Abstract
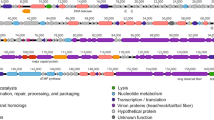
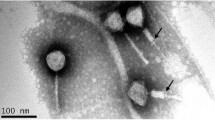
In the present study, we investigate the biological properties and genomic organization of virulent bacteriophage AM24, which specifically infects multidrug-resistant clinical Acinetobacter baumannii strains with a K9 capsular polysaccharide structure. The phage was identified as a member of the family Myoviridae by transmission electron microscopy. The AM24 linear double-stranded DNA genome of 97,177 bp contains 167 open reading frames. Putative functions were assigned for products of 40 predicted genes, including proteins involved in nucleotide metabolism and DNA replication, packaging of DNA into the capsid, phage assembly and structural proteins, and bacterial cell lysis. The gene encoding the tailspike, which possesses depolymerase activity towards the corresponding capsular polysaccharides, is situated in the phage genome outside of the structural module, upstream of the genes responsible for packaging of DNA into the capsid. The data on characterization of depolymerase-carrying phage AM24 contributes to our knowledge of the diversity of viruses infecting different capsular types of A. baumannii.


Similar content being viewed by others
References
Doi Y, Murray GL, Peleg AY (2015) Acinetobacter baumannii: evolution of antimicrobial resistance-treatment options. Semin Respir Crit Care Med 36(1):85–98. https://doi.org/10.1055/s-0034-1398388
Towner KJ (2009) Acinetobacter: an old friend, but a new enemy. J Hosp Infect 73(4):355–363. https://doi.org/10.1016/j.jhin.2009.03.032
Zarrilli R, Pournaras S, Giannouli M, Tsakris A (2013) Global evolution of multidrug-resistant Acinetobacter baumannii clonal lineages. Int J Antimicrob Agents 41(1):11–19. https://doi.org/10.1016/j.ijantimicag.2012.09.008
Kenyon JJ, Hall RM (2013) Variation in the Complex Carbohydrate Biosynthesis Loci of Acinetobacter baumannii Genomes. PLoS One 8(4):e62160. https://doi.org/10.1371/journal.pone.0062160
Popova AV, Lavysh DG, Klimuk EI et al (2017) Novel Fri1-like Viruses Infecting Acinetobacter baumannii—vB_AbaP_AS11 and vB_AbaP_AS12-characterization, comparative genomic analysis, and host-recognition strategy. Viruses. 9(7):188. https://doi.org/10.3390/v9070188
Adams MD (1959) Bacteriophages. Interscience Publishers Inc., New York (OCLC 326505)
Bartual SG, Seifert H, Hippler C et al (2005) Development of a multilocus sequence typing scheme for characterization of clinical isolates of Acinetobacter baumannii. J Clin Microbiol 43:4382–4390. https://doi.org/10.1128/JCM.43.9.4382-4390.2005
Diancourt L, Passet V, Nemec A et al (2010) The population structure of Acinetobacter baumannii: Expanding multiresistant clones from an ancestral susceptible genetic pool. PLoS One 5(4):e10034. https://doi.org/10.1371/journal.pone.0010034
Oliveira H, Costa AR, Konstantinides N et al (2017) Ability of phages to infect Acinetobacter calcoaceticus-Acinetobacter baumannii complex species through acquisition of different pectate lyase depolymerase domains. Environ Microbiol 19(12):5060–5077. https://doi.org/10.1111/1462-2920.13970
Sambrook J, Fritsch EF, Maniatis T (1989) Molecular cloning: a laboratory manual, 2nd edn. Laboratory Press, Cold Spring Harbor (ISBN: 0-87969-309-6)
Aziz RK, Bartels D, Best AA et al (2008) The RAST Server: rapid annotations using subsystems technology. BMC Genom 9:75. https://doi.org/10.1186/1471-2164-9-75
Laslett D, Canback B (2004) ARAGORN, a program for the detection of transfer RNA and transfer-messenger RNA genes in nucleotide sequences. Nucleic Acids Res 32(1):11–16. https://doi.org/10.1093/nar/gkh152
Söding J, Biegert A, Lupas AN (2005) The HHpred interactive server for protein homology detection and structure prediction. Nucleic Acids Res 33:W244–W248. https://doi.org/10.1093/nar/gki408
Novikova O, Topilina N, Belfort M (2014) Enigmatic distribution, evolution, and function of inteins. J Biol Chem 289(21):14490–14497. https://doi.org/10.1074/jbc.R114.548255
Turner D, Ackermann HW, Kropinski AM et al (2017) Comparative analysis of 37 Acinetobacter bacteriophages. Viruses 10(1):5. https://doi.org/10.3390/v10010005
Sievers F, Higgins DG (2014) Clustal Omega, accurate alignment of very large numbers of sequences. Methods Mol Biol. 1079:105–16. https://doi.org/10.1007/978-1-62703-646-7_6
Acknowledgements
The authors would like to thank Dr. Evgeny Zhilenkov (MicroWorld Ltd., Moscow, Russia) for assistance with TEM, Dr. Daria Lavysh (Heidelberg University, Heidelberg, Germany) for the participating in bioinformatic analysis, Dr. Yuriy Knirel’s group (N.D. Zelinsky Institute of Organic Chemistry, Russian Academy of Sciences, Moscow, Russia) for determination of the A. baumannii B05 CPS structure, and Dr. Olga Ershova (N. N. Burdenko Research Institute for Neurosurgery, Moscow, Russia) for the providing of clinical materials and A. baumannii strains.
Funding
The isolation of phage AM24 and determination of its infection parameters were supported by the Sectoral Scientific Program of the Russian Federal Service for Surveillance on Consumer Rights Protection and Human Wellbeing. Genomic analysis and phage host range determination were supported by the Russian Science Foundation (grant 18-15-00403).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
This article does not contain studies with human participants or animals performed by any of the authors.
Additional information
Handling Editor: Johannes Wittmann.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Popova, A.V., Shneider, M.M., Myakinina, V.P. et al. Characterization of myophage AM24 infecting Acinetobacter baumannii of the K9 capsular type. Arch Virol 164, 1493–1497 (2019). https://doi.org/10.1007/s00705-019-04208-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00705-019-04208-x