Abstract
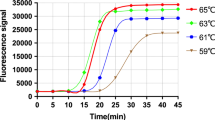
Human rhinoviruses (HRVs) have long been recognized as the cause of more than one-half of acute viral upper respiratory illnesses, and they are associated with more-serious diseases in children, such as asthma, acute otitis media and pneumonia. A rapid and universal test for of HRV infection is in high demand. In this study, a reverse transcription genome exponential amplification reaction (RT-GEAR) assay targeting the HRV 5′ untranslated region (UTR) was developed for pan-HRV detection. The reaction was performed in a single tube in one step at 65 °C for 60 min using a real-time fluorometer (Genie®II; Optigene). The RT-GEAR assay showed no cross-reactivity with common human enteroviruses, including HEV71, CVA16, CVA6, CVA10, CVA24, CVB5, Echo30, and PV1-3 or with other common respiratory viruses including FluA H3, FluB, PIV1-4, ADV3, RSVA, RSVB and HMPV. With in vitro-transcribed RNA containing the amplified regions of HRV-A60, HRV-B06 and HRV-C07 as templates, the sensitivity of the RT-GEAR assay was 5, 50 and 5 copies/reaction, respectively. Experiments to evaluate the clinical performance of the RT-GEAR assay were also carried out with a panel of 143 previously verified samples, and the results were compared with those obtained using a published semi-nested PCR assay followed by sequencing. The tested panel comprised 91 HRV-negative samples and 52 HRV-positive samples (18 HRV-A-positive samples, 3 HRV-B-positive samples and 31 HRV-C-positive samples). The sensitivity and specificity of the pan-HRVs RT-GEAR assay was 98.08 % and 100 %, respectively. The kappa correlation between the two methods was 0.985. The RT-GEAR assay based on a portable Genie®II fluorometer is a sensitive, specific and rapid assay for the universal detection of HRV infection.



Similar content being viewed by others
Abbreviations
- HRVs:
-
Human rhinoviruses
- RT-GEAR:
-
Reverse transcription genome exponential amplification reaction
- RT-LAMP:
-
Reverse transcription loop-mediated isothermal amplification
- UTR:
-
Untranslated region
- HEV:
-
Human enterovirus
- CVA:
-
Coxsackievirus A
- CVB:
-
Coxsackievirus B
- Echo:
-
Echovirus
- PV:
-
Poliovirus
- FluA H3:
-
Influenza A virus H3
- FluB:
-
Influenza B virus
- PIV:
-
Parainfluenza virus
- ADV:
-
Adenovirus
- RSV:
-
Respiratory syncytial virus
- HMPV:
-
Human metapneumovirus
- COPD:
-
Chronic obstructive pulmonary disease
References
Lemanske RF Jr, Jackson DJ, Gangnon RE, Evans MD, Li Z, Shult PA et al (2005) Rhinovirus illnesses during infancy predict subsequent childhood wheezing. J Allergy Clin Immunol 116:571–577
Mackay IM (2008) Human rhinoviruses: the cold wars resume. J Clin Virol 42:297–320
Corne JM, Marshall C, Smith S, Schreiber J, Sanderson G, Holgate ST et al (2002) Frequency, severity, and duration of rhinovirus infections in asthmatic and non-asthmatic individuals: a longitudinal cohort study. Lancet 359:831–834
Rawlinson WD, Waliuzzaman Z, Carter IW, Belessis YC, Gilbert KM, Morton JR (2003) Asthma exacerbations in children associated with rhinovirus but not human metapneumovirus infection. J Infect Dis 187:1314–1318
McIntyre CL, Knowles NJ, Simmonds P (2013) Proposals for the classification of human rhinovirus species A, B and C into genotypically assigned types. J Gen Virol 94:1791–1806
Oberste MS, Nix WA, Maher K, Pallansch MA (2003) Improved molecular identification of enteroviruses by RT-PCR and amplicon sequencing. J Clin Virol 26:375–377
Jacobs SE, Soave R, Shore TB, Satlin MJ, Schuetz AN, Magro C et al (2013) Human rhinovirus infections of the lower respiratory tract in hematopoietic stem cell transplant recipients. Transpl Infect Dis 15:474–486
Bochkov YA, Palmenberg AC, Lee WM, Rathe JA, Amineva SP, Sun X et al (2011) Molecular modeling, organ culture and reverse genetics for a newly identified human rhinovirus C. Nat Med 17:627–632
Olive DM, Al-Mufti S, Al-Mulla W, Khan MA, Pasca A, Stanway G et al (1990) Detection and differentiation of picornaviruses in clinical samples following genomic amplification. J Gen Virol 71:2141–2147
Hoek RA, Paats MS, Pas SD, Bakker M, Hoogsteden HC, Boucher CA et al (2013) Incidence of viral respiratory pathogens causing exacerbations in adult cystic fibrosis patients. Scand J Infect Dis 45:65–69
Bellau-Pujol S, Vabret A, Legrand L, Dina J, Gouarin S, Petitjean-Lecherbonnier J et al (2005) Development of three multiplex RT-PCR assays for the detection of 12 respiratory RNA viruses. J Virol Methods 126:53–63
Fry AM, Lu X, Olsen SJ, Chittaganpitch M, Sawatwong P, Chantra S et al (2011) Human rhinovirus infections in rural Thailand: epidemiological evidence for rhinovirus as both pathogen and bystander. PLoS One. doi:10.1371/journal.pone.0017780
Palmenberg AC, Spiro D, Kuzmickas R, Wang S, Djikeng A, Rathe JA et al (2009) Sequencing and analyses of all known human rhinovirus genomes reveal structure and evolution. Science 324:55–59
Weinberg GA, Schnabel KC, Erdman DD, Prill MM, Iwane MK, Shelley LM et al (2013) Field evaluation of TaqMan Array Card (TAC) for the simultaneous detection of multiple respiratory viruses in children with acute respiratory infection. J Clin Virol 57:254–260
Prithiviraj J, Hill V, Jothikumar N (2012) Rapid detection of microbial DNA by a novel isothermal genome exponential amplification reaction (GEAR) assay. Biochem Biophys Res Commun 420:738–742
Jothikumar P, Narayanan J, Hill VR (2014) Visual endpoint detection of Escherichia coli O157: H7 using isothermal Genome Exponential Amplification Reaction (GEAR) assay and malachite green. J Microbiol Methods 98:122–127
Song MH, Zhao LQ, Qian Y, Zhu RN, Deng J, Wang F et al (2013) Human rhinovirus with different genotypes in children with acute respiratory tract infections in Beijing. Bing Du Xue Bao 29:97–105
Loens K, Ieven M, Ursi D, De Laat C, Sillekens P, Oudshoorn P, Goossens H (2003) Improved detection of rhinoviruses by nucleic acid sequence-based amplification after nucleotide sequence determination of the 5’ noncoding regions of additional rhinovirus strains. J Clin Microbiol 41(5):1971–1976
Brebion A, Mirand A, Regagnon C, Archimbaud C, Chambon M, Peigue-Lafeuille H et al (2015) Evaluation of real-time RT-PCR Rhino&EV/Cc r-gene(®) (bioMerieux) kit versions 1 and 2 for rhinovirus detection. J Clin Virol 62:110–113
Faux CE, Arden KE, Lambert SB, Nissen MD, Nolan TM, Mackay IM et al (2011) Usefulness of published PCR primers in detecting human rhinovirus infection. Emerg Infect Dis 17(2):296–298
McLeish NJ, Witteveldt J, Clasper L, McIntyre C, McWilliam Leitch EC, Hardie A et al (2012) Development and assay of RNA transcripts of enterovirus species A to D, rhinovirus species a to C, and human parechovirus: assessment of assay sensitivity and specificity of real-time screening and typing methods. J Clin Microbiol 50:2910–2917
Nam YR, Lee U, Choi HS, Lee KJ, Kim N, Jang YJ et al (2015) Degenerate PCR primer design for the specific identification of rhinovirus C. J Virol Methods 214:15–24
Steven Oberste M, Penaranda Silvia, Rogers Shannon L, Elizabeth Henderson W, Nix Allan (2010) Comparative evaluation of Taqman real-time PCR and semi-nested VP1 PCR for detection of enteroviruses in clinical specimens. J Clin Virol 49:73–74
Sakthivel Senthilkumar K, Whitaker Brett, Xiaoyan Lu, Oliveira Danielle BL, Stockman Lauren J, Erdman Dean D et al (2012) Comparison of fast-track diagnostics respiratory pathogens multiplex real-time RTPCR assay with in-house singleplex assays for comprehensive detection of human respiratory viruses. J Virol Methods 185:259–266
Lu X, Chittaganpitch M, Olsen SJ, Mackay IM, Sloots TP, Fry AM, Erdman DD (2006) Real-time PCR assays for detection of bocavirus in human specimens. J Clin Microbiol 44:3231–3235
Lu X, Holloway B, Dare RK, Kuypers J, Yagi S, Erdman DD et al (2008) Real-time reverse transcription-PCR assay for comprehensive detection of human rhinoviruses. J Clin Microbiol 46:533–539
Bayko AA, Evtushenko OA, Avaeva SM et al (1988) A malachite green procedure for orthophosphate determination and its use in alkaline phosphatase-based enzyme immunoassay. Anal Biochem 171(2):266–270
Ma XJ, Shu YL, Nie K, Qin M, Wang DY, Dong XP et al (2010) Visual detection of pandemic influenza A H1N1 virus 2009 by reverse transcription loop mediated isothermal amplification with hydroxynaphthol blue dye. J Virol Methods 167(2):214–217
Nie K, Zhang Y, Luo L, Yang MJ, Xu WB, Ma XJ et al (2011) Visual detection of human enterovirus 71 subgenotype C4 and coxsackievirus A16 by reverse transcription loop-mediated isothermal amplification with hydroxynaphthol blue dye. J Virol Methods 175:283–286
Dupouey J, Ninove L, Ferrier V, Py O, Gazin C, Thirion-Perrier L, de Lamballerie X (2014) Molecular detection of human rhinoviruses in respiratory samples: a comparison of Taqman probe-, SYBR green I- and BOXTO-based real-time PCR assays. Virol J 18(11):31
Schibler M, Yerly S, Vieille G, Docquier M, Turin L, Kaiser L et al (2012) Critical analysis of rhinovirus RNA load quantification by real-time reverse transcription-PCR. J Clin Microbiol 50:2868–2872
Lu QB, Wo Y, Wang LY, Wang HY, Huang DD, Zhang XA et al (2014) Molecular epidemiology of human rhinovirus in children with acute respiratory diseases in Chongqing, China. Sci Rep 20:6686
Xiang Z, Gonzalez R, Wang Z, Xiao Y, Chen L, Li T et al (2010) Human rhinoviruses in Chinese adults with acute respiratory tract infection. J Infect 61:289–298
Martin EK, Kuypers J, Chu HY, Lacombe K, Qin X, Strelitz B et al (2015) Molecular epidemiology of human rhinovirus infections in the pediatric emergency department. J Clin Virol 62:25–31
Pretorius MA, Tempia S, Treurnicht FK, Walaza S, Cohen AL, Moyes J et al (2014) Genetic diversity and molecular epidemiology of human rhinoviruses in South Africa. Influenza Other Respir Viruses 8:567–573
Tsatsral S, Xiang Z, Fuji N, Maitsetseg C, Khulan J, Oshitani H et al (2015) Molecular epidemiology of human rhinovirus infection in Mongolian during 2008–2013. Jpn J Infect Dis 68:280–287
Li J, Mao NY, Zhang C, Yang MJ, Wang M, Xu WB et al (2012) The development of a GeXP-based multiplex reverse transcription-PCR assay for simultaneous detection of sixteen human respiratory virus types/subtypes. BMC Infect Dis 12:189–196
Sambrook Joseph, Russell David W (2001) Molecular cloning: a laboratory manual. Gold Spring Harbor, New York
Nancy Fenn Buderer (1996) Statistical methodology: I. Incorporating the prevalence of disease into the sample size calculation for sensitivity and specificity. Acad Emerg Med 3:895–900
Acknowledgements
We acknowledge the Laboratory of Virology, Capital Institute of Pediatrics, for providing nasopharyngeal aspirates, the National Laboratory for Poliomyelitis for providing the control viruses and clinical specimens, and the Chinese National Influenza Center for providing respiratory swabs used in this study. This work was supported by the China Mega-Project for Infectious Disease (2016ZX10004001-002, 2016ZX10004-001), Beijing Municipal Science & Technology Commission project (D151100002115003) and Guangzhou Municipal Science & Technology Commission project (2015B2150820).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Funding
This study was funded by the China Mega-Project for Infectious Disease (2016ZX10004001-002, 2016ZX10004-001), Beijing Municipal Science & Technology Commission project (D151100002115003) and Guangzhou Municipal Science & Technology Commission project (2015B2150820).
Conflict of interest
All authors declare that they have no competing interests.
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Informed consent
All aspects of the study were performed in accordance with national ethics regulations and appraised by the Institutional Review Boards of the Center for Disease Control and Prevention of China. Children’s parents were apprised of the study’s purpose and of their right to keep information confidential. Written informed consent was obtained from parents or caregivers.
Additional information
L. Guan, L.-Q. Zhao and H.-Y. Zhou contributed equally to this work.
Rights and permissions
About this article
Cite this article
Guan, L., Zhao, LQ., Zhou, HY. et al. Reverse transcription genome exponential amplification reaction assay for rapid and universal detection of human rhinoviruses. Arch Virol 161, 1891–1898 (2016). https://doi.org/10.1007/s00705-016-2858-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00705-016-2858-z