Abstract
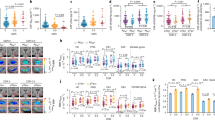
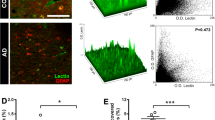
Parkinsonism–dementia complex of Guam (Guam PDC) is a neurodegenerative disease with parkinsonism and early onset Alzheimer-like dementia. Guam PDC belongs to the family of neurodegenerative disorders, known as tauopathies, which are histopathologically characterized by abnormal deposition of microtubule-associated protein tau. While changes in the blood–brain barrier (BBB) in Alzheimer’s disease are increasingly recognized, dysfunction of BBB in Guam PDC has not been extensively studied. In this study, we characterized cerebrovascular changes in the patients with Guam PDC. The brain tissue from ten post-mortem Guam PDC patients and six non-demented controls were assessed for structural and functional changes in BBB. Entorhinal cortex sections were immunostained for the markers of brain endothelial cells (claudin-5, occludin, and collagen IV) and inflammation (VCAM-1, ICAM-1, P-Selectin, and E-Selectin). The ultrastructure of brain capillaries was investigated by confocal microscopy and morphological changes and intensity alterations were evaluated. We found a significant decrease of tight junction proteins and the upregulation of adhesion molecules that correlated with the presence of neurofibrillary tangles. In addition, we showed the presence of CD3+-positive cells in the brain areas affected by pathological lesions. Our findings indicate that pathological lesions in Guam PDC are associated with inflammatory changes of brain capillaries and could mediate transmigration of cells to the brain parenchyma.






Similar content being viewed by others
Abbreviations
- AD:
-
Alzheimer’s disease
- BBB:
-
Blood–brain barrier
- NFT:
-
Neurofibrillary tangles
- Guam PDC:
-
Parkinsonism–dementia complex of Guam
References
Arpino V, Brock M, Gill SE (2015) The role of TIMPs in regulation of extracellular matrix proteolysis. Matrix Biol 44–46:247–254. https://doi.org/10.1016/j.matbio.2015.03.005
Blair LJ, Frauen HD, Zhang B, Nordhues BA, Bijan S, Lin YC, Zamudio F, Hernandez LD, Sabbagh JJ, Selenica ML, Dickey CA (2015) Tau depletion prevents progressive blood-brain barrier damage in a mouse model of tauopathy. Acta Neuropathol Commun 3:8. https://doi.org/10.1186/s40478-015-0186-2
Bradley WG, Mash DC (2009) Beyond Guam: the cyanobacteria/BMAA hypothesis of the cause of ALS and other neurodegenerative diseases. Amyotroph Lateral Scler 10(Suppl 2):7–20. https://doi.org/10.3109/17482960903286009
Buee L, Hof PR, Bouras C, Delacourte A, Perl DP, Morrison JH, Fillit HM (1994) Pathological alterations of the cerebral microvasculature in Alzheimer’s disease and related dementing disorders. Acta Neuropathol 87:469–480
Buee L, Hof PR, Delacourte A (1997) Brain microvascular changes in Alzheimer’s disease and other dementias. Ann N Y Acad Sci 826:7–24
Carrano A, Hoozemans JJ, van der Vies SM, van Horssen J, de Vries HE, Rozemuller AJ (2012) Neuroinflammation and blood–brain barrier changes in capillary amyloid angiopathy. Neurodegener Dis 10:329–331. https://doi.org/10.1159/000334916
Dickson DW (1999) Neuropathologic differentiation of progressive supranuclear palsy and corticobasal degeneration. J Neurol 246(Suppl 2):II6–II15
Dietrich JB (2002) The adhesion molecule ICAM-1 and its regulation in relation with the blood–brain barrier. J Neuroimmunol 128:58–68
DiPatre PL, Gelman BB (1997) Microglial cell activation in aging and Alzheimer disease: partial linkage with neurofibrillary tangle burden in the hippocampus. J Neuropathol Exp Neurol 56:143–149
Farkas E, De Jong GI, de Vos RA, Jansen Steur EN, Luiten PG (2000) Pathological features of cerebral cortical capillaries are doubled in Alzheimer’s disease and Parkinson’s disease. Acta Neuropathol 100:395–402
Ferrer I et al (2014) Glial and neuronal tau pathology in tauopathies: characterization of disease-specific phenotypes and tau pathology progression. J Neuropathol Exp Neurol 73:81–97. https://doi.org/10.1097/NEN.0000000000000030
Forman MS, Schmidt ML, Kasturi S, Perl DP, Lee VM, Trojanowski JQ (2002) Tau and alpha-synuclein pathology in amygdala of Parkinsonism–dementia complex patients of Guam. Am J Pathol 160:1725–1731
Forman MS, Lal D, Zhang B, Dabir DV, Swanson E, Lee VM, Trojanowski JQ (2005) Transgenic mouse model of tau pathology in astrocytes leading to nervous system degeneration. J Neurosci 25:3539–3550. https://doi.org/10.1523/JNEUROSCI.0081-05.2005
Garruto RM (1991) Pacific paradigms of environmentally-induced neurological disorders: clinical, epidemiological and molecular perspectives. Neurotoxicology 12:347–377
Garruto RM, Yanagihara R, Gajdusek DC (1985) Disappearance of high-incidence amyotrophic lateral sclerosis and parkinsonism–dementia on Guam. Neurology 35:193–198
Hirano A, Zimmerman HM (1962) Alzheimer’s neurofibrillary changes. A topographic study. Arch Neurol 7:227–242
Hirano A, Kurland LT, Krooth RS, Lessell S (1961a) Parkinsonism–dementia complex, an endemic disease on the island of Guam. I. Clinical features. Brain 84:642–661
Hirano A, Malamud N, Kurland LT (1961b) Parkinsonism–dementia complex, an endemic disease on the island of Guam. II. Pathological features. Brain 84:662–679
Jaworski T et al (2011) Dendritic degeneration, neurovascular defects, and inflammation precede neuronal loss in a mouse model for tau-mediated neurodegeneration. Am J Pathol 179:2001–2015. https://doi.org/10.1016/j.ajpath.2011.06.025
Kalaria RN, Pax AB (1995) Increased collagen content of cerebral microvessels in Alzheimer’s disease. Brain Res 705:349–352
Kovac A, Zilkova M, Deli MA, Zilka N, Novak M (2009) Human truncated tau is using a different mechanism from amyloid-beta to damage the blood–brain barrier. J Alzheimers Dis 18:897–906. https://doi.org/10.3233/JAD-2009-1197
Kovac A, Zilka N, Kazmerova Z, Cente M, Zilkova M, Novak M (2011) Misfolded truncated protein tau induces innate immune response via MAPK pathway. J Immunol 187:2732–2739. https://doi.org/10.4049/jimmunol.1100216
Lee VM, Goedert M, Trojanowski JQ (2001) Neurodegenerative tauopathies. Annu Rev Neurosci 24:1121–1159. https://doi.org/10.1146/annurev.neuro.24.1.1121
Lee DC et al (2010) LPS-induced inflammation exacerbates phospho-tau pathology in rTg4510 mice. J Neuroinflammation 7:56. https://doi.org/10.1186/1742-2094-7-56
Maccioni RB, Farias G, Morales I, Navarrete L (2010) The revitalized tau hypothesis on Alzheimer’s disease. Arch Med Res 41:226–231. https://doi.org/10.1016/j.arcmed.2010.03.007
Mena R, Edwards PC, Harrington CR, Mukaetova-Ladinska EB, Wischik CM (1996) Staging the pathological assembly of truncated tau protein into paired helical filaments in Alzheimer’s disease. Acta Neuropathol 91:633–641
Michalicova A, Banks WA, Legath J, Kovac A (2017) Tauopathies—focus on changes at the neurovascular unit. Curr Alzheimer Res. https://doi.org/10.2174/1567205014666170203143336
Mohandas E, Rajmohan V, Raghunath B (2009) Neurobiology of Alzheimer’s disease. Indian J Psychiatry 51:55–61. https://doi.org/10.4103/0019-5545.44908
Oyanagi K, Makifuchi T, Ohtoh T, Chen KM, Gajdusek DC, Chase TN (1997) Distinct pathological features of the gallyas- and tau-positive glia in the Parkinsonism–dementia complex and amyotrophic lateral sclerosis of Guam. J Neuropathol Exp Neurol 56:308–316
Schwab C, Steele JC, McGeer PL (1996) Neurofibrillary tangles of Guam parkinson-dementia are associated with reactive microglia and complement proteins. Brain Res 707:196–205
Serrano-Pozo A, Mielke ML, Gomez-Isla T, Betensky RA, Growdon JH, Frosch MP, Hyman BT (2011) Reactive glia not only associates with plaques but also parallels tangles in Alzheimer’s disease. Am J Pathol 179:1373–1384. https://doi.org/10.1016/j.ajpath.2011.05.047
Sheffield LG, Marquis JG, Berman NE (2000) Regional distribution of cortical microglia parallels that of neurofibrillary tangles in Alzheimer’s disease. Neurosci Lett 285:165–168
Soto C (2012) In vivo spreading of tau pathology. Neuron 73:621–623. https://doi.org/10.1016/j.neuron.2012.02.006
Vechterova L, Kontsekova E, Zilka N, Ferencik M, Ravid R, Novak M (2003) DC11: a novel monoclonal antibody revealing Alzheimer’s disease-specific tau epitope. NeuroReport 14:87–91. https://doi.org/10.1097/01.wnr.0000053064.88427.50
Wong D, Prameya R, Dorovini-Zis K (2007) Adhesion and migration of polymorphonuclear leukocytes across human brain microvessel endothelial cells are differentially regulated by endothelial cell adhesion molecules and modulate monolayer permeability. J Neuroimmunol 184:136–148. https://doi.org/10.1016/j.jneuroim.2006.12.003
Zilka N, Stozicka Z, Kovac A, Pilipcinec E, Bugos O, Novak M (2009) Human misfolded truncated tau protein promotes activation of microglia and leukocyte infiltration in the transgenic rat model of tauopathy. J Neuroimmunol 209:16–25. https://doi.org/10.1016/j.jneuroim.2009.01.013
Acknowledgements
We thank the Chamorro families and Guamanian patients who made this research possible. We also thank the Binghamton University Biospecimen Archive Facility (Binghamton NY, USA) for providing the brain tissue and Axon Neuroscience R&D Services SE for DC11 antibody. This work was supported by Grants APVV-14-0547 and VEGA 2/0088/18, and structural funds 26240220008 and 26240220046.
Author information
Authors and Affiliations
Contributions
PM and AK designed the study, acquired, analyzed, and interpreted the histologic data. RG contributed to the study design, tissue, and data acquisition and analysis. PM, AK, and RG drafted the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no competing interests.
Rights and permissions
About this article
Cite this article
Majerova, P., Garruto, R.M. & Kovac, A. Cerebrovascular inflammation is associated with tau pathology in Guam parkinsonism dementia. J Neural Transm 125, 1013–1025 (2018). https://doi.org/10.1007/s00702-018-1883-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00702-018-1883-3