Abstract
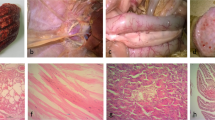
Whilst remarkable progress in elucidating the mechanisms governing interspecies transmission and pathogenicity of highly pathogenic avian influenza viruses (AIVs) has been made, similar studies focusing on low-pathogenic AIVs isolated from the wild waterfowl reservoir are limited. We previously reported that two AIV strains (subtypes H6N2 and H3N8) isolated from wild waterfowl in Zambia harbored some amino acid residues preferentially associated with human influenza virus proteins (so-called human signatures) and replicated better in the lungs of infected mice and caused more morbidity than a strain lacking such residues. To further substantiate these observations, we infected chickens and mice intranasally with AIV strains of various subtypes (H3N6, H3N8, H4N6, H6N2, H9N1 and H11N9) isolated from wild waterfowl in Zambia. Although some strains induced seroconversion, all of the tested strains replicated poorly and were nonpathogenic for chickens. In contrast, most of the strains having human signatures replicated well in the lungs of mice, and one of these strains caused severe illness in mice and induced lung injury that was characterized by a severe accumulation of polymorphonuclear leukocytes. These results suggest that some strains tested in this study may have the potential to infect mammalian hosts directly without adaptation, which might possibly be associated with the possession of human signature residues. Close monitoring and evaluation of host-associated signatures may help to elucidate the prevalence and emergence of AIVs with potential for causing zoonotic infections.



Similar content being viewed by others
References
Alexander DJ (2000) A review of avian influenza in different bird species. Vet Microbiol 74:3–13
Basler CF, Aguilar PV (2008) Progress in identifying virulence determinants of the 1918 H1N1 and the Southeast Asian H5N1 influenza A viruses. Antiviral Res 79:166–178
Berri F, Lê VB, Jandrot-Perrus M, Lina B, Riteau B (2014) Switch from protective to adverse inflammation during influenza: viral determinants and hemostasis are caught as culprits. Cell Mol Life Sci 71:885–898
Berri F, Rimmelzwaan GF, Hanss M, Albina E, Foucault-Grunenwald ML, Lê VB, Vogelzang-van Trierum SE, Gil P, Camerer E, Martinez D, Lina B, Lijnen R, Carmeliet P, Riteau B (2013) Plasminogen controls inflammation and pathogenesis of influenza virus infections via fibrinolysis. PLoS Pathog 9:e1003229
Chen GW, Chang SC, Mok CK, Lo YL, Kung YN, Huang JH, Shih YH, Wang JY, Chiang C, Chen CJ, Shih SR (2006) Genomic signatures of human versus avian influenza A viruses. Emerg Infect Dis 12:1353–1360
Chen GW, Shih SR (2009) Genomic signatures of influenza A pandemic (H1N1) 2009 virus. Emerg Infect Dis 15:1897–1903
Chen Y, Liang W, Yang S, Wu N, Gao H, Sheng J, Yao H, Wo J, Fang Q, Cui D, Li Y, Yao X, Zhang Y, Wu H, Zheng S, Diao H, Xia S, Zhang Y, Chan KH, Tsoi HW, Teng JL, Song W, Wang P, Lau SY, Zheng M, Chan JF, To KK, Chen H, Li L, Yuen KY (2013) Human infections with the emerging avian influenza A H7N9 virus from wet market poultry: clinical analysis and characterisation of viral genome. Lancet 381:1916–1925
Conenello GM, Zamarin D, Perrone LA, Tumpey T, Palese P (2007) A single mutation in the PB1-F2 of H5N1 (HK/97) and 1918 influenza A viruses contributes to increased virulence. PLoS Pathog 3:1414–1421
Driskell EA, Jones CA, Stallknecht DE, Howerth EW, Tompkins SM (2010) Avian influenza virus isolates from wild birds replicate and cause disease in a mouse model of infection. Virology 399:280–289
Finkelstein DB, Mukatira S, Mehta PK, Obenauer JC, Su X, Webster RG, Naeve CW (2007) Persistent host markers in pandemic and H5N1 influenza viruses. J Virol 81:10292–10299
Fujisawa H (2008) Neutrophils play an essential role in cooperation with antibody in both protection against and recovery from pulmonary infection with influenza virus in mice. J Virol 82:2772–2783
Gao R, Cao B, Hu Y, Feng Z, Wang D, Hu W, Chen J, Jie Z, Qiu H, Xu K, Xu X, Lu H, Zhu W, Gao Z, Xiang N, Shen Y, He Z, Gu Y, Zhang Z, Yang Y, Zhao X, Zhou L, Li X, Zou S, Zhang Y, Li X, Yang L, Guo J, Dong J, Li Q, Dong L, Zhu Y, Bai T, Wang S, Hao P, Yang W, Zhang Y, Han J, Yu H, Li D, Gao GF, Wu G, Wang Y, Yuan Z, Shu Y (2013) Human infection with a novel avian-origin influenza A (H7N9) virus. N Engl J Med 368:1888–1897
Gill JS, Webby R, Gilchrist MJR, Gray GC (2006) Avian influenza among waterfowl hunters and wildlife professionals. Emerg Infect Dis 12:1284–1286
Hinshaw VS, Bean WJ, Webster RG, Rehg JE, Fiorelli P, Early G, Geraci JR, St Aubin DJ (1984) Are seals frequently infected with avian influenza viruses? J Virol 51:863–865
Hossain MJ, Hickman D, Perez DR (2008) Evidence of expanded host range and mammalian-associated genetic changes in a duck H9N2 influenza virus following adaptation in quail and chickens. PLoS One 3:e3170
Ichinohe T, Pang IK, Iwasaki A (2010) Influenza virus activates inflammasomes via its intracellular M2 ion channel. Nat Immunol 11:404–410
Joseph T, McAuliffe J, Lu B, Jin H, Kemble G, Subbarao K (2007) Evaluation of replication and pathogenicity of avian influenza a H7 subtype viruses in a mouse model. J Virol 81:10558–10566
Kayali G, Barbour E, Dbaibo G, Tabet C, Saade M, Shaib HA, Debeauchamp J, Webby RJ (2011) Evidence of infection with H4 and H11 avian influenza viruses among Lebanese chicken growers. PLoS One 6:e26818
Khoufache K, Berri F, Nacken W, Vogel AB, Delenne M, Camerer E, Coughlin SR, Carmeliet P, Lina B, Rimmelzwaan GF, Planz O, Ludwig S, Riteau B (2013) PAR1 contributes to influenza A virus pathogenicity in mice. J Clin Invest 123:206–214
Kim HR, Lee YJ, Lee KK, Oem JK, Kim SH, Lee MH, Lee OS, Park CK (2010) Genetic relatedness of H6 subtype avian influenza viruses isolated from wild birds and domestic ducks in Korea and their pathogenicity in animals. J Gen Virol 91:208–219
Klenk HD, Garten W, Matrosovich M (2011) Molecular mechanisms of inter-species transmission and pathogenicity of influenza viruses: lessons from the 2009 pandemic. BioEssays 33:180–188
Myers KP, Setterquist SF, Capuano AW, Gray GC (2007) Infection due to 3 avian influenza subtypes in United States veterinarians. Clin Infect Dis 45:4–9
Narasaraju T, Yang E, Samy RP, Ng HH, Poh WP, Liew AA, Phoon MC, van Rooijen N, Chow VT (2011) Excessive neutrophils and neutrophil extracellular traps contribute to acute lung injury of influenza pneumonitis. Am J Pathol 179:199–210
Olsen B, Munster VJ, Wallensten A, Waldenström J, Osterhaus AD, Fouchier RA (2006) Global patterns of influenza A virus in wild birds. Science 312:384–388
Peiris JS, de Jong MD, Guan Y (2007) Avian influenza virus (H5N1): a threat to human health. Clin Microbiol Rev 20:243–267
Reed LJ, Muench H (1938) A simple method of estimating fi 354 fty percent endpoints. Am J Hyg 27:493–497
Shaw M, Cooper L, Xu X, Thompson W, Krauss S, Guan Y, Zhou N, Klimov A, Cox N, Webster R, Lim W, Shortridge K, Subbarao K (2002) Molecular changes associated with the transmission of avian influenza a H5N1 and H9N2 viruses to humans. J Med Virol 66:107–114
Simulundu E, Ishii A, Igarashi M, Mweene AS, Suzuki Y, Hang’ombe BM, Namangala B, Moonga L, Manzoor R, Ito K, Nakamura I, Sawa H, Sugimoto C, Kida H, Simukonda C, Chansa W, Chulu J, Takada A (2011) Characterization of influenza A viruses isolated from wild waterfowl in Zambia. J Gen Virol 92:1416–1427
Simulundu E, Mweene AS, Tomabechi D, Hang’ombe BM, Ishii A, Suzuki Y, Nakamura I, Sawa H, Sugimoto C, Ito K, Kida H, Saiwana L, Takada A (2009) Characterization of H3N6 avian influenza virus isolated from a wild white pelican in Zambia. Arch Virol 154:1517–1522
Song D, Kang B, Lee C, Jung K, Ha G, Kang D, Park S, Park B, Oh J (2008) Transmission of avian influenza virus (H3N2) to dogs. Emerg Infect Dis 14:741–746
Tumpey TM, García-Sastre A, Taubenberger JK, Palese P, Swayne DE, Pantin-Jackwood MJ, Schultz-Cherry S, Solórzano A, Van Rooijen N, Katz JM, Basler CF (2005) Pathogenicity of influenza viruses with genes from the 1918 pandemic virus: functional roles of alveolar macrophages and neutrophils in limiting virus replication and mortality in mice. J Virol 79:14933–14944
Wareing MD, Lyon AB, Lu B, Gerard C, Sarawar SR (2004) Chemokine expression during the development and resolution of a pulmonary leukocyte response to influenza A virus infection in mice. J Leukoc Biol 76:886–895
Webster RG, Bean WJ, Gorman OT, Chambers TM, Kawaoka Y (1992) Evolution and ecology of influenza A viruses. Microbiol Rev 56:152–179
Acknowledgments
We thank Hiroko Miyamoto and Mari Ishijima (Hokkaido University Research Center for Zoonosis Control) for excellent technical assistance. We are also grateful to Dr. Mathew Nyirenda and Dr. Collins Ateba (North West University, Mafikeng Campus) for proofreading the manuscript, and Dr. Yuji Sunden (Graduate School of Veterinary Medicine, Hokkaido University) for kindly supporting pathological studies. This work was supported by the Japan Initiative for Global Research Network on Infectious Diseases (J-GRID) and the Global COE Program ‘Establishment of International Collaboration Centers for Zoonosis Control’ from the Ministry of Education, Culture, Sports, Science and Technology (MEXT), Japan, as well as the Japan Science and Technology Agency (JST)/Japan International Cooperation Agency (JICA) within the framework of the Science and Technology Research Partnership for Sustainable Development (SATREPS).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Simulundu, E., Nao, N., Yabe, J. et al. The zoonotic potential of avian influenza viruses isolated from wild waterfowl in Zambia. Arch Virol 159, 2633–2640 (2014). https://doi.org/10.1007/s00705-014-2124-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00705-014-2124-1