Abstract
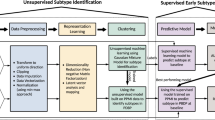
Responsiveness to levodopa varies greatly among patients with Parkinson’s disease (PD). The factors that affect it are ill defined. The aim of the study was to identify factors predictive of long-term response to levodopa. The medical records of 296 patients with PD (mean age of onset, 62.2 ± 9.7 years) were screened for demographics, previous treatments, and clinical phenotypes. All patients were assessed with the Unified PD Rating Scale (UPDRS)-III before and 3 months after levodopa initiation. Regression and machine-learning analyses were used to determine factors that are associated with levodopa responsiveness and might identify patients who will benefit from treatment. The UPDRS-III score improved by ≥ 30% (good response) in 128 patients (43%). On regression analysis, female gender, young age at onset, and early use of dopamine agonists predicted a good response. Time to initiation of levodopa treatment had no effect on responsiveness except in patients older than 72 years, who were less responsive. Machine-learning analysis validated these factors and added several others: symptoms of rigidity and bradykinesia, disease onset in the legs and on the left side, and fewer white vascular ischemic changes, comorbidities, and pre-non-motor symptoms. The main determinants of variations in levodopa responsiveness are gender, age, and clinical phenotype. Early use of dopamine agonists does not hamper levodopa responsiveness. In addition to validating the regression analysis results, machine-learning methods helped to determine the specific clinical phenotype of patients who may benefit from levodopa in terms of comorbidities and pre-motor and non-motor symptoms.



Similar content being viewed by others
References
Antonini A, Vitale C, Barone P, Cilia R, Righini A, Bonuccelli U, Abbruzzese G, Ramat S, Petrone A, Quatrale R, Marconi R, Ceravolo R, Stefani A, Lopiano L, Zappia M, Capus L, Morgante L, Tamma F, Tinazzi M, Colosimo C, Guerra UP (2012) The relationship between cerebral vascular disease and parkinsonism: The VADO study. Parkinsonism Relat Disord 18:775–780
Arena JE, Cerquetti D, Rossi M, Chaves H, Rollan C, Dossi DE, Merello M (2016) Influence of white matter MRI hyper-intensities on acute L-dopa response in patients with Parkinson’s disease. Parkinsonism Relat Disord 24:126–128. https://doi.org/10.1016/j.parkreldis.2016.01.017
Armananzas R, Bielza C, Chaudhuri KR, Martinez-Martin P, Larranaga P (2013) Unveiling relevant non-motor Parkinson’s disease severity symptoms using a machine learning approach. Artif Intell Med 58:195–202. https://doi.org/10.1016/j.artmed.2013.04.002
Avisar H, Guardia-Laguarta C, Area-Gomez E, Surface M, Chan AK, Alcalay RN, Lerner B (2021) Lipidomics prediction of Parkinson’s disease severity: A machine-learning analysis. J Parkinsons Dis 11:1141–1155. https://doi.org/10.3233/JPD-202476
Avisar H, Guardia-Laguarta C, Surface M, Papagiannakis N, Maniati M, Antonellou R, Papadimitriou D, Koros C, Athanassiadou A, Przedborski S, Lerner B, Stefanis L, Area-Gomez E, Alcalay RN (2022) Lipid level alteration in human and cellular models of alpha synuclein mutations. NPJ Parkinsons Dis 8:52. https://doi.org/10.1038/s41531-022-00313-y
Bind S, Tiwari AK, Sahani AK (2015) A survey of machine learning based approaches for Parkinson disease prediction. Int J Comput Sci Inf Technol 6:1648–1655
Cilia R, Cereda E, Akpalu A, Sarfo FS, Cham M, Laryea R, Obese V, Oppon K, Del Sorbo F, Bonvegna S, Zecchinelli AL, Pezzoli G (2020) Natural history of motor symptoms in Parkinson’s disease and the long-duration response to levodopa. Brain 143:2490–2501. https://doi.org/10.1093/brain/awaa181
Davidson MB, McGhee DJ, Counsell CE (2012) Comparison of patient rated treatment response with measured improvement in Parkinson’s disease. J Neurol Neurosurg Psychiatry 83:1001–1005. https://doi.org/10.1136/jnnp-2012-302741
Doder M, Rabiner EA, Turjanski N, Lees AJ, Brooks DJ (2003) Tremor in Parkinson’s disease and serotonergic dysfunction: an 11C-WAY 100635 PET study. Neurology 60:601–605. https://doi.org/10.1212/01.wnl.0000031424.51127.2b
Duda R, Hart P, Stork D (2001) Pattern Classification, 2nd edn. Wiley, New York
Gordon J, Lerner B (2019) Insights into amyotrophic lateral sclerosis from a machine learning perspective. J Clin Med 8:1578. https://doi.org/10.3390/jcm8101578
Gower JC (1971) A general coefficient of similarity and some of its properties. Biometrics 27:857–871
Guin D, Mishra MK, Talwar P, Rawat C, Kushwaha SS, Kukreti S, Kukreti R (2017) Asystematic review and integrative approach to decode the common molecular link between levodopa response and Parkinson’s disease. BMC Med Genom 10:56
Hauser RA, Auinger P, Oakes D, Parkinson Study Group (2009) Levodopa response in early Parkinson’s disease. Mov Disord 24:2328–2336. https://doi.org/10.1002/mds.22759
Helmich RC, Janssen MJ, Oyen WJ, Bloem BRI, Toni I (2011) Pallidal dysfunction drives a cerebellothalamic circuit into Parkinson tremor. Ann Neurol 69:269–281. https://doi.org/10.1002/ana.22361
Hughes AJ, Daniel SE, Kilford L, Lees AJ (1992) Accuracy of clinical diagnosis of idiopathic Parkinson’s disease. A clinico-pathological study of 100 cases. JNNP 55:181–184
Imbach LL, Sommerauer M, Leuenberger K, Schreglmann SR, Maier O, Uhl M, Gassert R, Baumann CR (2014) Dopamine-responsive pattern in tremor patients. Parkinsonism Relat Disord 20:1283–1286. https://doi.org/10.1016/j.parkreldis.2014.09.007
Isaias IU, Marzegan A, Pezzoli G, Marotta G, Canesi M, Biella GE, Volkmann J, Cavallari P (2012) A role for locus coeruleus in Parkinson tremor. Front Hum Neurosci 5:179. https://doi.org/10.3389/fnhum.2011.00179
Lyons KE, Hubble JP, Troster AI, Pahwa R, Koller WC (1998) Gender differences in Parkinson’s disease. Clin Neuropharmacol 21:118–121. https://doi.org/10.1136/jnnp.2006.103788
Malek N, Kanavou S, Lawton MA, Pitz V, Grosset KA, Bajaj N, Barker RA, Ben-Shlomo Y, Burn DJ, Foltynie T, Hardy J, Williams NM, Wood N, Morris HR, Grosset DG, ProBaND clinical consortium, (2019) L-dopa responsiveness in early Parkinson’s disease is associated with the rate of motor progression. Parkinsonism Relat Disord 65:55–61. https://doi.org/10.1016/j.parkreldis.2019.05.022
Martin WRW, Miles M, Zhong Q, Hartlein J, Racette BA, Norris SA, Ushe M, Maiti B, Criswell S, Davis AA, Kotzbauer PT, Cairns NJ, Perrin RJ, Perlmutter JS (2021) Is levodopa response a valid indicator of Parkinson’s disease? Mov Disord 36:948–954. https://doi.org/10.1002/mds.28406
Merello M, Nouzeilles MI, Arce GP, Leiguarda R (2002) Accuracy of term levodopa response as a major criterion for idiopathic Parkinson’s disease diagnosis. Mov Disord 17:795–798. https://doi.org/10.1002/mds.10123
Merello M, Gerschcovich ER, Ballesteros D, Cerquetti D (2011) Correlation between the movement disorders society unified Parkinson’s disease rating scale (MDS-UPDRS) and the Unified Parkinson’s Disease rating scale (UPDRS) during L-dopa acute challenge. Parkinsonism Relat Disord 17:705–707. https://doi.org/10.1016/j.parkreldis.2011.07.002
Pitz V, Malek N, Tobias ES, Grosset KA, Gentleman S, Grosset DG (2020) The levodopa response varies in pathologically confirmed Parkinson’s disease: a systematic review. Mov Disord Clin Pract 7:218–222. https://doi.org/10.1002/mdc3.12885
Qamhawi Z, Towey D, Shah B, Pagano G, Seibyl J, Marek K, Borghammer P, Brooks DJ, Pavese N (2015) Clinical correlates of raphe serotonergic dysfunction in early Parkinson’s disease. Brain 138(Pt 10):2964–2973. https://doi.org/10.1093/brain/awv215
Reilly DK, Rivera-Calimlim L, Van Dyke D (1980) Catechol-O-methyltransferase activity: a determinant of levodopa response. Clin Pharmacol Ther 28:278–286. https://doi.org/10.1038/clpt.1980.161
Rousseeuw PJ (1987) Silhouettes: a graphical aid to the interpretation and validation of cluster analysis. J Comput Appl Math 20:53–65. https://doi.org/10.1016/0377-0427(87)90125-7
Sampaio TF, Dos Santos EUD, de Lima GDC, Dos Anjos RSG, da Silva RC, Asano AGC, Asano NMJ, Crovella S, de Souza PRE (2018) MAO-B and COMT genetic variations associated with levodopa treatment response in patients with Parkinson’s disease. J Clin Pharmacol 58:920–926. https://doi.org/10.1002/jcph.1096
Saranza G, Lang AE (2021) Levodopa challenge test: indications, protocol, and guide. J Neurol 268:3135–3143. https://doi.org/10.1007/s00415-020-09810-7
Schade S, Sixel-Döring F, Ebentheuer J, Schulz X, Trenkwalder C, Mollenhauer B (2017) Acute levodopa challenge test in patients with de novo Parkinson’s disease: data from the DeNoPa cohort. Mov Disord Clin Pract 4:755–762. https://doi.org/10.1002/mdc3.12511
Sriram TVS, Rao MV, Narayana GVS, Kaladhar DSVGK, Vital TPR (2013) Intelligent Parkinson disease prediction using machine learning algorithms. Int J Eng Innov Technol 3:212–215
Sung YH, Chung SJ, Kim SR, Lee MC (2008) Factors predicting response to dopaminergic treatment for resting tremor of Parkinson’s disease. Mov Disord 23:137–140. https://doi.org/10.1002/mds.21793
Tahir N, Manap HH (2012) Parkinson disease gait classification based on machine learning approach. J Appl Sci 12:180–185. https://doi.org/10.3923/jas.2012.180.185
Tsanas A, Little MA, McSharry PE, Spielman J, Ramig LO (2012) Novel speech signal processing algorithms for high-accuracy classification of Parkinson’s disease. IEEE Trans Biomed Eng 59:1264–1271
Velseboer DC, Broeders M, Post B, van Geloven N, Speelman JD, Schmand B, de Haan RJ, de Bie RMA, CARPA Study Group (2013) Prognostic factors of motor impairment, disability, and quality of life in newly diagnosed PD. Neurology 80:627–633. https://doi.org/10.1212/WNL.0b013e318281cc99
Verschuur CVM, Suwijn SR, Boel JA, Post B, Bloem BR, van Hilten JJ, van Laar T, Tissingh G, Munts AG, Deuschl G, Lang AE, Dijkgraaf MGW, de Haan RJ, de Bie RMA, LEAP Study Group (2019) Randomized delayed-start trial of levodopa in Parkinson’s disease. N Engl J Med 380:315–324. https://doi.org/10.1056/NEJMoa1809983
Wickremaratchi MM, Ben-Shlomo Y, Morris HR (2009) The effect of onset age on the clinical features of Parkinson’s disease. Eur J Neurol 16:450–456. https://doi.org/10.1111/j.1468-1331.2008.02514.x
Zach H, Dirkx MF, Roth D, Pasman JW, Bloem BR, Helmich RC (2020) Dopamine-responsive and dopamine-resistant resting tremor in Parkinson disease. Neurology 95:e1461–e1470. https://doi.org/10.1212/WNL.0000000000010316
Zappia M, Montesanti R, Colao R, Branca D, Nicoletti G, Aguglia U, Quattrone A (1997) Short-term levodopa test assessed by movement time accurately predicts dopaminergic responsiveness in Parkinson’s disease. Mov Disord 12:103–106. https://doi.org/10.1002/mds.870120118
Funding
The authors have no relevant financial or non-financial interests to disclose.
Author information
Authors and Affiliations
Contributions
(1) Research project: A. Conception, B. Organization, C. Execution; (2) Experimental procedures and statistical analysis: A. Design, B. Execution, C. Review and critique; (3) Manuscript: A. Writing of the first draft, B. Review and critique. RD: 1A, 1B, 1C, 2C, 3A, and 3B. BH: 2A, 2B, 2C, 3A, and 3B. JR: 1B, 1C, and 3B. BAK: 1B, 1C, and 3B. BL: 1A, 1C, 2A, 2B, 2C, 3A, and 3B.
Corresponding author
Ethics declarations
Conflict of interest
None.
Ethics approval
The authors confirm that all procedures involving experiments on human subjects were done in accord with the ethical standards of the Committee on Human Experimentation of the institution in which the experiments were done or in accord with the Helsinki Declaration of 1975.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Djaldetti, R., Hadad, B., Reiner, J. et al. Levodopa responsiveness in Parkinson’s disease: harnessing real-life experience with machine-learning analysis. J Neural Transm 129, 1289–1297 (2022). https://doi.org/10.1007/s00702-022-02540-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00702-022-02540-2