Abstract
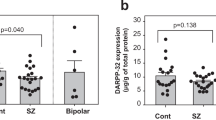
Bipolar disorder shares symptoms and pathological pathways with other neurodegenerative diseases, including frontotemporal dementia (FTD). Since TAR DNA-binding protein 43 (TDP-43) is a neuropathological marker of frontotemporal dementia and it is involved in synaptic transmission, we explored the role of TDP-43 as a molecular feature of bipolar disorder (BD). Homogenates were acquired from frozen hippocampus of postmortem brains of bipolar disorder subjects. TDP-43 levels were quantified using an ELISA-sandwich method and compared between the postmortem brains of bipolar disorder subjects and age-matched control group. We found higher levels of TDP-43 protein in the hippocampus of BD (n = 15) subjects, when compared to controls (n = 15). We did not find associations of TDP-43 with age at death, postmortem interval, or age of disease onset. Our results suggest that protein TDP-43 may be potentially implicated in behavioral abnormalities seen in BD. Further investigation is needed to validate these findings and to examine the role of this protein during the disease course and mood states.


Similar content being viewed by others
Availability of data and material
Data have not been published elsewhere and available on request from the authors.
Code availability
Not applicable.
References
American Psychiatry Association (2013) Diagnostic and statistical manual of mental disorders, 5th edn. APA, Washington, DC
Arloth J, Bogdan R, Weber P, Frishman G, Menke A, Wagner KV, Major Depressive Disorder Working Group of the Psychiatric Genomics Consortium, P. G. C. (2015) Genetic differences in the immediate transcriptome response to stress predict risk-related brain function and psychiatric disorders. Neuron 86(5):1189–1202. https://doi.org/10.1016/j.neuron.2015.05.034
Bauer IE, Soares JC, Selek S, Meyer TD (2017) The link between refractoriness and neuroprogression in treatment-resistant bipolar disorder. Mod Trends Pharmacopsychiatry 31:10–26. https://doi.org/10.1159/000470803
Berk M, Kapczinski F, Andreazza AC, Dean OM, Giorlando F, Maes M, Malhi GS (2011) Pathways underlying neuroprogression in bipolar disorder: focus on inflammation, oxidative stress and neurotrophic factors. Neurosci Biobehav Rev 35(3):804–817. https://doi.org/10.1016/j.neubiorev.2010.10.001
Bott NT, Radke A, Stephens ML, Kramer JH (2014) Frontotemporal dementia: diagnosis, deficits and management. Neurodegener Dis Manage 4(6):439–454. https://doi.org/10.2217/nmt.14.34
Cao B, Passos IC, Mwangi B, Amaral-Silva H, Tannous J, Wu MJ, Soares JC (2017) Hippocampal subfield volumes in mood disorders. Mol Psychiatry 22(9):1352–1358. https://doi.org/10.1038/mp.2016.262
Carvalho AF, Firth J, Vieta E (2020) Bipolar disorder. N Engl J Med 383(1):58–66. https://doi.org/10.1056/NEJMra1906193
Chiang PM, Ling J, Jeong YH, Price DL, Aja SM, Wong PC (2010) Deletion of TDP-43 down-regulates Tbc1d1, a gene linked to obesity, and alters body fat metabolism. Proc Natl Acad Sci USA 107(37):16320–16324. https://doi.org/10.1073/pnas.1002176107
Clelland CL, Read LL, Panek LJ, Nadrich RH, Bancroft C, Clelland JD (2013) Utilization of never-medicated bipolar disorder patients towards development and validation of a peripheral biomarker profile. PLoS ONE 8(6):e69082. https://doi.org/10.1371/journal.pone.0069082
Cohen TJ, Lee VM, Trojanowski JQ (2011) TDP-43 functions and pathogenic mechanisms implicated in TDP-43 proteinopathies. Trends Mol Med 17(11):659–667. https://doi.org/10.1016/j.molmed.2011.06.004
Cullen B, Smith DJ, Deary IJ, Evans JJ, Pell JP (2017) The “cognitive footprint” of psychiatric and neurological conditions: cross-sectional study in the UK Biobank cohort. Acta Psychiatr Scand 135(6):593–605. https://doi.org/10.1111/acps.12733
Cummings JL (1997) The Neuropsychiatric Inventory: assessing psychopathology in dementia patients. Neurology 48(5 Suppl 6):S10-16
Darby MM, Yolken RH, Sabunciyan S (2016) Consistently altered expression of gene sets in postmortem brains of individuals with major psychiatric disorders. Transl Psychiatry 6(9):e890. https://doi.org/10.1038/tp.2016.173
de Oliveira KC, Nery FG, Ferreti RE, Lima MC, Cappi C, Machado-Lima A, Grinberg LT (2012) Brazilian psychiatric brain bank: a new contribution tool to network studies. Cell Tissue Bank 13(2):315–326. https://doi.org/10.1007/s10561-011-9258-0
Endo R, Takashima N, Nekooki-Machida Y, Komi Y, Hui KK, Takao M, Tanaka M (2018) TAR DNA-binding protein 43 and disrupted in schizophrenia 1 coaggregation disrupts dendritic local translation and mental function in frontotemporal lobar degeneration. Biol Psychiatry. https://doi.org/10.1016/j.biopsych.2018.03.008
Ferretti REL, Damin AE, Brucki SMD, Morillo LS, Perroco TR, Campora F, Nitrini R (2010) Post-mortem diagnosis of dementia by informant interview. Dement Neuropsychol 4(2):138–144. https://doi.org/10.1590/S1980-57642010DN40200011
Franceschini A, Lin J, von Mering C, Jensen LJ (2016) SVD-phy: improved prediction of protein functional associations through singular value decomposition of phylogenetic profiles. Bioinformatics 32(7):1085–1087. https://doi.org/10.1093/bioinformatics/btv696
Fries GR, Quevedo J, Zeni CP, Kazimi IF, Zunta-Soares G, Spiker DE, Soares JC (2017) Integrated transcriptome and methylome analysis in youth at high risk for bipolar disorder: a preliminary analysis. Transl Psychiatry 7(3):e1059. https://doi.org/10.1038/tp.2017.32
Gandal MJ, Zhang P, Hadjimichael E, Walker RL, Chen C, Liu S, Geschwind DH (2018) Transcriptome-wide isoform-level dysregulation in ASD, schizophrenia, and bipolar disorder. Science 362(6420):eaat8127. https://doi.org/10.1126/science.aat8127
Handley EE, Pitman KA, Dawkins E, Young KM, Clark RM, Jiang TC, Blizzard CA (2017) Synapse dysfunction of layer V pyramidal neurons precedes neurodegeneration in a mouse model of TDP-43 proteinopathies. Cereb Cortex 27(7):3630–3647. https://doi.org/10.1093/cercor/bhw185
Harrison JK, Stott DJ, McShane R, Noel-Storr AH, Swann-Price RS, Quinn TJ (2016) Informant questionnaire on cognitive decline in the elderly (IQCODE) for the early diagnosis of dementia across a variety of healthcare settings. Cochrane Database Syst Rev 11:CD011333. https://doi.org/10.1002/14651858.CD011333.pub2
Heyburn L, Hebron ML, Smith J, Winston C, Bechara J, Li Z, Moussa CE (2016) Tyrosine kinase inhibition reverses TDP-43 effects on synaptic protein expression, astrocytic function and amino acid dis-homeostasis. J Neurochem 139(4):610–623. https://doi.org/10.1111/jnc.13763
Ichikawa T, Baba H, Maeshima H, Shimano T, Inoue M, Ishiguro M, Arai H (2019) Serum levels of TDP-43 in late-life patients with depressive episode. J Affect Disord 250:284–288. https://doi.org/10.1016/j.jad.2019.03.024
Kapczinski F, Magalhaes PV, Balanza-Martinez V, Dias VV, Frangou S, Gama CS, Berk M (2014) Staging systems in bipolar disorder: an International Society for Bipolar Disorders Task Force Report. Acta Psychiatr Scand 130(5):354–363. https://doi.org/10.1111/acps.12305
Kim HK, Nunes PV, Oliveira KC, Young LT, Lafer B (2016) Neuropathological relationship between major depression and dementia: a hypothetical model and review. Prog Neuropsychopharmacol Biol Psychiatry 67:51–57. https://doi.org/10.1016/j.pnpbp.2016.01.008
Lee Y, Zhang Y, Kim S, Han K (2018) Excitatory and inhibitory synaptic dysfunction in mania: an emerging hypothesis from animal model studies. Exp Mol Med 50(4):12. https://doi.org/10.1038/s12276-018-0028-y
Morris JC (1993) The clinical dementia rating (CDR): current version and scoring rules. Neurology 43(11):2412–2414
Nascimento C, Nunes PV, Rodriguez RD, Takada L, Suemoto CK, Grinberg LT, Lafer B (2019) A review on shared clinical and molecular mechanisms between bipolar disorder and frontotemporal dementia. Progr Neuropsychopharmacol Biol Psychiatry. https://doi.org/10.1016/j.pnpbp.2019.04.008
Neumann M, Sampathu DM, Kwong LK, Truax AC, Micsenyi MC, Chou TT, Lee VM (2006) Ubiquitinated TDP-43 in frontotemporal lobar degeneration and amyotrophic lateral sclerosis. Science 314(5796):130–133. https://doi.org/10.1126/science.1134108
Passos IC, Mwangi B, Vieta E, Berk M, Kapczinski F (2016) Areas of controversy in neuroprogression in bipolar disorder. Acta Psychiatr Scand 134(2):91–103. https://doi.org/10.1111/acps.12581
Ratti A, Buratti E (2016) Physiological functions and pathobiology of TDP-43 and FUS/TLS proteins. J Neurochem 138(Suppl 1):95–111. https://doi.org/10.1111/jnc.13625
Schubert KO, Focking M, Cotter DR (2015) Proteomic pathway analysis of the hippocampus in schizophrenia and bipolar affective disorder implicates 14–3-3 signaling, aryl hydrocarbon receptor signaling, and glucose metabolism: potential roles in GABAergic interneuron pathology. Schizophr Res 167(1–3):64–72. https://doi.org/10.1016/j.schres.2015.02.002
Spitzer R, Gibbon M, Williams J (1995) Structured clinical interview for axis I DSM-IV disorders (SCID). American Psychiatric, Washington, DC
Stallings NR, Puttaparthi K, Dowling KJ, Luther CM, Burns DK, Davis K, Elliott JL (2013) TDP-43, an ALS linked protein, regulates fat deposition and glucose homeostasis. PLoS ONE 8(8):e71793. https://doi.org/10.1371/journal.pone.0071793
Suemoto CK, Ferretti-Rebustini RE, Rodriguez RD, Leite RE, Soterio L, Brucki SM, Grinberg LT (2017) Neuropathological diagnoses and clinical correlates in older adults in Brazil: a cross-sectional study. PLoS Med 14(3):e1002267. https://doi.org/10.1371/journal.pmed.1002267
Szklarczyk D, Gable AL, Lyon D, Junge A, Wyder S, Huerta-Cepas J, Mering CV (2019) STRING v11: protein-protein association networks with increased coverage, supporting functional discovery in genome-wide experimental datasets. Nucleic Acids Res 47(D1):D607–D613. https://doi.org/10.1093/nar/gky1131
Taylor JB, Prager LM, Quijije NV, Schaefer PW (2018) Case 21–2018: a 61-year-old man with grandiosity, impulsivity, and decreased sleep. N Engl J Med 379(2):182–189. https://doi.org/10.1056/NEJMcpc1712229
Velakoulis D, Walterfang M, Mocellin R, Pantelis C, Dean B, McLean C (2009) Abnormal hippocampal distribution of TDP-43 in patients with-late onset psychosis. Aust N Z J Psychiatry 43(8):739–745. https://doi.org/10.1080/00048670903001984
Vieta E, Berk M, Schulze TG, Carvalho AF, Suppes T, Calabrese JR, Grande I (2018) Bipolar disorders. Nat Rev Dis Primers 4:18008. https://doi.org/10.1038/nrdp.2018.8
Witt SH, Juraeva D, Sticht C, Strohmaier J, Meier S, Treutlein J, Rietschel M (2014) Investigation of manic and euthymic episodes identifies state- and trait-specific gene expression and STAB1 as a new candidate gene for bipolar disorder. Transl Psychiatry 4:e426. https://doi.org/10.1038/tp.2014.71
Woolley JD, Khan BK, Murthy NK, Miller BL, Rankin KP (2011) The diagnostic challenge of psychiatric symptoms in neurodegenerative disease: rates of and risk factors for prior psychiatric diagnosis in patients with early neurodegenerative disease. J Clin Psychiatry 72(2):126–133. https://doi.org/10.4088/JCP.10m06382oli
Acknowledgements
This study was supported by Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq) [grant number 466763/2014-0] and by a generous private donation from Suzana and Carlos Melzer to the USP Bipolar Disorder Research Program (PROMAN). We would like to thank the families of the subjects that donated the brains and provided all information. Camila Nascimento was supported by a post-doctoral scholarship from Fundação de Amparo à Pesquisa do Estado de São Paulo (FAPESP) [grant number 2017/07089-8].
Funding
This study was supported by Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq) [grant number 466763/2014-0] and by a generous private donation from Suzana and Carlos Melzer to the USP Bipolar Disorder Research Program (PROMAN). Camila Nascimento was supported by a post-doctoral scholarship from Fundação de Amparo à Pesquisa do Estado de São Paulo (FAPESP) [grant number 2017/07089-8].
Author information
Authors and Affiliations
Contributions
All the authors contributed to the study conception and design. Experiment conduction, material preparation, data collection and analysis were performed by CN. PVN was responsible for the diagnostic of the bipolar disorder subjects and HK contributed with experiment conduction and analysis. The first draft of the manuscript was written by CN and all the authors commented on previous versions of the manuscript. All the authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors have no conflicts of interest to declare that are relevant to the content of this article.
Ethics approval
Approval was obtained from the ethics committee of University of Sao Paulo. The procedures used in this study adhere to the tenets of the Declaration of Helsinki.
Consent to participate
Informed consent was obtained from all next of kin of the individual participants included in the study.
Consent for publication
Next of kin of all participants signed informed consent regarding publishing data generated in the brain tissue.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Nascimento, C., Nunes, P.V., Kim, H.K. et al. Increased levels of TAR DNA-binding protein 43 in the hippocampus of subjects with bipolar disorder: a postmortem study. J Neural Transm 129, 95–103 (2022). https://doi.org/10.1007/s00702-021-02455-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00702-021-02455-4