Abstract
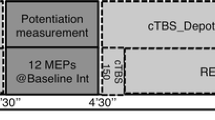
Abnormal plasticity has been reported in the brain of patients with Parkinson’s disease (PD), especially in the striatum. Although both l-Dopa and dopamine agonist remain to be the mainstay of the treatment in PD, their differential effects on cortical plasticity are unclear. We applied quadripulse stimulation (QPS) over the primary motor cortex (M1) in ten normal subjects to induce bidirectional long-term motor cortical plasticity. A long-term potentiation (LTP)-like effect was induced in the primary motor cortex (M1) by high-frequency QPS5 (interpulse interval of 5 ms) over M1, whereas a long-term depression (LTD)-like effect was induced by low-frequency QPS50 (interpulse interval of 50 ms), and the effects lasted up to 90 min after the stimulation pulses have ceased. In a double-blind randomized placebo-controlled crossover design, l-Dopa carbidopa 100 mg, pramipexole 1.5 mg [150 mg LED (l-Dopa equivalent dose)], or placebo was administered to the subjects 30 min before applying QPS. l-Dopa enhanced both LTP- and LTD-like plasticity as compared to placebo. In contrast, neither an LTP-like effect nor an LTD-like effect was modulated by pramipexole. The lack of LTP enhancement by pramipexole is compatible with the finding that D1 activation strengthens LTP because pramipexole is almost purely a D2 agonist. The lack of LTD enhancement by pramipexole is also consistent with the finding that both D1 and D2 coactivation is required for LTD. This is the first report to show that dopamine enhances LTD as well as LTP in the human brain and that coactivation of D1 and D2 is a requisite for LTD enhancement in normal humans.



Similar content being viewed by others
References
Antonini A, Calandrella D (2011) Pharmacokinetic evaluation of pramipexole. Expert Opin Drug Metab Toxicol 7(10):1307–1314. doi:10.1517/17425255.2011.614232
Bagetta V, Ghiglieri V, Sgobio C, Calabresi P, Picconi B (2010) Synaptic dysfunction in Parkinson’s disease. Biochem Soc Trans 38(2):493–497. doi:10.1042/bst0380493
Bagnato S, Agostino R, Modugno N, Quartarone A, Berardelli A (2006) Plasticity of the motor cortex in Parkinson’s disease patients on and off therapy. Mov Disord Off J Mov Disord Soc 21(5):639–645. doi:10.1002/mds.20778
Bezard E, Gross CE, Brotchie JM (2003) Presymptomatic compensation in Parkinson’s disease is not dopamine-mediated. Trends Neurosci 26(4):215–221. doi:10.1016/S0166-2236(03)00038-9
Bienenstock EL, Cooper LN, Munro PW (1982) Theory for the development of neuron selectivity: orientation specificity and binocular interaction in visual cortex. J Neurosci 2(1):32–48
Bliss TV, Collingridge GL (1993) A synaptic model of memory: long-term potentiation in the hippocampus. Nature 361(6407):31–39. doi:10.1038/361031a0
Calabresi P, Maj R, Mercuri NB, Bernardi G (1992) Coactivation of D1 and D2 dopamine receptors is required for long-term synaptic depression in the striatum. Neurosci Lett 142(1):95–99
Calabresi P, Picconi B, Tozzi A, Di Filippo M (2007) Dopamine-mediated regulation of corticostriatal synaptic plasticity. Trends Neurosci 30(5):211–219. doi:10.1016/j.tins.2007.03.001
Chen Z, Ito K, Fujii S, Miura M, Furuse H, Sasaki H, Kaneko K, Kato H, Miyakawa H (1996) Roles of dopamine receptors in long-term depression: enhancement via D1 receptors and inhibition via D2 receptors. Recept Channels 4(1):1–8
Cooke SF, Bliss TV (2006) Plasticity in the human central nervous system. Brain 129(Pt 7):1659–1673. doi:10.1093/brain/awl082
Deleu D, Northway MG, Hanssens Y (2002) Clinical pharmacokinetic and pharmacodynamic properties of drugs used in the treatment of Parkinson’s disease. Clin Pharmacokinet 41(4):261–309. doi:10.2165/00003088-200241040-00003
Eggers C, Fink GR, Nowak DA (2010) Theta burst stimulation over the primary motor cortex does not induce cortical plasticity in Parkinson’s disease. J Neurol 257(10):1669–1674. doi:10.1007/s00415-010-5597-1
Enomoto H, Ugawa Y (2012) Abnormal plasticity and drug effect in Parkinson’s disease. Rinsho shinkeigaku (Clin Neurol) 52(11):1204–1206
Floel A, Hummel F, Breitenstein C, Knecht S, Cohen LG (2005) Dopaminergic effects on encoding of a motor memory in chronic stroke. Neurology 65(3):472–474. doi:10.1212/01.wnl.0000172340.56307.5e
Fresnoza S, Paulus W, Nitsche MA, Kuo MF (2014) Nonlinear dose-dependent impact of D1 receptor activation on motor cortex plasticity in humans. J Neurosci 34(7):2744–2753. doi:10.1523/JNEUROSCI.3655-13.2014
Gentner R, Wankerl K, Reinsberger C, Zeller D, Classen J (2008) Depression of human corticospinal excitability induced by magnetic theta-burst stimulation: evidence of rapid polarity-reversing metaplasticity. Cereb Cortex 18(9):2046–2053. doi:10.1093/cercor/bhm239
Goetz CG, Blasucci L, Stebbins GT (1999) Switching dopamine agonists in advanced Parkinson’s disease: is rapid titration preferable to slow? Neurology 52(6):1227–1229
Goto Y, Yang CR, Otani S (2010) Functional and dysfunctional synaptic plasticity in prefrontal cortex: roles in psychiatric disorders. Biol Psychiatry 67(3):199–207. doi:10.1016/j.biopsych.2009.08.026
Hallett M (2007) Transcranial magnetic stimulation: a primer. Neuron 55(2):187–199. doi:10.1016/j.neuron.2007.06.026
Hamada M, Terao Y, Hanajima R, Shirota Y, Nakatani-Enomoto S, Furubayashi T, Matsumoto H, Ugawa Y (2008) Bidirectional long-term motor cortical plasticity and metaplasticity induced by quadripulse transcranial magnetic stimulation. J Physiol 586(16):3927–3947. doi:10.1113/jphysiol.2008.152793
Hamada M, Hanajima R, Terao Y, Okabe S, Nakatani-Enomoto S, Furubayashi T, Matsumoto H, Shirota Y, Ohminami S, Ugawa Y (2009) Primary motor cortical metaplasticity induced by priming over the supplementary motor area. J Physiol 587(Pt 20):4845–4862. doi:10.1113/jphysiol.2009.179101
Hamada M, Murase N, Hasan A, Balaratnam M, Rothwell JC (2013) The role of interneuron networks in driving human motor cortical plasticity. Cereb Cortex 23(7):1593–1605. doi:10.1093/cercor/bhs147
Hebb DO (1949) The organization of behavior. Wiley, New York
Huang YZ, Chen RS, Rothwell JC, Wen HY (2007) The after-effect of human theta burst stimulation is NMDA receptor dependent. Clin Neurophysiol 118(5):1028–1032. doi:10.1016/j.clinph.2007.01.021
Huang YZ, Rothwell JC, Edwards MJ, Chen RS (2008) Effect of physiological activity on an NMDA-dependent form of cortical plasticity in human. Cereb Cortex 18(3):563–570. doi:10.1093/cercor/bhm087
Huang YZ, Rothwell JC, Lu CS, Chuang WL, Lin WY, Chen RS (2010) Reversal of plasticity-like effects in the human motor cortex. J Physiol 588(Pt 19):3683–3693. doi:10.1113/jphysiol.2010.191361
Huang YZ, Rothwell JC, Lu CS, Chuang WL, Chen RS (2011) Abnormal bidirectional plasticity-like effects in Parkinson’s disease. Brain 134(Pt 8):2312–2320. doi:10.1093/brain/awr158
Kawashima S, Ueki Y, Mima T, Fukuyama H, Ojika K, Matsukawa N (2013) Differences in dopaminergic modulation to motor cortical plasticity between Parkinson's disease and multiple system atrophy. PloS one 8(5):e62515. doi:10.1371/journal.pone.0062515
Koch G (2013) Do studies on cortical plasticity provide a rationale for using non-invasive brain stimulation as a treatment for Parkinson’s disease patients? Front Neurol 4:180. doi:10.3389/fneur.2013.00180
Kuo MF, Paulus W, Nitsche MA (2008) Boosting focally-induced brain plasticity by dopamine. Cereb Cortex 18(3):648–651. doi:10.1093/cercor/bhm098
Monte-Silva K, Kuo MF, Thirugnanasambandam N, Liebetanz D, Paulus W, Nitsche MA (2009) Dose-dependent inverted U-shaped effect of dopamine (D2-like) receptor activation on focal and nonfocal plasticity in humans. J Neurosci 29(19):6124–6131. doi:10.1523/jneurosci.0728-09.2009
Monte-Silva K, Ruge D, Teo JT, Paulus W, Rothwell JC, Nitsche MA (2011) D2 receptor block abolishes theta burst stimulation-induced neuroplasticity in the human motor cortex. Neuropsychopharmacol Off Publ Am Coll Neuropsychopharmacol 36(10):2097–2102. doi:10.1038/npp.2011.100
Morgante F, Espay AJ, Gunraj C, Lang AE, Chen R (2006) Motor cortex plasticity in Parkinson’s disease and levodopa-induced dyskinesias. Brain 129(Pt 4):1059–1069. doi:10.1093/brain/awl031
Müller T (2013) Pharmacokinetic considerations for the use of levodopa in the treatment of Parkinson disease: focus on levodopa/carbidopa/entacapone for treatment of levodopa-associated motor complications. Clin Neuropharmacol 36(3):84–91. doi:10.1097/WNF.0b013e31828f3385
Nitsche MA, Kuo MF, Grosch J, Bergner C, Monte-Silva K, Paulus W (2009) D1-receptor impact on neuroplasticity in humans. J Neurosci 29(8):2648–2653. doi:10.1523/jneurosci.5366-08.2009
Oldfield RC (1971) The assessment and analysis of handedness: the Edinburgh inventory. Neuropsychologia 9(1):97–113
Picconi B, Centonze D, Hakansson K, Bernardi G, Greengard P, Fisone G, Cenci MA, Calabresi P (2003) Loss of bidirectional striatal synaptic plasticity in l-DOPA-induced dyskinesia. Nat Neurosci 6(5):501–506. doi:10.1038/nn1040
Scheidtmann K, Fries W, Muller F, Koenig E (2001) Effect of levodopa in combination with physiotherapy on functional motor recovery after stroke: a prospective, randomised, double-blind study. Lancet 358(9284):787–790. doi:10.1016/S0140-6736(01)05966-9
Schwingenschuh P, Katschnig P, Saurugg R, Ott E, Bhatia KP (2010) Artistic profession: a potential risk factor for dopamine dysregulation syndrome in Parkinson’s disease? Mov Disord Off J Mov Disord Soc 25(4):493–496. doi:10.1002/mds.22936
Sonde L, Lokk J (2007) Effects of amphetamine and/or l-dopa and physiotherapy after stroke—a blinded randomized study. Acta Neurol Scand 115(1):55–59. doi:10.1111/j.1600-0404.2006.00728.x
Stefan K, Kunesch E, Benecke R, Cohen LG, Classen J (2002) Mechanisms of enhancement of human motor cortex excitability induced by interventional paired associative stimulation. J Physiol 543(2):699–708. doi:10.1113/jphysiol.2002.023317
Suarez LM, Bustamante J, Orensanz LM, Martin del Rio R, Solis JM (2014) Cooperation of taurine uptake and dopamine D1 receptor activation facilitates the induction of protein synthesis-dependent late LTP. Neuropharmacology 79:101–111. doi:10.1016/j.neuropharm.2013.10.035
Suppa A, Marsili L, Belvisi D, Conte A, Iezzi E, Modugno N, Fabbrini G, Berardelli A (2011) Lack of LTP-like plasticity in primary motor cortex in Parkinson’s disease. Exp Neurol 227(2):296–301. doi:10.1016/j.expneurol.2010.11.020
Thirugnanasambandam N, Grundey J, Paulus W, Nitsche MA (2011) Dose-dependent nonlinear effect of l-DOPA on paired associative stimulation-induced neuroplasticity in humans. J Neurosci 31(14):5294–5299. doi:10.1523/jneurosci.6258-10.2011
Tomlinson CL, Stowe R, Patel S, Rick C, Gray R, Clarke CE (2010) Systematic review of levodopa dose equivalency reporting in Parkinson’s disease. Mov Disord Off J Mov Disord Soc 25(15):2649–2653. doi:10.1002/mds.23429
Udupa K, Chen R (2013) Motor cortical plasticity in Parkinson’s disease. Front Neurol 4:128. doi:10.3389/fneur.2013.00128
Ueki Y, Mima T, Kotb MA, Sawada H, Saiki H, Ikeda A, Begum T, Reza F, Nagamine T, Fukuyama H (2006) Altered plasticity of the human motor cortex in Parkinson’s disease. Ann Neurol 59(1):60–71. doi:10.1002/ana.20692
Ueno T, Yamada J, Nishijima H, Arai A, Migita K, Baba M, Ueno S, Tomiyama M (2014) Morphological and electrophysiological changes in intratelencephalic-type pyramidal neurons in the motor cortex of a rat model of levodopa-induced dyskinesia. Neurobiol Dis 64:142–149. doi:10.1016/j.nbd.2013.12.014
Acknowledgments
This work was supported in part by grants from the Research Project Grant-in-aid for Scientific Research from the Ministry of Education, Culture, Sports, Science and Technology No. 25461322 (HE), No. 22590954 (YT), No. 23591270 (RH), No. 22390181(YU), No. 25293206 (YU), a research grant from the Catecholamine and the Neurological Disorders Symposium (RH), and research funds from the Magnetic Health Science Foundation (RH, YT) and the Kato Memorial Trust for Nambyo Research, Japan (YT), and the Research Committee on Degenerative Ataxia from the Ministry of Health and Welfare of Japan; the Magnetic Health Science Foundation; the Uehara Memorial Foundation and The Novartis Foundation (Japan) for the Promotion of Science (YU).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Enomoto, H., Terao, Y., Kadowaki, S. et al. Effects of l-Dopa and pramipexole on plasticity induced by QPS in human motor cortex. J Neural Transm 122, 1253–1261 (2015). https://doi.org/10.1007/s00702-015-1374-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00702-015-1374-8