Abstract
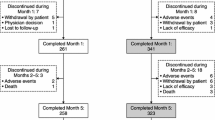
This was a retrospective pooled analysis of data from four comparably designed, double-blind, placebo-controlled, Phase III studies and their long-term open-label extensions. Patients on levodopa and a dopa decarboxylase inhibitor (DDCI) were randomized to entacapone or to placebo in the 6-month, double-blind phase, with all patients subsequently receiving entacapone in the extension phase. UPDRS III motor scores improved by −2.1 points during the first 6 months of levodopa/DDCI and entacapone therapy, and remained below baseline for up to 2 years. Increased daily ‘ON’ time, together with response duration to a single morning dose of levodopa and clinical global evaluation, also supported the long-term efficacy of levodopa/DDCI and entacapone. The mean daily dose of levodopa did not increase over the 5-year follow-up period. Long-term therapy with levodopa/DDCI and entacapone was well-tolerated.





Similar content being viewed by others
References
Alves G, Wentzel-Larsen T, Aarsland D, Larsen JP (2005) Progression of motor impairment and disability in Parkinson disease: a population-based study. Neurology 65(9):1436–1441
Bracco F, Battaglia A, Chouza C, Dupont E, Gershanik O, Marti Masso JF et al (2004) The long-acting dopamine receptor agonist cabergoline in early Parkinson’s disease: final results of a 5-year, double-blind, levodopa-controlled study. CNS Drugs 18(11):733–746
Brooks DJ, Sagar H (2003) Entacapone is beneficial in both fluctuating and non-fluctuating patients with Parkinson’s disease: a randomised, placebo controlled, double blind, six month study. J Neurol Neurosurg Psychiatry 74(8):1071–1079
Goetz CG, Stebbins GT, Blasucci LM (2000) Differential progression of motor impairment in levodopa-treated Parkinson’s disease. Mov Disord 15(3):479–484
Holloway RG, Shoulson I, Fahn S, Kieburtz K, Lang A, Marek K et al (2004) Pramipexole vs levodopa as initial treatment for Parkinson disease: a 4-year randomized controlled trial. Arch Neurol 61(7):1044–1053
Larsen JP, Worm-Petersen J, Siden A, Gordin A, Reinikainen K, Leinonen M (2003) The tolerability and efficacy of entacapone over 3 years in patients with Parkinson’s disease. Eur J Neurol 10(2):137–146
Myllyla V, Kultalahti ER, Haapaniemi H, Leinonen M (2001) Twelve-month safety of entacapone in patients with Parkinson’s disease. Eur J Neurol 8(1):53–60
Oertel WH, Wolters E, Sampaio C, Gimenez-Roldan S, Bergamasco B, Dujardin M et al (2006) Pergolide versus levodopa monotherapy in early Parkinson’s disease patients: The PELMOPET study. Mov Disord 21(3):343–353
Olanow CW, Agid Y, Mizuno Y, Albanese A, Bonuccelli U, Damier P et al (2004) Levodopa in the treatment of Parkinson’s disease: current controversies. Mov Disord 19(9):997–1005
Olanow CW, Obeso JA, Stocchi F (2006) Continuous dopamine-receptor treatment of Parkinson´s disease: scientific rationale and clinical implications. Lancet Neurol 5:677–687
Poewe WH, Deuschl G, Gordin A, Kultalahti ER, Leinonen M (2002) Efficacy and safety of entacapone in Parkinson’s disease patients with suboptimal levodopa response: a 6-month randomized placebo-controlled double-blind study in Germany and Austria (Celomen study). Acta Neurol Scand 105(4):245–255
PSG (1997) Entacapone improves motor fluctuations in levodopa-treated Parkinson’s disease patients. Parkinson Study Group. Ann Neurol 42(5):747–755
Rajput AH (2001) Levodopa prolongs life expectancy and is non-toxic to substantia nigra. Parkinsonism Relat Disord 8(2):95–100
Rascol O, Brooks DJ, Korczyn AD, De Deyn PP, Clarke CE, Lang AE (2000) A five-year study of the incidence of dyskinesia in patients with early Parkinson’s disease who were treated with ropinirole or levodopa. 056 Study Group. N Engl J Med 342(20):1484–1491
Rinne UK, Larsen JP, Siden A, Worm-Petersen J (1998) Entacapone enhances the response to levodopa in parkinsonian patients with motor fluctuations. Nomecomt Study Group. Neurology 51(5):1309–1314
Stocchi F (2006) The levodopa wearing-off phenomenon in Parkinson’s disease: pharmacokinetic considerations. Expert Opin Pharmacother 7(10):1399–1407
Acknowledgments
Studies included in this paper were sponsored by Orion Corporation, Orion Pharma. The authors would like to thank Diya Lahiri MSc PhD, for her editorial assistance.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Brooks, D.J., Leinonen, M., Kuoppamäki, M. et al. Five-year efficacy and safety of levodopa/DDCI and entacapone in patients with Parkinson’s disease. J Neural Transm 115, 843–849 (2008). https://doi.org/10.1007/s00702-008-0025-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00702-008-0025-8