Identification of a new Polerovirus (family Luteoviridae) associated with cotton bunchy top disease in Australia. Australas Plant Pathol 42:261–269
Fiallo-Olivé E, Navas-Hermosilla E, Ferro CG, Zerbini FM, Navas-Castillo J (2018) Evidence for a complex of emergent poleroviruses affecting pepper worldwide. Adv Virol 163:1171–1178
Filardo F, Nancarrow N, Kehoe MA, McTaggart AR, Congdon B, Kumari S, Aftab M, Trebicki P, Rodoni B, Thomas JE, Sharman M (2021) Genomic diversity and recombination between turnip yellows viruses in Australia. Arch Virol 166:813–829
Filardo FF, Sharman M (2019) Siratro latent polerovirus (SLPV): a new polerovirus from Australia with a non-functional open reading frame 0. Australas Plant Pathol 48:491–501
Filardo FF, Thomas JE, Webb M, Sharman M (2019) Faba bean polerovirus 1 (FBPV-1); a new polerovirus infecting legume crops in Australia. Adv Virol 164:1915–1921
Gibbs AJ, Gower JC (1960) The use of multiple-transfer method in plant virus transmission studies—some statistical points arising in the analysis of results. Ann Appl Biol 48:75–83
Gibbs MJ, Cooper JI (1995) A recombinational event in the history of Luteoviruses probably induced by base-pairing between the genomes of two distinct viruses. Virology 206:1129–1132
Guyader S, Ducray DG (2002) Sequence analysis of Potato leafroll virus isolates reveals genetic stability, major evolutionary events and differential selection pressure between overlapping reading frame products. J Gen Virol 83:1799–1807
Hadfield J, Thomas JE, Schwinghamer MW, Kraberger S, Stainton D, Dayaram A, Parry JN, Pande D, Martin DP, Varsani A (2012) Molecular characterisation of dicot-infecting mastreviruses from Australia. Virus Res 166:13–22
Herrbach E (1999) Vector-virus interaction. In: Smith HG, Barker H (eds) The luteoviridae. CAB International, Wallingford, pp 84–146
Hogenhout SA, Ammar E-D, Whitfield AE, Redinbaugh MG (2008) Insect vector interactions with persistently transmitted viruses. Annu Rev Phytopathol 46:327–359
Huson DH, Beier S, Flade I, Górska A, El-Hadidi M, Mitra S, Ruscheweyh H-J, Tappu R (2016) MEGAN community edition—interactive exploration and analysis of large-scale microbiome sequencing data. PLOS Comput Biol 12:e1004957
Jones RAC, Coutts BA (1996) Alfalfa mosaic and cucumber mosaic virus infection in chickpea and lentil: incidence and seed transmission. Ann Appl Biol 129:491–506
Katul L (1992) Characterization by serology and molecular biology of bean leaf roll virus and faba bean necrotic yellows virus. University of Göttingen, Göttingen, p 115
Kehoe MA, Coutts BA, Buirchell BJ, Jones RAC (2014) Plant virology and next generation sequencing: experiences with a Potyvirus. PLoS ONE 9:e104580
King AMQ, Adams MJ, Carstens EB, Lefkowitz EJ (2012) Virus taxonomy: ninth report of the international committee on taxonomy of viruses. Elsevier Acedemic Press, San Diego
Knierim D, Deng TC, Tsai WS, Green SK, Kenyon L (2010) Molecular identification of three distinct Polerovirus species and a recombinant Cucurbit aphid-borne yellows virus strain infecting cucurbit crops in Taiwan. Plant Pathol 59:991–1002
Kumar S, Stecher G, Tamua K (2016) MEGA7: molecular evolutionary genetics analysis version 7.0 for bigger datasets. Mol Biol Evol 33:1870–1874
Latham LJ, Jones RAC (2001) Incidence of virus infection in experimental plots, commercial crops, and seed stocks of cool season crop legumes. Aust J Agric Res 52:397–413
Lotos L, Maliogka VI, Katis NI (2016) New poleroviruses associated with yellowing symptoms in different vegetable crops in Greece. Arch Virol 161:431–436
Makkouk KM, Comeau A (1994) Evaluation of various methods for the detection of barley yellow dwarf virus by the tissue-blot immunoassay and its use for virus detection in cereals inoculated at different growth stages. Eur J Plant Pathol 100:71–80
Martin DP, Posada D, Crandall KA, Williamson C (2005) A modified Bootscan algorithm for automated identification of recombinant sequences and recombination breakpoints. AIDS Res Hum Retroviruses 21:98–102
Martin DP, Williamson C, Posada D (2005) RDP2: recombination detection and analysis from sequence alignments. Bioinformatics 21:260–262
Martin DP, Murrell B, Golden M, Khoosal A, Muhire B (2015) RDP4: Detection and analysis of recombination patterns in virus genomes. Virus Evol 1:1–5
Mayo MA, Miller WA (1999) The structure and expression of luteovirus genomes. In: Smith HG, Barker H (eds) The Luteoviridae. CAB International, Wallingford, pp 23–42
Moonan F, Molina J, Mirkov TE (2000) Sugarcane yellow leaf virus: an emerging virus that has evolved by recombination between luteoviral and poleroviral ancestors. Virology 269:156–171
Najar A, Kumari SG, Attar N, Lababidi S (2011) Present status of some virus diseases affecting legume crops in Tunisia, and partial characterization of Chickpea chlorotic stunt virus. Phytopathol Mediterr 50:310–315
Padidam M, Sawyer S, Fauquet CM (1999) Possible emergence of new geminiviruses by frequent recombination. Virology 265:218–225
Pagan I, Holmes EC (2010) Long-term evolution of the Luteoviridae: time scale and mode of virus speciation. J Virol 84:6177–6187
Palanga E, Martin DP, Galzi S, Zabré J, Bouda Z, Neya JB, Sawadogo M, Traore O, Peterschmitt M, Roumagnac P, Filloux D (2017) Complete genome sequences of cowpea polerovirus 1 and cowpea polerovirus 2 infecting cowpea plants in Burkina Faso. Adv Virol 162:2149–2152
Plchova H, Cerovska N, Moravec T, Dedic P (2009) Short communication: molecular analysis of Potato leafroll virus isolates from the Czech Republic. Virus Genes 39:153–155
Rathjen JP, Karageorgos LE, Habili N, Waterhouse PM, Symons RH (1994) Soybean dwarf luteovirus contains the third variant genome type in the luteovirus group. Virology 198:671–679
Robinson JT, Thorvaldsdóttir H, Winckler W, Guttman M, Lander ES, Getz G, Mesirov JP (2011) Integrative genomics viewer. Nat Biotechnol 29:24–26
Schwinghamer MW, Johnstone GR, Johnston-Lord CE (1999) First records of bean leafroll luteovirus in Australia. Australas Plant Pathol 28:260–260
Sharman M, Wilson CR, Thomas JE (2012) A distinct polerovirus infecting crop and weed legumes from eastern Australia is transmitted by cowpea aphid. 10th Australasian Plant Virology Workshop, Hanmer Springs, New Zealand
Sharman M, Thomas JE, Persley DM (2015) Natural host range, thrips and seed transmission of distinct Tobacco streak virus strains in Queensland, Australia. Ann Appl Biol 167:197–207
Sharman M, Kehoe M, Coutts B, van Leur J, Filardo F, Thomas J (2016) Two complete genome sequences of phasey bean mild yellows virus, a novel member of the luteoviridae from Australia. Genome Announc 4:e01569-e11515
Smirnova E, Firth AE, Miller WA, Scheidecker D, Brault V, Reinbold C, Rakotondrafara AM, Chung BYW, Ziegler-Graff V (2015) Discovery of a small non-AUG-initiated ORF in poleroviruses and luteoviruses that is required for long-distance movement. PLoS Pathogy 11:e1004868
Smith JM (1992) Analyzing the mosaic structure of genes. J Mol Evol 34:126–129
Stevens M, Smith HG, Hallsworth PB (1994) The host range of beet yellowing viruses among common arable weed species. Plant Pathol 43:579–588
Thomas JE, Schwinghamer MW, Parry JN, Sharman M, Schilg MA, Dann EK (2004) First report of Tomato spotted wilt virus infecting chickpea (Circer arietinum) in Australia. Australas Plant Pathol 33:597–599
Thompson JD, Higgins DG, Gibson TJ (1994) CLUSTAL W: improving the sensitivity of progressive multiple sequence alignment through sequence weighting, position-specific gap penalties and weight matrix choice. Nucleic Acids Res 22:4673–4680
Wang F, Zhou BG, Gao ZL, Xu DF (2016) A new species of the genus polerovirus causing symptoms similar to maize yellow dwarf virus-RMV of maize in China. Plant Dis 100:1508–1508
Wilson CR, Jones RAC (1992) Resistance to phloem transport of potato leafroll virus in potato plants. J Gen Virol 73:3219–3224
Wilson CR, Lambert SJ, Dann AL, Cross P, Hay FS (2012) Occurrence of viruses within Tasmanian vegetable crops and identification of a novel Polerovirus infecting pea. Australas Plant Pathol 41:311–319
Wilson CR (2014) Applied plant virology. CAB International, Wallingford
Wylie S, Wilson CR, Jones RAC, Jones MGK (1993) A polymerase chain reaction assay for cucumber mosaic virus in Lupin seeds. Aust J Agric Res 44:41–51
Xiang H-Y, Dong S-W, Shang Q-X, Zhou C-J, Li D-W, Yu J-L, Han C-G (2011) Molecular characterization of two genotypes of a new polerovirus infecting brassicas in China. Adv Virol 156:2251–2255
Xiang Y, Bernardy M, Bhagwat B, Wiersma PA, DeYoung R, Bouthillier M (2015) The complete genome sequence of a new polerovirus in strawberry plants from eastern Canada showing strawberry decline symptoms. Adv Virol 160:553–556
Zhang X-Y, Xiang H-Y, Zhou C-J, Li D-W, Yu J-L, Han C-G (2014) Complete genome sequence analysis identifies a new genotype of brassica yellows virus that infects cabbage and radish in China. Adv Virol 159:2177–2180
Acknowledgements
We gratefully acknowledge the contributions of Dr. Alison Dann, Mr. Peter Cross, Mr. Shane Hossel, and Dr. Tamilarasan Thangavel. Dr. Joop van Leur, Dr. John Thomas, and Ms. Judy Parry kindly provided virus-infected samples for this study.
Funding
This work was supported by the Department of Agriculture and Fisheries, Queensland, the Tasmanian Institute of Agriculture, and funds from the Australian Grains Research and Development Corporation projects DAQ00186 and DAN00202. Dr. Appiah was recipient of an Australian Development Scholarship provided by AusAID.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflicts of interest.
Ethical approval
All applicable international, national, and/or institutional guidelines for the care and use of animals were followed.
Additional information
Handling Editor: Elvira Fiallo-Olivé.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Sharman, M., Appiah, A.S., Filardo, F. et al. Biology and genetic diversity of phasey bean mild yellows virus, a common virus in legumes in Australia. Arch Virol 166, 1575–1589 (2021). https://doi.org/10.1007/s00705-021-05022-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00705-021-05022-0