Summary.
To study the relationship between the metabolism of amyloid precursor protein (APP) and cellular energy failure, HEK 293 cells stably transfected with βAPP 695 underwent graded energy failure induced either by i) hypoxia (pO2 25 mm Hg), ii) inhibition of the respiratory chain by sodium azide (NaN3), or iii) by combined glucose deprivation/hypoxia of different duration and severity.
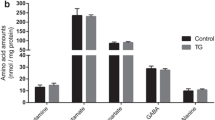
Secreted APP (APPs) and the derivative βA4 were quantified autoradiographically by immunoprecipitation, and [35S] methionine labeling. APP holoprotein (APPh) was determined by Western blot analysis. The concentrations of the energy-rich metabolites ATP, ADP, creatine phosphate (CrP), and adenosine were measured by high performance liquid chromatography.
Mild to moderate energy failure after NaN3 treatment (2h, 4h) and hypoxia (2h, 8h) was characterized by normal ATP concentration but also by a high reduction in CrP. A stress condition indicated by an increased ATP turnover and adenosine increase was obtained. Intracellular APPh increased but its metabolites APPs and βA4 as measured in the extracellular compartment decreased. These changes may point to a compensatory response of APP but also to a initial disturbance in intracellular APP metabolism. Severe abnormalities in both energy formation and utilization after 8h NaN3 and hypoxia glucose deprivation were found to be accompanied by a drastic fall in intracellular APPh concentration by at least 50%, paralleled by an accelerating reduction in the extracellular concentrations of both APPs and βA4.
A significant linear correlation between APPh and ATP and between CrP and βA4 became obvious.
The data of the present study indicate that abnormalities in APP metabolism were generated in an energy-dependent manner. The obvious similarities to sporadic Alzheimer s disease are discussed.
Similar content being viewed by others
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Hoyer, A., Bardenheuer, H., Martin, E. et al. Amyloid precursor protein (APP) and its derivatives change after cellular energy depletion. An in vitro-study. J Neural Transm 112, 239–253 (2005). https://doi.org/10.1007/s00702-004-0176-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00702-004-0176-1