Abstract
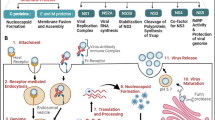
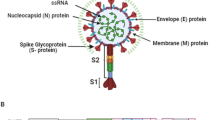
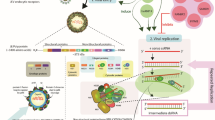
Chikungunya virus (CHIKV), a virus that induces pathogenic inflammatory host immune responses, is re-emerging worldwide, and there are currently no established antiviral control measures. Transient receptor potential vanilloid 1 (TRPV1), a non-selective Ca2+-permeable ion channel, has been found to regulate various host inflammatory responses including several viral infections. Immune responses to CHIKV infection in host macrophages have been reported recently. However, the possible involvement of TRPV1 during CHIKV infection in host macrophages has not been studied. Here, we investigated the possible role of TRPV1 in CHIKV infection of the macrophage cell line RAW 264.7. It was found that CHIKV infection upregulates TRPV1 expression in macrophages. To confirm this observation, the TRPV1-specific modulators 5ˊ-iodoresiniferatoxin (5ˊ-IRTX, a TRPV1 antagonist) and resiniferatoxin (RTX, a TRPV1 agonist) were used. Our results indicated that TRPV1 inhibition leads to a reduction in CHIKV infection, whereas TRPV1 activation significantly enhances CHIKV infection. Using a plaque assay and a time-of-addition assay, it was observed that functional modulation of TRPV1 affects the early stages of the viral lifecycle in RAW 264.7 cells. Moreover, CHIKV infection was found to induce of pNF-κB (p65) expression and nuclear localization. However, both activation and inhibition of TRPV1 were found to enhance the expression and nuclear localization of pNF-κB (p65) and production of pro-inflammatory TNF and IL-6 during CHIKV infection. In addition, it was demonstrated by Ca2+ imaging that TRPV1 regulates Ca2+ influx during CHIKV infection. Hence, the current findings highlight a potentially important regulatory role of TRPV1 during CHIKV infection in macrophages. This study might also have broad implications in the context of other viral infections as well.







Similar content being viewed by others
References
Tominaga M, Tominaga T (2005) Structure and function of TRPV1. Pflugers Arch 451:143–150. https://doi.org/10.1007/s00424-005-1457-8
Ortíz-Rentería M, Juárez-Contreras R, González-Ramírez R et al (2018) TRPV1 channels and the progesterone receptor Sig-1R interact to regulate pain. Proc Natl Acad Sci. https://doi.org/10.1073/pnas.1715972115
Choi JY, Lee HY, Hur J et al (2018) TRPV1 blocking alleviates airway inflammation and remodeling in a chronic asthma murine model. Allergy Asthma Immunol Res 10:216–224. https://doi.org/10.4168/aair.2018.10.3.216
Majhi RK, Sahoo SS, Yadav M et al (2015) Functional expression of TRPV channels in T cells and their implications in immune regulation. FEBS J 282:2661–2681. https://doi.org/10.1111/febs.13306
Yang F, Ma L, Cao X et al (2014) Divalent cations activate TRPV1 through promoting conformational change of the extracellular region. J General Physiol 143:91–103. https://doi.org/10.1085/jgp.201311024
Ramirez G, Coletto L, Sciorati C et al (2018) Ion channels and transporters in inflammation: special focus on TRP channels and TRPC6. Cells 7:70. https://doi.org/10.3390/cells7070070
Ramsey IS, Delling M, Clapham DE (2006) An introduction to TRP channels. Annu Rev Physiol 68:619–647. https://doi.org/10.1146/annurev.physiol.68.040204.100431
Gees M, Colsoul B, Nilius B (2010) The role of transient receptor potential cation channels in Ca2+ signaling. Cold Spring Harbor Perspect Biol 2:a003962–a003962. https://doi.org/10.1101/cshperspect.a003962
Vay L, Gu C, McNaughton PA (2012) The thermo-TRP ion channel family: Properties and therapeutic implications. Br J Pharmacol 165:787–801. https://doi.org/10.1111/j.1476-5381.2011.01601.x
Venkatachalam K, Montell C (2007) TRP channels. Ann Rev Biochem 76:387–417. https://doi.org/10.1146/annurev.biochem.75.103004.142819
Smani T, Shapovalov G, Skryma R et al (2015) Functional and physiopathological implications of TRP channels. Biochimica et Biophysica Acta - Molecular Cell Research 1853:1772–1782. https://doi.org/10.1016/j.bbamcr.2015.04.016
Pla AF, Gkika D (2013) Emerging role of TRP channels in cell migration: From tumor vascularization to metastasis. Front Physiol. https://doi.org/10.3389/fphys.2013.00311
Sukumaran P, Schaar A, Sun Y, Singh BB (2016) Functional role of TRP channels in modulating ER stress and Autophagy. Cell Calcium 60:123–132. https://doi.org/10.1016/j.ceca.2016.02.012
Lishko PV, Procko E, Jin X et al (2007) The Ankyrin repeats of TRPV1 bind multiple ligands and modulate channel sensitivity. Neuron 54:905–918. https://doi.org/10.1016/j.neuron.2007.05.027
Devesa I, Planells-Cases R, Fernández-Ballester G et al (2011) Role of the transient receptor potential vanilloid 1 in inflammation and sepsis. J Inflamm Res 4:67–81. https://doi.org/10.2147/JIR.S12978
Tsuji F, Aono H (2012) Role of transient receptor potential vanilloid 1 in inflammation and autoimmune diseases. Pharmaceuticals 5:837–852. https://doi.org/10.3390/ph5080837
Fernandes ES, Fernandes MA, Keeble JE (2012) The functions of TRPA1 and TRPV1: moving away from sensory nerves. Br J Pharmacol 166:510–521. https://doi.org/10.1111/j.1476-5381.2012.01851.x
Facer P, Casula MA, Smith GD et al (2007) Differential expression of the capsaicin receptor TRPV1 and related novel receptors TRPV3, TRPV4 and TRPM8 in normal human tissues and changes in traumatic and diabetic neuropathy. BMC Neurol 7:1–12. https://doi.org/10.1186/1471-2377-7-11
Omari SA, Adams MJ, Geraghty DP (2017) TRPV1 channels in immune cells and hematological malignancies, 1st edn. Elsevier Inc.
Marrone MC, Morabito A, Giustizieri M et al (2017) TRPV1 channels are critical brain inflammation detectors and neuropathic pain biomarkers in mice. Nat Commun. https://doi.org/10.1038/ncomms15292
Yoshida A, Furube E, Mannari T et al (2016) TRPV1 is crucial for proinflammatory STAT3 signaling and thermoregulation-associated pathways in the brain during inflammation. Sci Rep 6:1–11. https://doi.org/10.1038/srep26088
Felix A, Rosa E a Gestão Documental Como Suporte Ao Governo Eletrônico : Caso Da Secretaria De Estado Da Saúde De Santa Catarina ( Ses / Sc ). 36–50
Silva RO, Bingana RD, Sales TMAL et al (2018) Role of TRPV1 receptor in inflammation and impairment of esophageal mucosal integrity in a murine model of nonerosive reflux disease. Neurogastroenterol Motil 30:1–8. https://doi.org/10.1111/nmo.13340
Toledo-Mauriño JJ, Furuzawa-Carballeda J, Villeda-Ramírez MA et al (2018) The transient receptor potential vanilloid 1 is associated with active inflammation in ulcerative colitis. Mediators Inflamm 2018:1–7. https://doi.org/10.1155/2018/6570371
Kitagawa Y, Tamai I, Hamada Y et al (2013) Orally administered selective TRPV1 antagonist, JTS-653, attenuates chronic pain refractory to non-steroidal anti-inflammatory drugs in rats and mice including post-herpetic pain. J Pharmacol Sci 122:128–137. https://doi.org/10.1254/jphs.12276FP
Abdullah H, Heaney LG, Cosby SL, McGarvey LPA (2014) Rhinovirus upregulates transient receptor potential channels in a human neuronal cell line: implications for respiratory virus-induced cough reflex sensitivity. Thorax 69:46–54. https://doi.org/10.1136/thoraxjnl-2013-203894
Han SB, Kim H, Hyun Cho S et al (2016) Transient receptor potential vanilloid-1 in epidermal keratinocytes may contribute to acute pain in herpes zoster. Acta Dermato-Venereologica 96:319–322. https://doi.org/10.2340/00015555-2247
Cabrera JR, Viejo-Borbolla A, Alcamí A, Wandosell F (2016) Secreted herpes simplex virus-2 glycoprotein G alters thermal pain sensitivity by modifying NGF effects on TRPV1. J Neuroinflamm 13:1–9. https://doi.org/10.1186/s12974-016-0677-5
Omar S, Clarke R, Abdullah H et al (2017) Respiratory virus infection up-regulates TRPV1, TRPA1 and ASICS3 receptors on airway cells. PLoS ONE 12:1–21. https://doi.org/10.1371/journal.pone.0171681
Harford TJ, Rezaee F, Scheraga RG et al (2018) Asthma predisposition and respiratory syncytial virus infection modulate transient receptor potential vanilloid 1 function in children’s airways. J Allergy Clin Immunol 141:414–416. https://doi.org/10.1016/j.jaci.2017.07.015 (e4)
Alhmada Y, Selimovic D, Murad F et al (2017) Hepatitis C virus-associated pruritus: etiopathogenesis and therapeutic strategies. World J Gastroenterol 23:743–750. https://doi.org/10.3748/wjg.v23.i5.743
Dupuis-Maguiraga L, Noret M, Brun S et al (2012) Chikungunya disease: Infection-associated markers from the acute to the chronic phase of arbovirus-induced arthralgia. PLoS Neglected Trop Dis. https://doi.org/10.1371/journal.pntd.0001446
Silva LA, Dermody TS (2017) Chikungunya virus: epidemiology, replication, disease mechanisms, and prospective intervention strategies. J Clin Investig 127:737–749
Tanabe ISB, Tanabe ELL, Santos EC, Martins WV (2018) Cellular and molecular immune response to Chikungunya virus infection. Front Cell Infect Microbiol 8:1–15. https://doi.org/10.3389/fcimb.2018.00345
Nayak TK, Mamidi P, Kumar A et al (2017) Regulation of viral replication, apoptosis and pro-inflammatory responses by 17-aag during chikungunya virus infection in macrophages. Viruses. https://doi.org/10.3390/v9010003
Subudhi BB, Chattopadhyay S, Mishra P, Kumar A (2018) Current strategies for inhibition of Chikungunya infection. Viruses 10
Franken L, Schiwon M, Kurts C (2016) Microreview Macrophages: sentinels and regulators of the immune system. Cell Microbiol 18:475–487. https://doi.org/10.1111/cmi.12580
Pratheek BM, Suryawanshi AR, Chattopadhyay S, Chattopadhyay S (2015) In silico analysis of MHC-I restricted epitopes of Chikungunya virus proteins: Implication in understanding anti-CHIKV CD8+ T cell response and advancement of epitope based immunotherapy for CHIKV infection. Infect Genetics Evolut. https://doi.org/10.1016/j.meegid.2015.01.017
Nayak TK, Mamidi P, Sahoo SS et al (2019) P38 and JNK mitogen-activated protein kinases interact with Chikungunya virus non-structural protein-2 and regulate TNF induction during viral infection in macrophages. Front Immunol 10:1–15. https://doi.org/10.3389/fimmu.2019.00786
Bertin S, Aoki-Nonaka Y, De Jong PR et al (2014) The ion channel TRPV1 regulates the activation and proinflammatory properties of CD4+T cells. Nat Immunol 15:1055–1063. https://doi.org/10.1038/ni.3009
Khalil M, Alliger K, Weidinger C et al (2018) Functional role of transient receptor potential channels in immune cells and epithelia. Front Immunol 9:1–7
Fernandes ES, Liang L, Smillie S-J et al (2012) TRPV1 deletion enhances local inflammation and accelerates the onset of systemic inflammatory response syndrome. J Immunol 188:5741–5751. https://doi.org/10.4049/jimmunol.1102147
Clark N, Keeble J, Fernandes ES et al (2007) The transient receptor potential vanilloid 1 (TRPV1) receptor protects against the onset of sepsis after endotoxin. FASEB J 21:3747–3755. https://doi.org/10.1096/fj.06-7460com
Han SS, Keum YS, Chun KS, Surh YJ (2002) Suppression of phorbol ester-induced NF-κB activation by capsaicin in cultured human promyelocytic leukemia cells. Arch Pharmacal Res 25:475–479. https://doi.org/10.1007/BF02976605
Miyake T, Shirakawa H, Nakagawa T, Kaneko S (2015) Activation of mitochondrial transient receptor potential vanilloid 1 channel contributes to microglial migration. GLIA 63:1870–1882. https://doi.org/10.1002/glia.22854
Sappington RM, Calkins DJ (2008) Contribution of TRPV1 to microglia-derived IL-6 and NFκB translocation with elevated hydrostatic pressure. Invest Ophthalmol Vis Sci 49:3004–3017. https://doi.org/10.1167/iovs.07-1355
Wang Y, Wang DH (2009) Aggravated renal inflammatory responses in TRPV1 gene knockout mice subjected to DOCA-salt hypertension. Am J Physiol-Renal Physiol 297:F1550–F1559. https://doi.org/10.1152/ajprenal.00012.2009
Tsuji F, Murai M, Oki K et al (2010) Effects of SA13353, a transient receptor potential vanilloid 1 agonist, on leukocyte infiltration in lipopolysaccharide-induced acute lung injury and ovalbumin-induced allergic airway inflammation. J Pharmacol Sci 112:487–490. https://doi.org/10.1254/jphs.09295SC
Hsieh WS, Kung CC, Huang SL et al (2017) TDAG8, TRPV1, and ASIC3 involved in establishing hyperalgesic priming in experimental rheumatoid arthritis. Sci Rep 7:1–14. https://doi.org/10.1038/s41598-017-09200-6
Fernandes ES, Brito CXL, Teixeira SA et al (2014) TRPV1 antagonism by Capsazepine modulates innate immune response in mice infected with plasmodium berghei ANKA. Mediators Inflamm. https://doi.org/10.1155/2014/506450
Helyes Z, Elekes K, Nemeth J et al (2007) Role of transient receptor potential vanilloid 1 receptors in endotoxin-induced airway inflammation in the mouse. Am J Physiol Lung Cell Mol Physiol 292:L1173–L1181. https://doi.org/10.1152/ajplung.00406.2006
White JPM, Urban L, Nagy I (2011) TRPV1 function in health and disease. 130–144
Gouin O, L’Herondelle K, Lebonvallet N et al (2017) TRPV1 and TRPA1 in cutaneous neurogenic and chronic inflammation: pro-inflammatory response induced by their activation and their sensitization. Protein Cell 8:644–661. https://doi.org/10.1007/s13238-017-0395-5
Fox JM, Diamond MS (2016) Immune-mediated protection and pathogenesis of Chikungunya virus. J Immunol. https://doi.org/10.4049/jimmunol.1601426
Kulkarni SP, Ganu M, Jayawant P et al (2017) Regulatory T cells and IL-10 as modulators of chikungunya disease outcome: a preliminary study. Eur J Clin Microbiol Infect Dis 36:2475–2481. https://doi.org/10.1007/s10096-017-3087-4
Chattopadhyay SS, Kumar A, Mamidi P et al (2014) Development and characterization of monoclonal antibody against non-structural protein-2 of chikungunya virus and its applications. J Virol Methods 199:86–94. https://doi.org/10.1016/j.jviromet.2014.01.008
Elokely K, Velisetty P, Delemotte L et al (2016) Understanding TRPV1 activation by ligands: insights from the binding modes of capsaicin and resiniferatoxin. Proc Natl Acad Sci 113:E137–E145. https://doi.org/10.1073/pnas.1517288113
Kumar A, Mamidi P, Das I et al (2014) A Novel 2006 Indian outbreak strain of chikungunya virus exhibits different pattern of infection as compared to prototype strain. PLoS ONE. https://doi.org/10.1371/journal.pone.0085714
Saswat T, Kumar A, Kumar S et al (2015) High rates of co-infection of dengue and chikungunya virus in Odisha and Maharashtra, India during 2013. Infect Genetics Evol 35:134–141. https://doi.org/10.1016/j.meegid.2015.08.006
Raghavendhar S, Patel AK, Kabra SK et al (2019) Virus load and clinical features during the acute phase of Chikungunya infection in children. PLoS ONE 14:1–10. https://doi.org/10.1371/journal.pone.0211036
Bartholomeusz A, Locarnini S (2006) Associated with antiviral therapy. Antiviral Therapy 55:52–55. https://doi.org/10.1002/jmv
C A-U, Chen M, Hotta H (2018) Time-of-addition and temperature-shift assays to determine particular step(s) in the viral life cycle that is blocked by antiviral substances(s). Bio-protocol 8:1–12. https://doi.org/10.21769/BioProtoc.2830
Sahoo SS, Majhi RK, Tiwari A et al (2019) Transient Receptor Potential Ankyrin1 channel is endogenously expressed in T cells and is involved in immune functions. Biosci Rep. https://doi.org/10.1042/bsr20191437
Lapointe TK, Basso L, Iftinca MC et al (2015) TRPV1 sensitization mediates postinflammatory visceral pain following acute colitis. Am J Physiol 309:G87–G99. https://doi.org/10.1152/ajpgi.00421.2014
Lawton SK, Xu F, Tran A et al (2017) N-Arachidonoyl dopamine modulates acute systemic inflammation via nonhematopoietic TRPV1. J Immunol (Baltimore, Md: 1950) 199:1465–1475. https://doi.org/10.4049/jimmunol.1602151
Bassi MS, Gentile A, Iezzi E et al (2019) Transient receptor potential vanilloid 1 modulates central inflammation in multiple sclerosis. Front Neurol 10:1–8. https://doi.org/10.3389/fneur.2019.00030
Westlund KN, Kochukov MY, Lu Y, McNearney TA (2010) Impact of central and peripheral TRPV1 and ROS levels on proinflammatory mediators and nociceptive behavior. Mol Pain. https://doi.org/10.1186/1744-8069-6-46
Labadie K (2010) Chikungunya disease in nonhuman primates leads to long-term viral persistence in macrophages. J Clin Invest 120:1–13. https://doi.org/10.1172/JCI40104.894
Venugopalan A, Ghorpade RP, Chopra A (2014) Cytokines in acute chikungunya. PLoS ONE. https://doi.org/10.1371/journal.pone.0111305
Picazo-Juárez G, Romero-Suárez S, Nieto-Posadas AS et al (2011) Identification of a binding motif in the S5 helix that confers cholesterol sensitivity to the TRPV1 ion channel. J Biol Chem 286:24966–24976. https://doi.org/10.1074/jbc.M111.237537
Sudji IR, Subburaj Y, Frenkel N et al (2015) Membrane disintegration caused by the steroid saponin digitonin is related to the presence of cholesterol. Molecules 20:20146–20160. https://doi.org/10.3390/molecules201119682
Frenkel N, Makky A, Sudji IR et al (2014) Mechanistic investigation of interactions between steroidal saponin digitonin and cell membrane models. J Phys Chem B 118:14632–14639. https://doi.org/10.1021/jp5074939
Mitra S, Dungan SR (2001) Cholesterol solubilization in aqueous micellar solutions of quillaja saponin, bile salts, or nonionic surfactants. J Agric Food Chem 49:384–394. https://doi.org/10.1021/jf000568r
Szoke É, Börzsei R, Tóth DM et al (2010) Effect of lipid raft disruption on TRPV1 receptor activation of trigeminal sensory neurons and transfected cell line. Eur J Pharmacol 628:67–74. https://doi.org/10.1016/j.ejphar.2009.11.052
Liu M, Huang W, Wu D, Priestley JV (2006) TRPV1, but not P2X3, requires cholesterol for its function and membrane expression in rat nociceptors. Eur J Neurosci 24:1–6. https://doi.org/10.1111/j.1460-9568.2006.04889.x
Sághy É, Szoke É, Payrits M et al (2015) Evidence for the role of lipid rafts and sphingomyelin in Ca2+-gating of Transient Receptor Potential channels in trigeminal sensory neurons and peripheral nerve terminals. Pharmacol Res 100:101–116. https://doi.org/10.1016/j.phrs.2015.07.028
Saha S, Ghosh A, Tiwari N et al (2017) Preferential selection of Arginine at the lipid-water-interface of TRPV1 during vertebrate evolution correlates with its snorkeling behaviour and cholesterol interaction. Sci Rep. https://doi.org/10.1038/s41598-017-16780-w
Jeong JH, Lee DK, Liu S, et al (2018) Activation of temperature-sensitive TRPV1-like receptors in ARC POMC neurons reduces food intake. 1–24
Baboota RK, Singh DP, Sarma SM, et al (2014) Capsaicin Induces ‘“ Brite ”’ Phenotype in Differentiating 3T3-L1 Preadipocytes. 9:1–12. https://doi.org/https://doi.org/10.1371/journal.pone.0103093
Takahashi N, Matsuda Y, Yamada H, et al (2014) Epithelial TRPV1 Signaling Cell Proliferation. 1141–1147. https://doi.org/https://doi.org/10.1177/0022034514552826
To I (2009) Transient Receptor Potential Vanilloid-1 Signaling as a Regulator of Human Sebocyte Biology. https://doi.org/https://doi.org/10.1038/jid.2008.258
Tak PP, Firestein GS (2001) NF- κ B in defense and disease NF- κ B : a key role in inflammatory diseases. 107:7–11
Lawrence T (2009) The nuclear factor NF-kappaB pathway in inflammation. Cold Spring Harbor perspectives in biology 1:1–10. https://doi.org/10.1101/cshperspect.a001651
Wang Y, Wang DH (2013a) Role of the transient receptor potential vanilloid type 1 channel in renal inflammation induced by lipopolysaccharide in mice. Am J Physiol 304:R1–R9. https://doi.org/10.1152/ajpregu.00163.2012
Tóth A, Blumberg PM, Chen Z, Kozikowski AP (2004) Design of a high-affinity competitive antagonist of the Vanilloid receptor selective for the calcium entry-linked receptor population. Mol Pharmacol 65:282–291. https://doi.org/10.1124/mol.65.2.282
Kárai LJ, Russell JT, Iadarola MJ, Oláh Z (2004) Vanilloid receptor 1 regulates multiple calcium compartments and contributes to Ca2+-induced Ca2+ release in sensory neurons. J Biol Chem 279:16377–16387. https://doi.org/10.1074/jbc.M310891200
Grant ER, Dubin AE, Zhang SP et al (2002) Simultaneous intracellular calcium and sodium flux imaging in human vanilloid receptor 1 (VR1)-transfected human embryonic kidney cells: a method to resolve ionic dependence of VR1-mediated cell death. J Pharmacol Exp Ther 300:9–17. https://doi.org/10.1124/jpet.300.1.9
Assas BM, Wakid MH, Zakai HA et al (2016) Transient receptor potential vanilloid 1 expression and function in splenic dendritic cells: a potential role in immune homeostasis. Immunology 147:292–304. https://doi.org/10.1111/imm.12562
Kim HS, Kwon HJ, Kim GE et al (2014) Attenuation of natural killer cell functions by capsaicin through a direct and TRPV1-independent mechanism: capsaicin-induced NK cell dysfunction. Carcinogenesis 35:1652–1660. https://doi.org/10.1093/carcin/bgu091
Szitter I, Pozsgai G, Sandor K et al (2010) The role of transient receptor potential vanilloid 1 (Trpv1) receptors in dextran sulfate-induced colitis in mice. J Mol Neurosci 42:80–88. https://doi.org/10.1007/s12031-010-9366-5
Messeguer A, Planells-Cases R, Ferrer-Montiel A (2006) Physiology and pharmacology of the vanilloid receptor. Curr Neuropharmacol 4:1–15. https://doi.org/10.2174/157015906775202995
Alawi K, Keeble J (2010) The paradoxical role of the transient receptor potential vanilloid 1 receptor in inflammation. Pharmacol Ther 125:181–195. https://doi.org/10.1016/j.pharmthera.2009.10.005
Wang Y, Wang DH (2013b) TRPV1 ablation aggravates inflammatory responses and organ damage during endotoxic shock. Clin Vaccine Immunol 20:1008–1015. https://doi.org/10.1128/CVI.00674-12
Zhao JF, Ching LC, Kou YR et al (2013) Activation of TRPV1 prevents OxLDL-induced lipid accumulation and TNF- α -Induced inflammation in macrophages: role of liver X receptor α. Mediators Inflamm. https://doi.org/10.1155/2013/925171
Zhong B, Rubinstein J, Ma S, Wang DH (2018) Genetic ablation of TRPV1 exacerbates pressure overload-induced cardiac hypertrophy. Biomed Pharmacother 99:261–270. https://doi.org/10.1016/j.biopha.2018.01.065
Palomer X, Coll T, Davidson MM, et al (2010) The p65 subunit of NF- k B binds to PGC-1 a , linking inflammation and metabolic disturbances in cardiac cells. 449–458. https://doi.org/https://doi.org/10.1093/cvr/cvq080
Pollock N, Taylor G, Jobe F, Guzman E (2019) Modulation of the transcription factor NF- k B in antigen- presenting cells by bovine respiratory syncytial virus small hydrophobic protein. 1587–1599. https://doi.org/https://doi.org/10.1099/jgv.0.000855
Gutierrez H, Davies AM (2011) Regulation of neural process growth, elaboration and structural plasticity by NF- k B. Trends Neurosci 34:316–325. https://doi.org/10.1016/j.tins.2011.03.001
Liu T, Zhang L, Joo D, Sun S (2017) NF- κ B signaling in in fl ammation. https://doi.org/10.1038/sigtrans.2017.23
Doñate-Macián P, Jungfleisch J, Pérez-Vilaró G et al (2018) The TRPV4 channel links calcium influx to DDX3X activity and viral infectivity. Nat Commun 9:1–13. https://doi.org/10.1038/s41467-018-04776-7
Rinkenberger N, Schoggins JW (2018) Mucolipin-2 cation channel increases trafficking efficiency of endocytosed viruses. mBio 9:1–16. https://doi.org/https://doi.org/10.1128/mBio.02314-17
Zhou Y, Frey TK, Yang JJ (2009) Viral calciomics: interplays between Ca2+ and virus. Cell Calcium 46:1–17. https://doi.org/10.1016/j.ceca.2009.05.005
Apaire-marchais V, Ogliastro M, Chandre F, et al (2016) Minireview Virus and calcium : an unexpected tandem to optimize insecticide efficacy. 8:168–178. https://doi.org/10.1111/1758-2229.12377
Chen X, Cao R, Zhong W (2020) Host calcium channels and pumps in viral infections. Cells 9(1):94
Ueda M, Daidoji T, Anariwa D, Yang C-S, Ibrahim MS, Ikuta K, Nakaya T (2010) Highly pathogenic H5N1 avian influenza virus induces extracellular Ca2+ influx, leading to apoptosis in avian cells. J Virol 84(6):3068–3078
Taverner WK, Jacobus EJ, Christianson J, Champion B, Paton AW, Paton JC, Weiheng S, Cawood R, Seymour LW, Lei-Rossmann J (2019) Calcium influx caused by ER stress inducers enhances oncolytic adenovirus enadenotucirev replication and killing through PKCα activation. Mol Ther Oncolytics 15:117–130
Acknowledgements
We are thankful to Dr. Manmohan Parida, DRDE, Gwalior, India, for kindly providing the virus strain (DRDE-06), Vero cell line, and E2 antibody. We are also thankful to the flow cytometry facility and the confocal facility of NISER and ILS for their support.
Funding
This study was partly funded by CSIR, India grant no. 37 (1542)/12/EMR-II, 37(1675)/16/EMR-II, DST FIST grant no. SR/FST/LSI-652/2015 and DST-SERB, India grant no. EMR/2016/000854. It was supported by Institute of Life Sciences, Bhubaneswar, under the Department of Biotechnology and National Institute of Science Education and Research, HBNI, Bhubaneswar, under the Department of Atomic Energy, Government of India.
Author information
Authors and Affiliations
Contributions
Conceptualization: P Sanjai Kumar, Soma Chattopadhyay, Subhasis Chattopadhyay; Methodology: P Sanjai Kumar, Tapas K. Nayak, Chandan Mahish, Subhransu S. Sahoo, Anukrishna R, Saikat De, Ankita Datey, Ram P. Sahu, Chandan Goswami, Soma Chattopadhyay, Subhasis Chattopadhyay; Formal analysis and investigation: P Sanjai Kumar, Soma Chattopadhyay, Subhasis Chattopadhyay; Writing - original draft preparation: P Sanjai Kumar, Soma Chattopadhyay, Subhasis Chattopadhyay; Writing - review and editing: P Sanjai Kumar, Tapas K. Nayak, Chandan Mahish, Subhransu S. Sahoo, Anukrishna R, Saikat De, Ankita Datey, Ram P. Sahu, Chandan Goswami, Soma Chattopadhyay, Subhasis Chattopadhyay; Funding acquisition: Soma Chattopadhyay, Subhasis Chattopadhyay; Resources: Soma Chattopadhyay, Subhasis Chattopadhyay; Supervision: Soma Chattopadhyay, Subhasis Chattopadhyay
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest. The funding sponsors had no role in the design of the study; in the collection, analysis, or interpretation of data; in the writing of the manuscript; or in the decision to publish the results.
Additional information
Handling Editor: Patricia Aguilar.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Sanjai Kumar, P., Nayak, T.K., Mahish, C. et al. Inhibition of transient receptor potential vanilloid 1 (TRPV1) channel regulates chikungunya virus infection in macrophages. Arch Virol 166, 139–155 (2021). https://doi.org/10.1007/s00705-020-04852-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00705-020-04852-8