Abstract
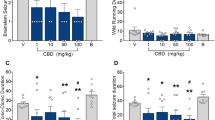
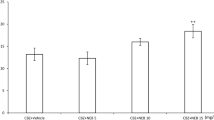
Several studies have shown that cannabinoids have anticonvulsant properties that are mediated through activation of the cannabinoid CB1 receptors. In addition, endogenous cannabinoid compounds (endocannabinoids) regulate synaptic transmission and dampen seizure activity via activation of the same receptors. The aim of this study was to evaluate the possible interactions between antiepileptic effects of cannabinoid compounds and diazepam using electroshock-induced model of seizure in mice. Electroconvulsions were produced by means of an alternating current (ear-clip electrodes, fixed current intensity 35 mA, stimulus duration 0.2 s) and tonic hindlimb extension was taken as the endpoint. All experiments were performed on groups of ten mice and the number of animals who did not display seizure reported as percent protection. Intraperitoneal (i.p.) administration of diazepam (0.25–2 mg/kg) and CB1 receptor agonist WIN55212-2 (0.5–4 mg/kg) dose dependently produced an antiepileptic effect evaluated in terms of increased percentage of protection against electroshock-induced seizure. Logistic regression analysis indicated synergistic interactions in anticonvulsant action after co-administration of diazepam and WIN55212-2 in fixed-ratio combination of 3:1 (diazepam:WIN55212-2), while an additive effect was resulted after co-administration of 1:1 and 1:3 fixed-ratio combinations. Administration of various doses of the endocannabinoid reuptake inhibitor, AM404, did not produce any effect on electroshock-induced seizure. Moreover, co-administration of AM404 and diazepam did not produce significant interaction in antiepileptic properties of these compounds. Administration of the fatty acid amide hydrolase inhibitor, URB597, produced significant antiepileptic effect. Co-administration of URB597 and diazepam led to an antagonistic interaction in protection against shock-induced seizure. Co-administration of different doses of the cannabinoid CB1 receptor antagonist, AM251 did not alter the antiepileptic effect of diazepam in the electroshock-induced seizure test. These results demonstrate that endocannabinoid system participates in the modulation of seizure and combination of small doses of exogenous CB1 receptor agonists with diazepam may have effective consequences in seizure control. Furthermore, inhibiting the endocannabinoid degradation could be more efficacious in modulating seizure than preventing their uptake. This study also suggests that the effects of cannabinoids on epilepsy depend on the relative cannabinoid responsiveness of GABAergic and glutamatergic neurotransmission. While, the antiepileptic effects of cannabinoid compounds are likely by affecting excitatory glutamate neurotransmission, the antagonistic interaction between cannabinoid compounds and diazepam to protect seizure is due to the cannabinoid action on inhibitory GABAergic system.
Similar content being viewed by others
References
Abood ME, Rizvi G, Sallapudi N, McAllister SD (2001) Activation of the CB1 cannabinoid receptor protects cultured mouse spinal neurons against excitotoxicity. Neurosci Lett 309:197–201
Adams IB, Martin BR (1996) Cannabis: pharmacology and toxicology in animals and humans. Addiction 91:1585–1614
Alger BE (2002) Retrograde signaling in the regulation of synaptic transmission: focus on endocannabinoids. Prog Neurobiol 68:247–286
Alger BE (2004) Endocannabinoids and their implications for epilepsy. Epilepsy Curr 4:169–173
Azad SC, Eder M, Marsicano G, Lutz B, Zieglgansberger W, Rammes G (2003) Activation of the cannabinoid receptor type 1 decreases glutamatergic and GABAergic synaptic transmission in the lateral amygdala of the mouse. Learn Mem 10:116–128
Baker D, Pryce G, Croxford JL, Brown P, Pertwee RG, Makriyannis A, Khanolkar A, Layward L, Fezza F, Bisogno T, Di Marzo V (2001) Endocannabinoids control spasticity in a multiple sclerosis model. Faseb J 15:300–302
Beltramo M, Stella N, Calignano A, Lin SY, Makriyannis A, Piomelli D (1997) Functional role of high-affinity anandamide transport, as revealed by selective inhibition. Science 277:1094–1097
Ben Amar M (2006) Cannabinoids in medicine: a review of their therapeutic potential. J Ethnopharmacol 105:1–25
Blair RE, Deshpande LS, Sombati S, Falenski KW, Martin BR, DeLorenzo RJ (2006) Activation of the cannabinoid type-1 receptor mediates the anticonvulsant properties of cannabinoids in the hippocampal neuronal culture models of acquired epilepsy and status epilepticus. J Pharmacol Exp Ther 317:1072–1078
Browne TR, Holmes GL (2001) Epilepsy. N Engl J Med 344:1145–1151
Chen K, Baram TZ, Soltesz I (1999) Febrile seizures in the developing brain result in persistent modification of neuronal excitability in limbic circuits. Nat Med 5:888–894
Chen K, Ratzliff A, Hilgenberg L, Gulyas A, Freund TF, Smith M, Dinh TP, Piomelli D, Mackie K, Soltesz I (2003) Long-term plasticity of endocannabinoid signaling induced by developmental febrile seizures. Neuron 39:599–611
Clement AB, Hawkins EG, Lichtman AH, Cravatt BF (2003) Increased seizure susceptibility and proconvulsant activity of anandamide in mice lacking fatty acid amide hydrolase. J Neurosci 23:3916–3923
Consroe P, Martin A, Singh V (1981) Antiepileptic potential of cannabidiol analogs. J Clin Pharmacol 21:428S–436S
Consroe P, Benedito MA, Leite JR, Carlini EA, Mechoulam R (1982) Effects of cannabidiol on behavioral seizures caused by convulsant drugs or current in mice. Eur J Pharmacol 83:293–298
Cunha JM, Carlini EA, Pereira AE, Ramos OL, Pimentel C, Gagliardi R, Sanvito WL, Lander N, Mechoulam R (1980) Chronic administration of cannabidiol to healthy volunteers and epileptic patients. Pharmacology 21:175–185
Czuczwar SJ, Borowicz KK (2002) Polytherapy in epilepsy: the experimental evidence. Epilepsy Res 52:15–23
Deckers CL, Czuczwar SJ, Hekster YA, Keyser A, Kubova H, Meinardi H, Patsalos PN, Renier WO, Van Rijn CM (2000) Selection of antiepileptic drug polytherapy based on mechanisms of action: the evidence reviewed. Epilepsia 41:1364–1374
Deshpande LS, Sombati S, Blair RE, Carter DS, Martin BR, DeLorenzo RJ (2007) Cannabinoid CB1 receptor antagonists cause status epilepticus-like activity in the hippocampal neuronal culture model of acquired epilepsy. Neurosci Lett 411:11–16
Devane WA, Hanus L, Breuer A, Pertwee RG, Stevenson LA, Griffin G, Gibson D, Mandelbaum A, Etinger A, Mechoulam R (1992) Isolation and structure of a brain constituent that binds to the cannabinoid receptor. Science 258:1946–1949
Di Marzo V, Bifulco M, De Petrocellis L (2004) The endocannabinoid system and its therapeutic exploitation. Nat Rev Drug Discov 3:771–784
Fegley D, Gaetani S, Duranti A, Tontini A, Mor M, Tarzia G, Piomelli D (2005) Characterization of the fatty acid amide hydrolase inhibitor cyclohexyl carbamic acid 3′-carbamoyl-biphenyl-3-yl ester (URB597): effects on anandamide and oleoylethanolamide deactivation. J Pharmacol Exp Ther 313:352–358
Freund TF, Katona I, Piomelli D (2003) Role of endogenous cannabinoids in synaptic signaling. Physiol Rev 83:1017–1066
Gennings C, Carter WH Jr, Campbell ED, Staniswalis JG, Martin TJ, Martin BR, White KL Jr (1990) Isobolographic characterization of drug interactions incorporating biological variability. J Pharmacol Exp Ther 252:208–217
Gerdeman GL, Lovinger DM (2003) Emerging roles for endocannabinoids in long-term synaptic plasticity. Br J Pharmacol 140:781–789
Gerdeman GL, Ronesi J, Lovinger DM (2002) Postsynaptic endocannabinoid release is critical to long-term depression in the striatum. Nat Neurosci 5:446–451
Gonzalez S, Romero J, de Miguel R, Lastres-Becker I, Villanua MA, Makriyannis A, Ramos JA, Fernandez-Ruiz JJ (1999) Extrapyramidal and neuroendocrine effects of AM404, an inhibitor of the carrier-mediated transport of anandamide. Life Sci 65:327–336
Gordon E, Devinsky O (2001) Alcohol and marijuana: effects on epilepsy and use by patients with epilepsy. Epilepsia 42:1266–1272
Herkenham M, Lynn AB, Johnson MR, Melvin LS, de Costa BR, Rice KC (1991) Characterization and localization of cannabinoid receptors in rat brain: a quantitative in vitro autoradiographic study. J Neurosci 11:563–583
Howlett AC (1995) Pharmacology of cannabinoid receptors. Annu Rev Pharmacol Toxicol 35:607–634
Howlett AC, Barth F, Bonner TI, Cabral G, Casellas P, Devane WA, Felder CC, Herkenham M, Mackie K, Martin BR, Mechoulam R, Pertwee RG (2002) International union of pharmacology. XXVII. Classification of cannabinoid receptors. Pharmacol Rev 54:161–202
Howlett AC, Breivogel CS, Childers SR, Deadwyler SA, Hampson RE, Porrino LJ (2004) Cannabinoid physiology and pharmacology: 30 years of progress. Neuropharmacology 47(Suppl 1):345–358
Jonker DM, Voskuyl RA, Danhof M (2007) Synergistic combinations of anticonvulsant agents: what is the evidence from animal experiments? Epilepsia 48:412–434
Kalviainen R (2001) Monotherapy trial design: conversion versus de novo. Epilepsy Res 45:75–78 Discussion 79–80
Karler R, Cely W, Turkanis SA (1973) The anticonvulsant activity of cannabidiol and cannabinol. Life Sci 13:1527–1531
Karler R, Cely W, Turkanis SA (1974) Anticonvulsant properties of delta 9-tetrahydrocannabinol and other cannabinoids. Life Sci 15:931–947
Knudsen FU (2000) Febrile seizures: treatment and prognosis. Epilepsia 41:2–9
Litchfield JT Jr, Wilcoxon F (1949) A simplified method of evaluating dose–effect experiments. J Pharmacol Exp Ther 96:99–113
Loscher W (2002a) Animal models of epilepsy for the development of antiepileptogenic and disease-modifying drugs. A comparison of the pharmacology of kindling and post-status epilepticus models of temporal lobe epilepsy. Epilepsy Res 50:105–123
Loscher W (2002b) Basic pharmacology of valproate: a review after 35 years of clinical use for the treatment of epilepsy. CNS Drugs 16:669–694
Loscher W (2002c) Current status and future directions in the pharmacotherapy of epilepsy. Trends Pharmacol Sci 23:113–118
Lozovaya N, Yatsenko N, Beketov A, Tsintsadze T, Burnashev N (2005) Glycine receptors in CNS neurons as a target for nonretrograde action of cannabinoids. J Neurosci 25:7499–7506
Luszczki JJ, Czuczwar P, Cioczek-Czuczwar A, Czuczwar SJ (2006) Arachidonyl-2′-chloroethylamide, a highly selective cannabinoid CB1 receptor agonist, enhances the anticonvulsant action of valproate in the mouse maximal electroshock-induced seizure model. Eur J Pharmacol 547:65–74
Lutz B (2002) Molecular biology of cannabinoid receptors. Prostaglandins Leukot Essent Fatty Acids 66:123–142
Lutz B (2004) On-demand activation of the endocannabinoid system in the control of neuronal excitability and epileptiform seizures. Biochem Pharmacol 68:1691–1698
Marsicano G, Lutz B (1999) Expression of the cannabinoid receptor CB1 in distinct neuronal subpopulations in the adult mouse forebrain. Eur J Neurosci 11:4213–4225
Marsicano G, Goodenough S, Monory K, Hermann H, Eder M, Cannich A, Azad SC, Cascio MG, Gutierrez SO, van der Stelt M, Lopez-Rodriguez ML, Casanova E, Schutz G, Zieglgansberger W, Di Marzo V, Behl C, Lutz B (2003) CB1 cannabinoid receptors and on-demand defense against excitotoxicity. Science 302:84–88
Matsuda LA, Lolait SJ, Brownstein MJ, Young AC, Bonner TI (1990) Structure of a cannabinoid receptor and functional expression of the cloned cDNA. Nature 346:561–564
McCaughran JA Jr, Corcoran ME, Wada JA (1974) Anticonvulsant activity of delta-8- and delta-9-tetrahydrocannabinol in rats. Pharmacol Biochem Behav 2:227–233
McKinney MK, Cravatt BF (2005) Structure and function of fatty acid amide hydrolase. Annu Rev Biochem 74:411–432
Mechoulam R, Gaoni Y (1965) A total synthesis of Dl-delta-1-tetrahydrocannabinol, the active constituent of hashish. J Am Chem Soc 87:3273–3275
Mechoulam R, Hanus L (2002) Cannabidiol: an overview of some chemical and pharmacological aspects. Part I: chemical aspects. Chem Phys Lipids 121:35–43
Perucca E (2004) NICE guidance on newer drugs for epilepsy in adults. BMJ 328:1273–1274
Piomelli D (2003) The molecular logic of endocannabinoid signalling. Nat Rev Neurosci 4:873–884
Ray AM, Benham CD, Roberts JC, Gill CH, Lanneau C, Gitterman DP, Harries M, Davis JB, Davies CH (2003) Capsazepine protects against neuronal injury caused by oxygen glucose deprivation by inhibiting I(h). J Neurosci 23:10146–10153
Robbe D, Kopf M, Remaury A, Bockaert J, Manzoni OJ (2002) Endogenous cannabinoids mediate long-term synaptic depression in the nucleus accumbens. Proc Natl Acad Sci USA 99:8384–8388
Rosman NP, Colton T, Labazzo J, Gilbert PL, Gardella NB, Kaye EM, Van Bennekom C, Winter MR (1993) A controlled trial of diazepam administered during febrile illnesses to prevent recurrence of febrile seizures. N Engl J Med 329:79–84
Sander JW (1993) Some aspects of prognosis in the epilepsies: a review. Epilepsia 34:1007–1016
Smith PF (2005) Cannabinoids as potential anti-epileptic drugs. Curr Opin Investig Drugs 6:680–685
Stella N, Schweitzer P, Piomelli D (1997) A second endogenous cannabinoid that modulates long-term potentiation. Nature 388:773–778
Treiman DM (1989) Pharmacokinetics and clinical use of benzodiazepines in the management of status epilepticus. Epilepsia 30(Suppl 2):S4–S10
Veldhuis WB, van der Stelt M, Wadman MW, van Zadelhoff G, Maccarrone M, Fezza F, Veldink GA, Vliegenthart JF, Bar PR, Nicolay K, Di Marzo V (2003) Neuroprotection by the endogenous cannabinoid anandamide and arvanil against in vivo excitotoxicity in the rat: role of vanilloid receptors and lipoxygenases. J Neurosci 23:4127–4133
Wallace MJ, Wiley JL, Martin BR, DeLorenzo RJ (2001) Assessment of the role of CB1 receptors in cannabinoid anticonvulsant effects. Eur J Pharmacol 428:51–57
Wallace MJ, Martin BR, DeLorenzo RJ (2002) Evidence for a physiological role of endocannabinoids in the modulation of seizure threshold and severity. Eur J Pharmacol 452:295–301
Wallace MJ, Blair RE, Falenski KW, Martin BR, DeLorenzo RJ (2003) The endogenous cannabinoid system regulates seizure frequency and duration in a model of temporal lobe epilepsy. J Pharmacol Exp Ther 307:129–137
Wilson RI, Nicoll RA (2002) Endocannabinoid signaling in the brain. Science 296:678–682
Woodbury LA, Davenport VD (1952) Design and use of a new electroshock seizure apparatus, and analysis of factors altering seizure threshold and pattern. Arch Int Pharmacodyn Ther 92:97–107
Woodbury LA, Swinyard CA (1952) Stimulus parameters for electroshock seizures in rats. Am J Physiol 170:661–667
Acknowledgments
This work was supported by a grant (308/A/A) from Neuroscience Research Center, Shahid Beheshti University, MC, Iran.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Naderi, N., Aziz Ahari, F., Shafaghi, B. et al. Evaluation of interactions between cannabinoid compounds and diazepam in electroshock-induced seizure model in mice. J Neural Transm 115, 1501–1511 (2008). https://doi.org/10.1007/s00702-008-0076-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00702-008-0076-x