Abstract
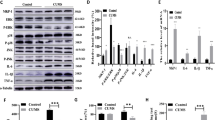
The c-Jun NH2-terminal kinases (JNKs) are important stress-responsive kinases. They regulate cellular activities by sequential phosphorylation and activation through a mitogen-activated protein kinase cascade, whereas JNKs activation is altered in response to various stressors. In the present study, we used immunoblotting to assess the effect of 21 day of social isolation as the chronic stressor, either sole and in combination with 2 h of acute immobilization or cold (4°C) stress on circulating corticosterone level and phosphorylation status of p46 (phospho-p46/total p46) and p54 (phospho-p54/total p54) JNK isoforms in the cytosolic and nuclear fraction of the prefrontal cortex and hippocampus of male Wistar rats. Also, the phosphorylation status of JNK nuclear down-stream target c-Jun (p–c-Jun/c-Jun) on Ser63 was examined. Both acute stressors with elevated CORT levels led to increased phosphorylation status of cytosolic p54 JNK isoforms but not p46 JNK isoforms only in the hippocampus and no change in phosphorylation status of c-jun in both brain regions. Chronic isolation with unaltered CORT level and reduced responsiveness to novel acute stressors, led to unchanged or reduced phosphorylation status of p46 and p54 JNK isoforms in both fractions and both brain regions, whereas the decrease of c-Jun phosphorylation status was found only in the prefrontal cortex. Our results suggest that compromised JNKs activation following chronic isolation may lead to interruption of JNK signaling, which could be related with neuropsychiatric disorders such as depression or long-lasting neuronal remodeling.





Similar content being viewed by others
References
Behrens A, Sibilia M, Wagner EF (1999) Amino-terminal phosphorylation of c-Jun regulates stress-induced apoptosis and cellular proliferation. Nat Genet 21:326–329. doi:10.1038/6854
Bird TA, Kyriakis JM, Tyshler L, Gayle M, Milne A, Virca GD (1994) Interleukin-1 activates p54 mitogen-activated protein (MAP) kinase/stress-activated protein kinase by a pathway that is independent of p21ras, Raf-1, and MAP kinase kinase. J Biol Chem 269:31836–31844
Broderick PA (2002) Interleukin 1α alters hippocampal serotonin and norepinephrine release during open-field behavior in Sprague–Dawley animals: differences from the Fawn-Hooded animal model of depression. Prog Neuropsychopharmacol Biol Psychiatry 26:1355–1372
Brooks KP, Robles TF (2009) Recent depressive and anxious symptoms predict cortisol responses to stress in men. Psychoneuroendocrinology 34:1041–1049. doi:10.1016/j.psyneuen.2009.02.005
Bruna A, Nicolàs M, Muñoz A, Kyriakis JM, Caelles C (2003) Glucocorticoid receptor-JNK interaction mediates inhibition of the JNK pathway by glucocorticoids. EMBO J 22:6035–6044. doi:10.1093/emboj/cdg590
Budziszewska B, Szymanska M, Leskiewicz M, Basta-Kaim A, Jaworska-Feil L, Kubera M, Jantas D, Lason W (2010) The decrease in JNK- and p38-MAP kinase activity is accompanied by the enhancement of PP2A phosphate level in the brain of prenatally stressed rats. J Physiol Pharmacol 61:207–215
Caelles C, González-Sancho JM, Muñoz A (1997) Nuclear hormone receptor antagonism with AP-1 by inhibition of the JNK pathway. Genes Dev 11:3351–3364. doi:10.1101/gad.11.24.3351
Cavigelli M, Dol F, Claret FX, Karin M (1995) Induction of c-fos expression through JNK-mediated TCF-ELK1 phosphorylation. EMBO J 14:5957–5964
Chrousos GP, Kino T (2007) Glucocorticoid action networks and complex psychiatric and/or somatic disorders. Stress 10:213–219. doi:10.1080/10253890701292119
Clark AR, Lasa M (2003) Crosstalk between glucocorticoids and mitogen-activated protein kinase signalling pathways. Curr Opin Pharmacol 3:404–411. doi:10.1016/S1471-4892(03)00073-0
Cruzalegui FH, Hardingham GE, Bading H (1999) c-Jun functions as a calcium-regulated transcriptional activator in the absence of JNK/SAPK1 activation. EMBO J 18:1335–1344. doi:10.1093/emboj/18.5.1335
Dronjak S, Gavrilović L, Filipović D, Radojcić MB (2004) Immobilization and cold stress affect sympatho-adrenomedullary system and pituitary-adrenocortical axis of rats exposed to long-term isolation and crowding. Physiol Behav 81:409–415. doi:10.1016/j.physbeh.2004.01.011
Filipović D, Pajović SB (2009) Differential regulation of CuZnSOD expression in rat brain by acute and/or chronic stress. Cell Mol Neurobiol 29:673–681. doi:10.1007/s10571-009-9375-5
Filipović D, Gavrilović LJ, Dronjak S, Radojčić M (2005) Brain glucocorticoid receptor and heat shock protein 70 levels in rats exposed to chronic, acute or combined stress. Neuropsychobiology 51:107–114. doi:10.1159/000084168
Filipović D, Zlatković J, Pajović SB (2009) The effect of acute or/and chronic stress on the MnSOD protein expression in rat prefrontal cortex and hippocampus. Gen Physiol Biophys 28 Spec No 53-61
Filipović D, Zlatković J, Inta D, Bjelobaba I, Stojiljkovic M, Gass P (2011) Chronic isolation stress predisposes the frontal cortex but not the hippocampus to the potentially detrimental release of cytochrome c from mitochondria and the activation of caspase-3. J Neurosci Res 89:1461–1470. doi:10.1002/jnr.22687
Fuchs SY, Adler V, Pincus MR, Ronai Z (1998) MEKK1/JNK signaling stabilizes and activates p53. Proc Natl Acad Sci USA 95:10541–10546. doi:10.1073/pnas.95.18.10541
Garcia A, Marti O, Valles A, Dal-Zotto S, Armario A (2000) Recovery of the hypothalamic–pituitary–adrenal response to stress. Effect of stress intensity, stress duration and previous stress exposure. Neuroendocrinology 72:114–125. doi:10.1159/000054578
Garzón J, Del Río J (1981) Hyperactivity induced in rats by long-term isolation: further studies on a new animal model for the detection of antidepressants. Eur J Pharmacol 74:287–294. doi:10.1016/0014-2999(81)90047-9
Gavrilović L, Dronjak S (2005) Activation of rat pituitary-adrenocortical and sympatho-adrenomedullary system in response to different stressors. Neuro Endocrinol Lett 26:515–520
González MV, Jiménez B, Berciano MT, González-Sancho JM, Caelles C, Lafarga M, Muñoz A (2000) Glucocorticoids antagonize AP-1 by inhibiting the activation/phosphorylation of JNK without affecting its subcellular distribution. J Cell Biol 150:1199–1207. doi:10.1083/jcb.150.5.1199
Gupta S, Campbell D, Derijard B, Davis RJ (1995) Transcription factor ATF-2 regulation by the JNK signal transduction pathway. Science 267:389–393. doi:10.1126/science.7824938
Gupta S, Barrett T, Whitmarsh AJ, Cavanagh J, Sluss HK, Dérijard B, Davis RJ (1996) Selective interaction of JNK protein kinase isoforms with transcription factors. EMBO J 15:2760–2770
Ham J, Eilers A, Whitfield J, Neame SJ, Shah B (2000) c-Jun and the transcriptional control of neuronal apoptosis. Biochem Pharmacol 60:1015–1021. doi:10.1016/S0006-2952(00)00372-5
Herdegen T, Skene P, Bahr M (1997) The c-Jun transcriptional factor: bipotential mediator of neuronal death, survival and regeneration. Trends Neurosci 20:227–231. doi:10.1016/S0166-2236(96)01000-4
Holsboer F (2000) The corticosteroid receptor hypothesis of depression. Neuropsychopharmacology 23:477–501. doi:10.1038/sj.npp.1395567
Jarrard LE (1993) On the role of the hippocampus in learning and memory in the rat. Behav Neural Biol 60:9–26. doi:10.1016/0163-1047(93)90664-4
Kamata H, Honda S, Maeda S, Chang L, Hirata H, Karin M (2005) Reactive oxygen species promote TNFalpha-induced death and sustained JNK activation by inhibiting MAP kinase phosphatases. Cell 120:649–661. doi:10.1016/j.cell.2004.12.041
Karin M, Liu Z, Zandi E (1997) AP-1 function and regulation. Curr Opin Cell Biol 9:240–246. doi:10.1016/S0955-0674(97)80068-3
Krstic MD, Rogatsky I, Yamamoto KR, Garabedian MJ (1997) Mitogen-activated and cyclin-dependent protein kinases selectively and differentially modulate transcriptional enhancement by the glucocorticoid receptor. Mol Cell Biol 17:3947–3954
Kvetnansky R, Mikulaj L (1970) Adrenal and urinary catecholamines in rats during adaptation to repeated immobilization stress. Endocrinology 87:738–743. doi:10.1210/endo-87-4-738
Leppä S, Bohmann D (1999) Diverse functions of JNK signaling and c-Jun in stress response and apoptosis. Oncogene 18:6158–6162. doi:10.1038/sj.onc.1203173
Li L, Feng Z, Porter AG (2004) JNK-dependent phosphorylation of c-Jun on serine 63 mediates nitric oxide-induced apoptosis of neuroblastoma cells. J Biol Chem 279:4058–4065. doi:10.1074/jbc.M310415200
Liu YF, Bertram K, Perides G, McEwen BS, Wang D (2004a) Stress induces activation of stress-activated kinases in the mouse brain. J Neurochem 89:1034–1043. doi:10.1111/j.1471-4159.2004.02391.x
Liu J, Minemoto Y, Lin A (2004b) c-Jun N-Terminal protein kinase 1 (JNK1), but not JNK2, is essential for tumor necrosis factor alpha-induced c-Jun kinase activation and apoptosis. Mol Cell Biol 24:10844–10856. doi:10.1128/MCB.24(24),10844-10856.2004
Lopez-Ilasaca M (1998) Signaling from G-protein-coupled receptors to mitogen-activated protein (MAP)-kinase cascades. Biochem Pharmacol 56:269–277. doi:10.1016/S0006-2952(98)00059-8
Lowry OH, Rosebrough NJ, Farr AJ, Randall RJ (1951) Protein measurement with the Folin phenol reagent. J Biol Chem 123:265–275. doi:10.1111/j.1471-4159.2009.05892.x
Marinissen MJ, Gutkind JS (2001) G-protein-coupled receptors and signaling networks: emerging paradigms. Trends Pharmacol Sci 22:368–376. doi:10.1016/S0165-6147(00)01678-3
Maroney AC, Glicksman MA, Basma AN, Walton KM, Knight E Jr, Murphy CA, Bartlett BA, Finn JP, Angeles T, Matsuda Y, Neff NT, Dionne CA (1998) Motoneuron apoptosis is blocked by CEP-1347 (KT 7515), a novel inhibitor of the JNK signaling pathway. J Neurosci 18:104–111
Maundrell K, Antonsson B, Magnenat E, Camps M, Muda M, Chabert C, Gillieron C, Boschert U, Vial-Knecht E, Martinou JC, Arkinstall S (1997) Bcl-2 undergoes phosphorylation by c-Jun N-terminal kinase/stress-activated protein kinase in the presence of the constitutively active GTP-binding protein. Rac1. J Biol Chem 272:25238–25242. doi:10.1074/jbc.272.40.25238
McEwen BS (1998) Protective and damaging effects of stress mediators. N Engl J Med 338:171–179. doi:10.1056/NEJM199801153380307
McEwen BS (2006) Protective and damaging effects of stress mediators: central role of the brain. Dialog Clin Neurosci 8:367–381
Meller E (2007) 5-HT1A receptor-mediated apoptosis: death by JNK? Biochim Biophys Acta 1773(6):691–693. doi:10.1016/j.bbamcr.2007.01.003
Meller E, Shen C, Nikolao T, Jensen C, Tsimberg Y, Chen J, Gruen RJ (2003) Region-specific effects of acute and repeated restraint stress on the phosphorylation of mitogen-activated protein kinases. Brain Res 979:57–64. doi:10.1016/S0006-8993(03)02866-X
Mielke K, Herdegen T (2000) JNK and p38 stress kinases degenerative effectors of signal-transduction-cascades in the nervous system. Prog Neurobiol 61:45–60. doi:10.1016/S0301-0082(99)00042-8
Minden A, Karin M (1997) Regulation and function of the JNK subgroup of MAP kinases. Biochim Biophys Acta 1333:85–104. doi:10.1016/S0304-419X(97)00018-8
Morton S, Davis RJ, McLaren A, Cohen P (2003) A reinvestigation of the multisite phosphorylation of the transcription factor c-Jun. EMBO J 22:3876–3886. doi:10.1093/emboj/cdg388
Nankova BB, Fuchs SY, Serova LI, Ronai Z, Wild D, Sabban EL (1998) Selective in vivo stimulation of stress-activated protein kinase in different rat tissues by immobilization stress. Stress 2:289–298
Nemoto S, Takeda K, Yu ZX, Ferrans VJ, Finkel T (2000) Role for mitochondrial oxidants as regulators of cellular metabolism. Mol Cell Biol 20:7311–7318. doi:10.1128/MCB.20.19.7311-7318.2000
Pacak K, Palkovits M, Yadid G, Kvetnansky R, Kopin IJ, Goldstein DS (1998) Heterogeneous neurochemical responses to different stressors: a test of Selye’s doctrine of nonspecificity. Am J Physiol Regul Integr Comp Physiol 275:R1247–R1255
Pulverer BJ, Kyriakis JM, Avruch J, Nikolakaki E, Woodgett JR (1991) Phosphorylation of c-Jun mediated by MAP kinases. Nature 353:670–674. doi:10.1038/353670a0
Rogatsky I, Logan SK, Garabedian MI (1998) Antagonism of glucocorticoid receptor transcriptional activation by the c-Jun N-terminal kinase. Proc Natl Acad Sci USA 95:2050–2055. doi:10.1073/pnas.95.5.2050
Saporito MS, Thomas BA, Scott RW (2000) MPTP activates c-Jun NH(2)-terminal kinase (JNK) and its upstream regulatory kinase MKK4 in nigrostriatal neurons in vivo. J Neurochem 75:1200–1208. doi:10.1046/j.1471-4159.2000.0751200.x
Shen HM, Liu Z (2006) JNK signaling pathway is a key modulator in cell death mediated by reactive and nitrogen species. Free Radic Biol Med 40:928–939. doi:10.1016/j.freeradbiomed.2005.10.056
Shen CP, Tsimberg Y, Salvadore C, Meller E (2004) Activation of Erk and JNK MAPK pathways by acute swim stress in rat brain regions. BMC Neurosci 5:36. doi:10.1186/1471-2202-5-36
Sherrin T, Blank T, Hippel C, Rayner M, Davis RJ, Todorovic C (2010) Hippocampal c-Jun-N-terminal kinases serve as negative regulators of associative learning. J Neurosci 30:13348–13361. doi:10.1523/JNEUROSCI.3492-10.2010
Simmons DA, Broderick PA (2005) Cytokines, stressors, and clinical depression: augmented adaptation responses underlie depression pathogenesis. Prog Neuropsychopharmacol Biol Psychiatry 29:793–807. doi:10.1016/j.pnpbp.2005.03.009
Smeal T, Binetruy B, Mercola DA, Birrer M, Karin M (1991) Oncogenic and transcriptional cooperation with Ha-Ras requires phosphorylation of c-Jun on serines 63 and 73. Nature 354:494–496. doi:10.1038/354494a0
Spencer RL, Kalman BA, Cotter CS, Deak T (2000) Discrimination between changes in glucocorticoid receptor expression and activation in rat brain using western blot analysis. Brain Res 868:275–286. doi:10.1016/S0006-8993(00)02341-6
Spiliotaki M, Salpeas V, Malitas P, Alevizos V, Moutsatsou P (2006) Altered glucocorticoid receptor signaling cascade in lymphocytes of bipolar disorder patients. Psychoneuroendocrinology 31:748–760. doi:10.1016/j.psyneuen.2006.02.006
Torres M (2002) Mitogen-activated protein kinase pathways in redox signaling. Front Biosci 8:369–391. doi:10.2741/999
Tournier C, Dong C, Turner TK, Jones SN, Flavell RA, Davis RJ (2001) MKK7 is an essential component of the JNK signal transduction pathway activated by proinflammatory cytokines. Genes Dev 15:1419–1426. doi:10.1101/gad.888501
Turner JH, Garnovskaya MN, Raymond JR (2007) Serotonin 5-HT1A receptor stimulates c-Jun N-terminal kinase and induces apoptosis in Chinese hamster ovary fibroblasts. Biochim Biophys Acta 1773:391–399. doi:10.1016/j.bbamcr.2006.12.003
van Dam H, Wilhelm D, Herr I, Steen A, Herrlich P, Angel P (1995) ATF-2 is preferentially activated by stress-activated protein kinase to mediated c-jun induction in response to genotoxic agents. EMBO J 14:1798–1811
Whitmarsh AJ, Davis RJ (1996) Transcription factor AP-1 regulation by mitogen-activated protein kinase signal transduction pathways. J Mol Med 74:589–607. doi:10.1007/s001090050063
Xia ZG, Dickens M, Raingeaud J, Davis RJ, Greenberg ME (1995) Opposite effect of ERK and JNK-p38 MAP kinases on apoptosis. Science 270:1326–1331. doi:10.1126/science.270.5240.1326
Zlatković J, Filipović D (2012) Chronic isolation stress increases nNOS and iNOS protein expression in the prefrontal cortex of rats. http://ibro2012.shp.hu/hpc/web.php?a=ibro2012&o=detailed_program_5U9d, p 240
Zlatković J, Filipović D (2012) Increase of nNOS protein expression has no influence on redox balance in the hippocampus of chronically isolated rats. http://ibro2012.shp.hu/hpc/web.php?a=ibro2012&o=detailed_program_5U9d, p 241
Acknowledgments
This work was supported by the Ministry of Education and Science of the Republic of Serbia, Grant 173044.
Conflict of interest
The authors declare that they have no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Filipović, D., Zlatković, J., Pavićević, I. et al. Chronic isolation stress compromises JNK/c-Jun signaling in rat brain. J Neural Transm 119, 1275–1284 (2012). https://doi.org/10.1007/s00702-012-0776-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00702-012-0776-0