Abstract
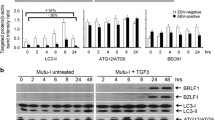
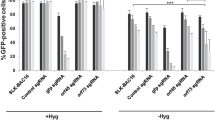
Kaposi’s sarcoma-associated herpesvirus (KSHV) is the infectious cause of the angioproliferative neoplasm Kaposi’s sarcoma (KS). We first confirmed the susceptibility of NIH 3T3 fibroblasts to KSHV by infecting them with BCP-1-derived KSHV. Lytic replication of KSHV was confirmed by PCR amplification of viral DNA isolated from culture supernatants of KSHV-infected cells. The template from KSHV-infected NIH 3T3 cells resulted in an intense viral DNA PCR product. A time course experiment revealed the disappearance of KSHV-specific DNA in culture supernatant of NIH 3T3 cells during a period between 48 h and 72 h postinfection. Furthermore, 3 days postinfection, infected NIH 3T3 cells showed no evidence of latent or lytic transcripts, including LANA, vFLIP, vCyclin, and vIL-6. These results imply that KSHV infection in NIH 3T3 cells is unstable and is rapidly lost on subsequent culturing. Additionally, we detected an enhancement of autophagy early in infection with KSHV. More interestingly, inhibition of autophagy by Beclin 1 siRNA or 3-methyladenine significantly increased the amount of KSHV-specific DNA in the culture supernatant of NIH 3T3 cells when compared to the group treated with KSHV infection alone, implying that autophagy prevents lytic replication of KSHV. Taken together, our data suggest that autophagy could be one of the cellular mechanisms utilized by host cells to promote viral clearance.




Similar content being viewed by others
References
Chang Y, Cesarman E, Pessin MS, Lee F, Culpepper J, Knowles DM, Moore PS (1994) Identification of herpesvirus-like DNA sequences in AIDS-associated Kaposi’s sarcoma. Science 266(5192):1865–1869
Cesarman E, Chang Y, Moore PS, Said JW, Knowles DM (1995) Kaposi’s sarcoma-associated herpesvirus-like DNA sequences in AIDS-related body-cavity-based lymphomas. N Engl J Med 332(18):1186–1191
Soulier J, Grollet L, Oksenhendler E, Cacoub P, Cazals-Hatem D, Babinet P, d’Agay MF, Clauvel JP, Raphael M, Degos L, Sigaux F (1995) Kaposi’s sarcoma-associated herpesvirus-like DNA sequences in multicentric Castleman’s disease. Blood 86(4):1276–1280
Veettil MV, Bandyopadhyay C, Dutta D, Chandran B (2014) Interaction of KSHV with host cell surface receptors and cell entry. Viruses 6(10):4024–4046
Deng H, Liang Y, Sun R (2007) Regulation of KSHV lytic gene expression. Curr Top Microbiol Immunol 312:157–183
Yang Z, Yan Z, Wood C (2008) Kaposi’s sarcoma-associated herpesvirus transactivator RTA promotes degradation of the repressors to regulate viral lytic replication. J Virol 82(7):3590–3603
Ye F, Lei X, Gao SJ (2011) Mechanisms of Kaposi’s sarcoma-associated herpesvirus latency and reactivation. Adv Virol 2011:193860
Tempera I, Lieberman PM (2010) Chromatin organization of gammaherpesvirus latent genomes. Biochim Biophys Acta 1799(3–4):236–245
Dittmer D, Lagunoff M, Renne R, Staskus K, Haase A, Ganem D (1998) A cluster of latently expressed genes in Kaposi’s sarcoma-associated herpesvirus. J Virol 72(10):8309–8315
Rainbow L, Platt GM, Simpson GR, Sarid R, Gao SJ, Stoiber H, Herrington CS, Moore PS, Schulz TF (1997) The 222- to 234-kilodalton latent nuclear protein (LNA) of Kaposi’s sarcoma-associated herpesvirus (human herpesvirus 8) is encoded by orf73 and is a component of the latency-associated nuclear antigen. J Virol 71(8):5915–5921
Sarid R, Flore O, Bohenzky RA, Chang Y, Moore PS (1998) Transcription mapping of the Kaposi’s sarcoma-associated herpesvirus (human herpesvirus 8) genome in a body cavity-based lymphoma cell line (BC-1). J Virol 72(2):1005–1012
Bechtel JT, Liang Y, Hvidding J, Ganem D (2003) Host range of Kaposi’s sarcoma-associated herpesvirus in cultured cells. J Virol 77(11):6474–6481
Akula SM, Naranatt PP, Walia NS, Wang FZ, Fegley B, Chandran B (2003) Kaposi’s sarcoma-associated herpesvirus (human herpesvirus 8) infection of human fibroblast cells occurs through endocytosis. J Virol 77(14):7978–7990
Pertel PE (2002) Human herpesvirus 8 glycoprotein B (gB), gH, and gL can mediate cell fusion. J Virol 76(9):4390–4400
Chaumorcel M, Souquère S, Pierron G, Codogno P, Esclatine A (2007) Human cytomegalovirus controls a new autophagy-dependent cellular antiviral defense mechanism. Autophagy 4(1):46–53
Orvedahl A, Alexander D, Tallóczy Z, Sun Q, Wei Y, Zhang W, Burns D, Leib DA, Levine B (2007) HSV-1 ICP34.5 confers neurovirulence by targeting the Beclin 1 autophagy protein. Cell Host Microbe 1(1):23–35
Orvedahl A, Levine B (2008) Viral evasion of autophagy. Autophagy 4(3):280–285
Kudchodkar SB, Levine B (2009) Viruses and autophagy. Rev Med Virol 19(6):359–378
Leidal AM, Cyr DP, Hill RJ, Lee PWK, McCormick C (2012) Subversion of autophagy by Kaposi’s sarcoma-associated herpesvirus impairs oncogene-induced senescence. Cell Host Microbe 11(2):167–180
Lee DY, Sugden B (2008) The latent membrane protein 1 oncogene modifies B-cell physiology by regulating autophagy. Oncogene 27(20):2833–2842
Wen HJ, Yang Z, Zhou Y, Wood C (2010) Enhancement of autophagy during lytic replication by the Kaposi’s sarcoma-associated herpesvirus replication and transcription activator. J Virol 84(15):7448–7458
Boshoff C, Gao SJ, Healy LE, Matthews S, Thomas AJ, Coignet L, Warnke RA, Strauchen JA, Matutes E, Kamel OW, Moore PS, Weiss RA, Chang Y (1998) Establishing a KSHV+ cell line (BCP-1) from peripheral blood and characterizing its growth in Nod/SCID mice. Blood 91(5):1671–1679
McGeoch DJ, Davison AJ (1999) The descent of human herpesvirus 8. Semin Cancer Biol 9(3):201–209
Poole LJ, Zong JC, Ciufo DM, Alcendor DJ, Cannon JS, Ambinder R, Orenstein JM, Reitz MS, Hayward GS (1999) Comparison of genetic variability at multiple loci across the genomes of the major subtypes of Kaposi’s sarcoma-associated herpesvirus reveals evidence for recombination and for two distinct types of open reading frame K15 alleles at the right-hand end. J Virol 73(8):6646–6660
Miller G, Rigsby MO, Heston L, Grogan E, Sun R, Metroka C, Levy JA, Gao SJ, Chang Y, Moore P (1996) Antibodies to butyrate inducible antigens of Kaposi’s sarcoma-associated herpesvirus in patients with HIV-1 infection. N Engl J Med 334(20):1292–1297
Berenson JR, Vescio RA (1999) HHV-8 is present in multiple myeloma patients. Blood 93(10):3157–3159
Pan L, Milligan L, Michaeli J, Cesarman E, Knowles DM (2001) Polymerase chain reaction detection of Kaposi’s sarcoma-associated herpesvirus-optimized protocols and their application to myeloma. J Mol Diagn 3(1):32–38
Greene W, Kuhne K, Ye F, Chen J, Zhou F, Lei X, Gao SJ (2007) Molecular biology of KSHV in relation to AIDS-associated oncogenesis. Cancer Treat Res 133:69–127
Zhu FX, Cusano T, Yuan Y (1999) Identification of the immediate-early transcripts of Kaposi’s sarcoma-associated herpesvirus. J Virol 73(7):5556–5567
Sun Q, Fan W, Zhong Q (2009) Regulation of Beclin 1 in autophagy. Autophagy 5(5):713–716
Hartford CM, Ratain MJ (2007) Rapamycin: something old, something new, sometimes borrowed and now renewed. Clin Pharmacol Ther 82:381–388
Renne R, Blackbourn D, Whitby D, Levy J, Ganem D (1998) Limited transmission of Kaposi’s sarcoma-associated herpesvirus in cultured cells. J Virol 72(6):5182–5188
Hahn A, Birkmann A, Wies E, Dorer D, Mahr K, Stürzl M, Titgemeyer F, Neipel F (2009) Kaposi’s sarcoma-associated herpesvirus gH/gL: glycoprotein export and interaction with cellular receptors. J Virol 83(1):396–407
Nicholas J, Ruvolo VR, Burns WH, Sandford G, Wan X, Ciufo D, Hendrickson SB, Guo HG, Hayward GS, Reitz MS (1997) Kaposi’s sarcoma-associated human herpesvirus-8 encodes homologues of macrophage inflammatory protein-1 and interleukin-6. Nat Med 3(3):287–292
Mesri EA, Cesarman E, Boshoff C (2010) Kaposi’s sarcoma and its associated herpesvirus. Nat Rev Cancer 10(10):707–719
Lee JS, Li Q, Lee JY, Lee SH, Jeong JH, Lee HR, Chang H, Zhou FC, Gao SJ, Liang C, Jung JU (2009) FLIP-mediated autophagy regulation in cell death control. Nat Cell Biol 11(11):1355–1362
Mijaljica D, Devenish RJ (2013) Nucleophagy at a glance. J Cell Sci 126(Pt 19):4325–4330
English L, Chemali M, Duron J, Rondeau C, Laplante A, Gingras D, Alexander D, Leib D, Norbury C, Lippé R, Desjardins M (2009) Autophagy enhances the presentation of endogenous viral antigens on MHC class I molecules during HSV-1 infection. Nat Immunol 10(5):480–487
Pankiv S, Lamark T, Bruun JA, Øvervatn A, Bjørkøy G, Johansen T (2010) Nucleocytoplasmic shuttling of p62/SQSTM1 and its role in recruitment of nuclear polyubiquitinated proteins to promyelocytic leukemia bodies. J Biol Chem 285(8):5941–5953
Kirkegaard K, Taylor MP, Jackson WT (2004) Cellular autophagy: surrender, avoidance and subversion by microorganisms. Nat Rev Microbiol 2(4):301–304
Levine B (2005) Eating oneself and uninvited guests: autophagy-related pathways in cellular defense. Cell 120(2):159–162
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
Jang, GH., Lee, J., Kim, NY. et al. Suppression of lytic replication of Kaposi’s sarcoma-associated herpesvirus by autophagy during initial infection in NIH 3T3 fibroblasts. Arch Virol 161, 595–604 (2016). https://doi.org/10.1007/s00705-015-2698-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00705-015-2698-2