Abstract
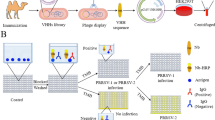
Porcine reproductive and respiratory syndrome virus (PRRSV) causes great economic losses to the swine industry worldwide. Typically, an N protein-coated indirect enzyme-linked immunosorbent assay (N-coated iELISA) is used to detect PRRSV antibodies. Non-structural protein (NSP) 4 is essential to the PRRSV life cycle and contains B-cell epitopes. Yet, no specific antibody against NSP4 has been detected in clinical samples. In this study, we developed an NSP4-coated iELISA and compared its effectiveness with the N-coated iELISA. The NSP4-coated iELISA was developed with a cut-off value of 0.406 at an optical density of 450 nm by testing a panel of 70 PRRSV positive and 80 PRRSV negative pig serum samples, which generated a specificity and sensitivity of 100%. Agreement between the NSP4-coated and N-coated iELISAs was 92.2%. Interestingly, 50 serum samples, mostly from pigs vaccinated with the HP-PRRSV live strain, tested positive for PRRSV antibodies with the NSP4-coated iELISA, but were negative with the N-coated iELISA. These results were further confirmed by western blot analysis and another iELISA based on the N-terminus of NSP2 (NSP2-1-coated iELISA). The agreement between the results of western blot analysis with the NSP4-coated and NSP2-1-coated iELISA analyses were 92% and 96.1%, respectively, showing that the developed NSP4-coated iELISA is a useful tool to discriminate a false negative from a true negative response to the HP-PRRSV vaccine.


Similar content being viewed by others
References
Baron T, Albina E, Leforban Y, Madec F, Guilmoto H, Plana Duran J, Vannier P (1992) Report on the first outbreaks of the porcine reproductive and respiratory syndrome (PRRS) in France. Diagnosis and viral isolation. Annales de recherches veterinaires Ann Vet Res 23:161–166
Barrette-Ng IH, Ng KK, Mark BL, Van Aken D, Cherney MM, Garen C, Kolodenko Y, Gorbalenya AE, Snijder EJ, James MN (2002) Structure of arterivirus nsp4. The smallest chymotrypsin-like proteinase with an alpha/beta C-terminal extension and alternate conformations of the oxyanion hole. J Biol Chem 277:39960–39966
Brockmeier SL, Loving CL, Vorwald AC, Kehrli ME Jr, Baker RB, Nicholson TL, Lager KM, Miller LC, Faaberg KS (2012) Genomic sequence and virulence comparison of four type 2 porcine reproductive and respiratory syndrome virus strains. Virus Res 169:212–221
Brown E, Lawson S, Welbon C, Gnanandarajah J, Li J, Murtaugh MP, Nelson EA, Molina RM, Zimmerman JJ, Rowland RR, Fang Y (2009) Antibody response to porcine reproductive and respiratory syndrome virus (PRRSV) nonstructural proteins and implications for diagnostic detection and differentiation of PRRSV types I and II. Clin Vaccine Immunol 16:628–635
Cavanagh D (1997) Nidovirales: a new order comprising Coronaviridae and Arteriviridae. Arch Virol 142:629–633
Chen Z, Li M, He Q, Du J, Zhou L, Ge X, Guo X, Yang H (2014) The amino acid at residue 155 in nonstructural protein 4 of porcine reproductive and respiratory syndrome virus contributes to its inhibitory effect for interferon-beta transcription in vitro. Virus Res 189:226–234
de Lima M, Pattnaik AK, Flores EF, Osorio FA (2006) Serologic marker candidates identified among B-cell linear epitopes of Nsp2 and structural proteins of a North American strain of porcine reproductive and respiratory syndrome virus. Virology 353:410–421
Greiner M (1995) Two-graph receiver operating characteristic (TG-ROC): a Microsoft-EXCEL template for the selection of cut-off values in diagnostic tests. J Immunol Methods 185:145–146
Huang C, Du Y, Yu Z, Zhang Q, Liu Y, Tang J, Shi J, Feng WH (2016) Highly pathogenic porcine reproductive and respiratory syndrome virus Nsp4 cleaves VISA to impair antiviral responses mediated by RIG-I-like receptors. Sci Rep 6:28497
Hopper SA, White ME, Twiddy N (1992) An outbreak of blue-eared pig disease (porcine reproductive and respiratory syndrome) in four pig herds in Great Britain. Vet Record 131:140–144
Johnson CR, Yu W, Murtaugh MP (2007) Cross-reactive antibody responses to nsp1 and nsp2 of Porcine reproductive and respiratory syndrome virus. J General Virol 88:1184–1195
Li Y, Wang X, Bo K, Tang B, Yang B, Jiang W, Jiang P (2007) Emergence of a highly pathogenic porcine reproductive and respiratory syndrome virus in the Mid-Eastern region of China. Vet J 174:577–584
Lunney JK, Fang Y, Ladinig A, Chen N, Li Y, Rowland B, Renukaradhya GJ (2016) Porcine reproductive and respiratory syndrome virus (PRRSV): pathogenesis and interaction with the immune system. Ann Rev Animal Biosci 4:129–154
Meulenberg JJ, Hulst MM, de Meijer EJ, Moonen PL, den Besten A, de Kluyver EP, Wensvoort G, Moormann RJ (1993) Lelystad virus, the causative agent of porcine epidemic abortion and respiratory syndrome (PEARS), is related to LDV and EAV. Virology 192:62
Oleksiewicz MB, Botner A, Toft P, Normann P, Storgaard T (2001) Epitope mapping porcine reproductive and respiratory syndrome virus by phage display: the nsp2 fragment of the replicase polyprotein contains a cluster of B-cell epitopes. J Virol 75:3277–3290
Rascon-Castelo E, Burgara-Estrella A, Mateu E, Hernandez J (2015) Immunological features of the non-structural proteins of porcine reproductive and respiratory syndrome virus. Viruses 7:873–886
Snijder EJ, Wassenaar AL, van Dinten LC, Spaan WJ, Gorbalenya AE (1996) The arterivirus nsp4 protease is the prototype of a novel group of chymotrypsin-like enzymes, the 3C-like serine proteases. J Biol Chem 271:4864–4871
Tian K, Yu X, Zhao T, Feng Y, Cao Z, Wang C, Hu Y, Chen X, Hu D, Tian X, Liu D, Zhang S, Deng X, Ding Y, Yang L, Zhang Y, Xiao H, Qiao M, Wang B, Hou L, Wang X, Yang X, Kang L, Sun M, Jin P, Wang S, Kitamura Y, Yan J, Gao GF (2007) Emergence of fatal PRRSV variants: unparalleled outbreaks of atypical PRRS in China and molecular dissection of the unique hallmark. PLoS One 2:e526
Tong GZ, Zhou YJ, Hao XF, Tian ZJ, An TQ, Qiu HJ (2007) Highly pathogenic porcine reproductive and respiratory syndrome, China. Emerg Infect Dis 13:1434–1436
Wang X, Qiu H, Zhang M, Cai X, Qu Y, Hu D, Zhao X, Zhou E, Liu S, Xiao Y (2015) Distribution of highly pathogenic porcine reproductive and respiratory syndrome virus (HP-PRRSV) in different stages of gestation sows: HP-PRRSV distribution in gestation sows. Vet Immunol Immunopathol 166:88–94
Xiao YH, Wang TT, Zhao Q, Wang CB, Lv JH, Nie L, Gao JM, Ma XC, Hsu WH, Zhou EM (2014) Development of indirect ELISAs for differential serodiagnosis of classical and highly pathogenic porcine reproductive and respiratory syndrome virus. Transbound emerg Dis 61:341–349
Zhang M, Li X, Cai X, Qu Y, Hu D, Lv L, Liu S, Zhao X, Wang L, Xiao Y (2016) Evaluation of infection status in Chinese swine with porcine reproductive and respiratory syndrome virus by nested RT-PCR targeting nsp2 gene. Infect Genet Evol 44:55–60
Zhou L, Wang Z, Ding Y, Ge X, Guo X, Yang H (2015) NADC30-like strain of porcine reproductive and respiratory syndrome virus, China. Emerg Infect Dis 21:2256–2257
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Funding
This study was funded by the National Natural Science Foundation of China (Grant no. 31772708), the Natural Science Foundation of Shandong Province (Grant no. ZR2014CM024) and the Funds of Shandong “Double Tops” Program.
Conflict of interest
The authors have no conflicts of interest to declare.
Ethical approval
All samples in this study were handled in accordance with the guidelines of the Animal Care and Use Committee of Shandong Agricultural University. This study did not involve human participants.
Additional information
Handling Editor: Zhenhai Chen.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Cao, S., Cai, X., Tan, M. et al. Serodiagnosis, targeting nonstructural protein 4, of porcine reproductive and respiratory syndrome virus. Arch Virol 163, 411–418 (2018). https://doi.org/10.1007/s00705-017-3625-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00705-017-3625-5