Abstract
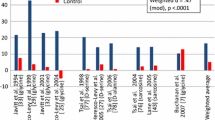
d-Cycloserine is a partial agonist at the glycine site of the N-methyl-D-aspartate (NMDA) receptor. Results have been inconsistent in trials on the efficacy of d-Cycloserine in patients with schizophrenia. We examined the efficacy of d-Cycloserine against negative and cognitive symptoms (primary and co-primary outcomes). Secondary outcomes were efficacy of d-Cycloserine against positive symptoms and the examination of early treatment outcomes. A systematic literature search was carried out using following selection criteria: Population = Patients with Schizophrenia; Intervention = Trials using d-Cycloserine either as monotherapy or adjuvant therapy; Comparison = Placebo or active comparator; Outcome = Change in negative symptoms, cognitive symptoms and positive symptoms; Study design = Randomized controlled trials with parallel design. We used the Cochrane Collaboration tool for risk of bias for study quality appraisal. Effect sizes for trials were calculated separately for negative, positive and cognitive symptom dimensions using the DerSimonian–Laird random effects model. Seven studies (pooled N = 413) provided data for meta-analysis. The pooled Standardized Mean Difference (SMD) for negative, cognitive, and positive symptom change scores were − 0.32 (95% CI, − 0.75 to 0.11), − 0.05 (95% CI, − 0.91 to 0.81), and − 0.08 (95% CI, − 0.37 to 0.20), respectively. No significant improvement was noted with regard to early outcome. I2 values for heterogeneity were 61%, 67%, and 0% for studies assessing negative, cognitive, and positive symptom ratings, respectively. d-Cycloserine did not exhibit significant efficacy in treating negative, cognitive, or positive symptoms of schizophrenia at either study-defined endpoint (4–36 weeks) or at four weeks (early outcome).





Similar content being viewed by others
References
Bennett M (2009) Positive and negative symptoms in schizophrenia: the NMDA receptor hypofunction hypothesis, neuregulin/ErbB4 and synapse regression. Aust N Z J Psychiatry 43:711–721. https://doi.org/10.1080/00048670903001943
Buchanan RW, Javitt DC, Marder SR et al (2007) The Cognitive and Negative Symptoms in Schizophrenia Trial (CONSIST): the efficacy of glutamatergic agents for negative symptoms and cognitive impairments. Am J Psychiatry 164:1593–1602. https://doi.org/10.1176/appi.ajp.2007.06081358
Cain CK, McCue M, Bello I et al (2014) d-Cycloserine augmentation of cognitive remediation in schizophrenia. Schizophr Res 153:177–183. https://doi.org/10.1016/j.schres.2014.01.016
Chang CH, Lin CH, Liu CY et al (2020) Efficacy and cognitive effect of sarcosine (N-methylglycine) in patients with schizophrenia: a systematic review and meta-analysis of double-blind randomised controlled trials. J Psychopharmacol 34:495–505. https://doi.org/10.1177/0269881120908016
Coyle JT, Tsai G, Goff D (2003) Converging evidence of NMDA receptor hypofunction in the pathophysiology of schizophrenia. Ann N Y Acad Sci 1003:318–327. https://doi.org/10.1196/annals.1300.020
Dall’Olio R, Gaggi R, Bonfante V, Gandolfi O (1999) The non-competitive NMDA receptor blocker dizocilpine potentiates serotonergic function. Behav Pharmacol 10:63–71. https://doi.org/10.1097/00008877-199902000-00006
Dall’Olio R, Gandolfi O, Gaggi R (2000) d-Cycloserine, a positive modulator of NMDA receptors, inhibits serotonergic function. Behav Pharmacol 11:631–637. https://doi.org/10.1097/00008877-200011000-00010
Danysz W, Parsons CG (1998) Glycine and N-methyl-d-aspartate receptors: physiological significance and possible therapeutic applications. Pharmacol Rev 50:597–664
Diminich ED, Dickerson F, Bello I et al (2020) d-cycloserine augmentation of cognitive behavioral therapy for delusions: a randomized clinical trial. Schizophr Res 222:145–152. https://doi.org/10.1016/j.schres.2020.06.015
Duncan EJ, Szilagyi S, Schwartz MP et al (2004) Effects of d-cycloserine on negative symptoms in schizophrenia. Schizophr Res 71:239–248. https://doi.org/10.1016/j.schres.2004.03.013
Forsyth JK, Bachman P, Mathalon DH et al (2017) Effects of augmenting N-methyl-d-aspartate receptor signaling on working memory and experience-dependent plasticity in schizophrenia: an exploratory study using acute d-cycloserine. Schizophr Bull 43:1123–1133. https://doi.org/10.1093/schbul/sbw193
Gilmour G, Dix S, Fellini L et al (2012) NMDA receptors, cognition and schizophrenia–testing the validity of the NMDA receptor hypofunction hypothesis. Neuropharmacology 62:1401–1412. https://doi.org/10.1016/j.neuropharm.2011.03.015
Goff D (2016) The therapeutic role of d-Cycloserine in schizophrenia. In: Schwarcz R (ed) Advances in Pharmacology. Academic Press, pp 39–66
Goff DC, Tsai G, Levitt J et al (1999) A placebo-controlled trial of d-cycloserine added to conventional neuroleptics in patients with schizophrenia. Arch Gen Psychiatry 56:21–27. https://doi.org/10.1001/archpsyc.56.1.21
Goff DC, Herz L, Posever T et al (2005) A six-month, placebo-controlled trial of d-cycloserine co-administered with conventional antipsychotics in schizophrenia patients. Psychopharmacology 179:144–150. https://doi.org/10.1007/s00213-004-2032-2
Goff DC, Cather C, Gottlieb JD et al (2008) Once-weekly d-cycloserine effects on negative symptoms and cognition in schizophrenia: an exploratory study. Schizophr Res 106:320–327. https://doi.org/10.1016/j.schres.2008.08.012
Gottlieb JD, Cather C, Shanahan M et al (2011) d-cycloserine facilitation of cognitive behavioral therapy for delusions in schizophrenia. Schizophr Res 131:69–74. https://doi.org/10.1016/j.schres.2011.05.029
Heresco-Levy U, Javitt DC, Ermilov M et al (1998) Double-blind, placebo-controlled, crossover trial of d-cycloserine adjuvant therapy for treatment-resistant schizophrenia. Int J Neuropsychopharmacol 1:131–135. https://doi.org/10.1017/S1461145798001242
Heresco-Levy U, Ermilov M, Shimoni J et al (2002) Placebo-controlled trial of d-cycloserine added to conventional neuroleptics, olanzapine, or risperidone in schizophrenia. Am J Psychiatry 159:480–482. https://doi.org/10.1176/appi.ajp.159.3.480
Higgins J, Green S (2008) Cochrane Handbook for Systematic Reviews of Interventions. Wiley Online Library
Higgins JPT, Thompson SG, Deeks JJ, Altman DG (2003) Measuring inconsistency in meta-analyses. BMJ 327:557–560. https://doi.org/10.1136/bmj.327.7414.557
Higgins JPT, Altman DG, Gøtzsche PC et al (2011) The Cochrane Collaboration’s tool for assessing risk of bias in randomised trials. BMJ. https://doi.org/10.1136/bmj.d5928
Lakhan SE, Caro M, Hadzimichalis N (2013) NMDA receptor activity in neuropsychiatric disorders. Front Psychiatry. https://doi.org/10.3389/fpsyt.2013.00052
Laruelle M (2014) Schizophrenia: from dopaminergic to glutamatergic interventions. Curr Opin Pharmacol 14:97–102. https://doi.org/10.1016/j.coph.2014.01.001
Lin CH, Chen YM, Lane HY (2020) Novel treatment for the most resistant schizophrenia: dual activation of NMDA receptor and antioxidant. Curr Drug Targets 21:610–615. https://doi.org/10.2174/1389450120666191011163539
Moghaddam B, Adams B, Verma A, Daly D (1997) Activation of glutamatergic neurotransmission by ketamine: a novel step in the pathway from NMDA receptor blockade to dopaminergic and cognitive disruptions associated with the prefrontal cortex. J Neurosci 17:2921–2927. https://doi.org/10.1523/JNEUROSCI.17-08-02921.1997
Moher D, Liberati A, Tetzlaff J, Altman DG (2009) Preferred reporting items for systematic reviews and meta-analyses: the PRISMA statement. BMJ. https://doi.org/10.1136/bmj.b2535
Rolland B, Jardri R, Amad A et al (2014) Pharmacology of hallucinations: several mechanisms for one single symptom? Biomed Res Int 2014:307106. https://doi.org/10.1155/2014/307106
Rosse RB, Fay-McCarthy M, Kendrick K et al (1996) d-Cycloserine adjuvant therapy to molindone in the treatment of schizophrenia. Clin Neuropharmacol 19:444–450
Rowland LM, Astur RS, Jung RE et al (2005) Selective cognitive impairments associated with NMDA receptor blockade in humans. Neuropsychopharmacol 30:633–639. https://doi.org/10.1038/sj.npp.1300642
Takiguchi K, Uezato A, Itasaka M et al (2017) Association of schizophrenia onset age and white matter integrity with treatment effect of d-cycloserine: a randomized placebo-controlled double-blind crossover study. BMC Psychiatry 17:249. https://doi.org/10.1186/s12888-017-1410-3
Tsai GE, Lin P-Y (2010) Strategies to enhance N-methyl-d-aspartate receptor-mediated neurotransmission in schizophrenia, a critical review and meta-analysis. Curr Pharm Des 16:522–537. https://doi.org/10.2174/138161210790361452
Tuominen HJ, Tiihonen J, Wahlbeck K (2005) Glutamatergic drugs for schizophrenia: a systematic review and meta-analysis. Schizophr Res 72:225–234. https://doi.org/10.1016/j.schres.2004.05.005
Tuominen HJ, Tiihonen J, Wahlbeck K (2006) Glutamatergic drugs for schizophrenia. Cochrane Database Syst Rev. https://doi.org/10.1002/14651858.CD003730.pub2
Watanabe Y, Saito H, Abe K (1992) Effects of glycine and structurally related amino acids on generation of long-term potentiation in rat hippocampal slices. Eur J Pharmacol 223:179–184. https://doi.org/10.1016/0014-2999(92)94837-L
Watson GB, Bolanowski MA, Baganoff MP et al (1990) d-Cycloserine acts as a partial agonist at the glycine modulatory site of the NMDA receptor expressed in Xenopus oocytes. Brain Res 510:158–160. https://doi.org/10.1016/0006-8993(90)90745-W
Yurgelun-Todd DA, Coyle JT, Gruber SA et al (2005) Functional magnetic resonance imaging studies of schizophrenic patients during word production: effects of d-cycloserine. Psychiatry Res 138:23–31. https://doi.org/10.1016/j.pscychresns.2004.11.006
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Kuppili, P.P., Menon, V., Sathyanarayanan, G. et al. Efficacy of adjunctive d-Cycloserine for the treatment of schizophrenia: a systematic review and meta-analysis of randomized controlled trials. J Neural Transm 128, 253–262 (2021). https://doi.org/10.1007/s00702-020-02292-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00702-020-02292-x