Abstract
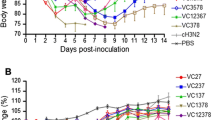
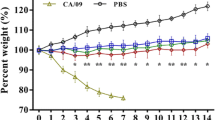
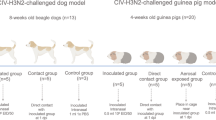
An outbreak of a canine influenza virus (CIV) H3N2 reassortant derived from pandemic (pdm) H1N1 and CIV H3N2 in companion animals has underscored the urgent need to monitor CIV infections for potential zoonotic transmission of influenza viruses to humans. In this study, we assessed the virulence of a novel CIV H3N2 reassortant, VC378, which was obtained from a dog that was coinfected with pdm H1N1 and CIV H3N2, in ferrets, dogs, and mice. Significantly enhanced virulence of VC378 was demonstrated in mice, although the transmissibility and pathogenicity of VC378 were similar to those of classical H3N2 in ferrets and dogs. This is notable because mice inoculated with an equivalent dose of classical CIV H3N2 showed no clinical signs and no lethality. We found that the PA and NS gene segments of VC378 were introduced from pdmH1N1, and these genes included the amino acid substitutions PA-P224S and NS-I123V, which were previously found to be associated with increased virulence in mice. Thus, we speculate that the natural reassortment between pdm H1N1 and CIV H3N2 can confer virulence and that continuous surveillance is needed to monitor the evolution of CIV in companion animals.





Similar content being viewed by others
References
Beeler E (2009) Influenza in dogs and cats. Vet Clin N Am Small Anim Pract 39:251–264
Brown EG (1990) Increased virulence of a mouse-adapted variant of influenza A/FM/1/47 virus is controlled by mutations in genome segments 4, 5, 7, and 8. J Virol 64:4523–4533
Brown EG, Bailly JE (1999) Genetic analysis of mouse-adapted influenza A virus identifies roles for the NA, PB1, and PB2 genes in virulence. Virus Res 61:63–76
Brown EG, Liu H, Kit LC, Baird S, Nesrallah M (2001) Pattern of mutation in the genome of influenza A virus on adaptation to increased virulence in the mouse lung: identification of functional themes. Proc Natl Acad Sci USA 98:6883–6888
Bunpapong N, Nonthabenjawan N, Chaiwong S, Tangwangvivat R, Boonyapisitsopa S, Jairak W, Tuanudom R, Prakairungnamthip D, Suradhat S, Thanawongnuwech R, Amonsin A (2014) Genetic characterization of canine influenza A virus (H3N2) in Thailand. Virus Genes 48:56–63
Chen LM, Davis CT, Zhou H, Cox NJ, Donis RO (2008) Genetic compatibility and virulence of reassortants derived from contemporary avian H5N1 and human H3N2 influenza A viruses. PLoS Pathog 4:e1000072
Christman MC, Kedwaii A, Xu J, Donis RO, Lu G (2011) Pandemic (H1N1) 2009 virus revisited: an evolutionary retrospective. Infect Genet Evol 11:803–811
Itoh Y, Shinya K, Kiso M, Watanabe T, Sakoda Y, Hatta M, Muramoto Y, Tamura D, Sakai-Tagawa Y, Noda T, Sakabe S, Imai M, Hatta Y, Watanabe S, Li C, Yamada S, Fujii K, Murakami S, Imai H, Kakugawa S, Ito M, Takano R, Iwatsuki-Horimoto K, Shimojima M, Horimoto T, Goto H, Takahashi K, Makino A, Ishigaki H, Nakayama M, Okamatsu M, Takahashi K, Warshauer D, Shult PA, Saito R, Suzuki H, Furuta Y, Yamashita M, Mitamura K, Nakano K, Nakamura M, Brockman-Schneider R, Mitamura H, Yamazaki M, Sugaya N, Suresh M, Ozawa M, Neumann G, Gern J, Kida H, Ogasawara K, Kawaoka Y (2009) In vitro and in vivo characterization of new swine-origin H1N1 influenza viruses. Nature 460:1021–1025
Jeoung HY, Lim SI, Shin BH, Lim JA, Song JY, Song DS, Kang BK, Moon HJ, An DJ (2013) A novel canine influenza H3N2 virus isolated from cats in an animal shelter. Vet Microbiol 165:281–286
Keenliside J (2013) Pandemic influenza A H1N1 in Swine and other animals. Curr Top Microbiol Immunol 370:259–271
Kim H, Song D, Moon H, Yeom M, Park S, Hong M, Na W, Webby RJ, Webster RG, Park B, Kim JK, Kang B (2013) Inter- and intraspecies transmission of canine influenza virus (H3N2) in dogs, cats, and ferrets. Influenza Other Respir Viruses 7:265–270
Lipatov AS, Andreansky S, Webby RJ, Hulse DJ, Rehg JE, Krauss S, Perez DR, Doherty PC, Webster RG, Sangster MY (2005) Pathogenesis of Hong Kong H5N1 influenza virus NS gene reassortants in mice: the role of cytokines and B- and T-cell responses. J Gen Virol 86:1121–1130
Lyoo KS, Kim JK, Kang B, Moon H, Kim J, Song M, Park B, Kim SH, Webster RG, Song D (2015) Comparative analysis of virulence of a novel, avian-origin H3N2 canine influenza virus in various host species. Virus Res 195:135–140
Ma W, Brenner D, Wang Z, Dauber B, Ehrhardt C, Hogner K, Herold S, Ludwig S, Wolff T, Yu K, Richt JA, Planz O, Pleschka S (2010) The NS segment of an H5N1 highly pathogenic avian influenza virus (HPAIV) is sufficient to alter replication efficiency, cell tropism, and host range of an H7N1 HPAIV. J Virol 84:2122–2133
Na W, Lyoo KS, Song EJ, Hong M, Yeom M, Moon H, Kang BK, Kim DJ, Kim JK, Song D (2015) Viral dominance of reassortants between canine influenza H3N2 and pandemic (2009) H1N1 viruses from a naturally co-infected dog. Virol J 12:134
Nakagawa Y, Oda K, Nakada S (1996) The PB1 subunit alone can catalyze cRNA synthesis, and the PA subunit in addition to the PB1 subunit is required for viral RNA synthesis in replication of the influenza virus genome. J Virol 70:6390–6394
Nelson MI, Vincent AL, Kitikoon P, Holmes EC, Gramer MR (2012) Evolution of novel reassortant A/H3N2 influenza viruses in North American swine and humans, 2009–2011. J Virol 86:8872–8878
Noronha JM, Liu M, Squires RB, Pickett BE, Hale BG, Air GM, Galloway SE, Takimoto T, Schmolke M, Hunt V, Klem E, Garcia-Sastre A, McGee M, Scheuermann RH (2012) Influenza virus sequence feature variant type analysis: evidence of a role for NS1 in influenza virus host range restriction. J Virol 86:5857–5866
Octaviani CP, Ozawa M, Yamada S, Goto H, Kawaoka Y (2010) High level of genetic compatibility between swine-origin H1N1 and highly pathogenic avian H5N1 influenza viruses. J Virol 84:10918–10922
Parrish CR, Murcia PR, Holmes EC (2015) Influenza virus reservoirs and intermediate hosts: dogs, horses, and new possibilities for influenza virus exposure of humans. J Virol 89:2990–2994
Ping J, Keleta L, Forbes NE, Dankar S, Stecho W, Tyler S, Zhou Y, Babiuk L, Weingartl H, Halpin RA, Boyne A, Bera J, Hostetler J, Fedorova NB, Proudfoot K, Katzel DA, Stockwell TB, Ghedin E, Spiro DJ, Brown EG (2011) Genomic and protein structural maps of adaptive evolution of human influenza A virus to increased virulence in the mouse. PLoS One 6:e21740
Regan JF, Liang Y, Parslow TG (2006) Defective assembly of influenza A virus due to a mutation in the polymerase subunit PA. J Virol 80:252–261
Romero-Tejeda A, Capua I (2013) Virus-specific factors associated with zoonotic and pandemic potential. Influenza and other respiratory viruses 7(Suppl 2):4–14
Smith GJ, Vijaykrishna D, Bahl J, Lycett SJ, Worobey M, Pybus OG, Ma SK, Cheung CL, Raghwani J, Bhatt S, Peiris JS, Guan Y, Rambaut A (2009) Origins and evolutionary genomics of the 2009 swine-origin H1N1 influenza A epidemic. Nature 459:1122–1125
Song D, Kang B, Lee C, Jung K, Ha G, Kang D, Park S, Park B, Oh J (2008) Transmission of avian influenza virus (H3N2) to dogs. Emerg Infect Dis 14:741–746
Song D, Moon HJ, An DJ, Jeoung HY, Kim H, Yeom MJ, Hong M, Nam JH, Park SJ, Park BK, Oh JS, Song M, Webster RG, Kim JK, Kang BK (2012) A novel reassortant canine H3N1 influenza virus between pandemic H1N1 and canine H3N2 influenza viruses in Korea. J Gen Virol 93:551–554
Song D, Kim H, Na W, Hong M, Park SJ, Moon H, Kang B, Lyoo KS, Yeom M, Jeong DG, An DJ, Kim JK (2015) Canine susceptibility to human influenza viruses (A/pdm 09H1N1, A/H3N2 and B). J Gen Virol 96:254–258
Song DS, An DJ, Moon HJ, Yeom MJ, Jeong HY, Jeong WS, Park SJ, Kim HK, Han SY, Oh JS, Park BK, Kim JK, Poo H, Webster RG, Jung K, Kang BK (2011) Interspecies transmission of the canine influenza H3N2 virus to domestic cats in South Korea, 2010. J Gen Virol 92:2350–2355
Sun Y, Xu Q, Shen Y, Liu L, Wei K, Sun H, Pu J, Chang KC, Liu J (2014) Naturally occurring mutations in the PA gene are key contributors to increased virulence of pandemic H1N1/09 influenza virus in mice. J Virol 88:4600–4604
Urbaniak K, Markowska-Daniel I (2014) In vivo reassortment of influenza viruses. Acta Biochim Pol 61:427–431
Vergara-Alert J, Busquets N, Ballester M, Chaves AJ, Rivas R, Dolz R, Wang Z, Pleschka S, Majo N, Rodriguez F, Darji A (2014) The NS segment of H5N1 avian influenza viruses (AIV) enhances the virulence of an H7N1 AIV in chickens. Vet Res 45:7
Wang H, Jia K, Qi W, Zhang M, Sun L, Liang H, Du G, Tan L, Shao Z, Ye J, Sun L, Cao Z, Chen Y, Zhou P, Su S, Li S (2013) Genetic characterization of avian-origin H3N2 canine influenza viruses isolated from Guangdong during 2006–2012. Virus Genes 46:558–562
Zhang K, Zhang Z, Yu Z, Li L, Cheng K, Wang T, Huang G, Yang S, Zhao Y, Feng N, Fu J, Qin C, Gao Y, Xia X (2013) Domestic cats and dogs are susceptible to H9N2 avian influenza virus. Virus Res 175:52–57
Zhang YB, Chen JD, Xie JX, Zhu WJ, Wei CY, Tan LK, Cao N, Chen Y, Zhang MZ, Zhang GH, Li SJ (2013) Serologic reports of H3N2 canine influenza virus infection in dogs in northeast China. J Vet Med Sci 75:1061–1062
Zhu H, Hughes J, Murcia PR (2015) Origins and Evolutionary Dynamics of H3N2 Canine Influenza Virus. J Virol 89:5406–5418
Acknowledgments
This work was supported by the Korea Healthcare Technology R&D Project, Ministry of Health and Welfare, Republic of Korea (Grant No. A103001) and BioNano Health-Guard Research Center funded by the Ministry of Science, ICT & Future Planning (MSIP) of Korea as a Global Frontier Project (Grant No. H-GUARD_2013M3A6B2078954).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare no conflict of interest.
Additional information
K. S. Lyoo and W. Na contributed equally and are co-first authors.
Rights and permissions
About this article
Cite this article
Lyoo, KS., Na, W., Yeom, M. et al. Virulence of a novel reassortant canine H3N2 influenza virus in ferret, dog and mouse models. Arch Virol 161, 1915–1923 (2016). https://doi.org/10.1007/s00705-016-2868-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00705-016-2868-x