Abstract
Purpose
Triggering of inflammatory responses and disruption of blood-spinal cord barrier (BSCB) integrity are considered pivotal events in the pathophysiology of traumatic spinal cord injury (TSCI). Yet, these events are poorly understood and described in humans. This study aims to describe inflammatory responses and BSCB integrity in human TSCI.
Methods
Fifteen TSCI patients and fifteen non-TSCI patients were prospectively recruited from Aarhus University Hospital, Denmark. Peripheral blood (PB) and cerebrospinal fluid (CSF) were collected at median day 0 [IQR: 1], median day 9 [IQR: 2], and median day 148 [IQR: 49] after injury. PB and CSF were analyzed for immune cells by flow cytometry, cytokines by multiplex immunoassay, and BSCB integrity by IgG Index.
Results
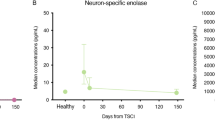
Eleven TSCI patients completed follow-up. Results showed alterations in innate and adaptive immune cell counts over time. TSCI patients had significantly increased cytokine concentrations in CSF at the first and second follow-up, while only concentrations of interleukin (IL)-4, IL-8, and tumor necrosis factor-α remained significantly increased at the third follow-up. In PB, TSCI patients had significantly increased IL-6, IL-8, and IL-10 concentrations and significantly decreased interferon-γ concentrations at the first follow-up. Results further showed increased IgG Index indicative of BSCB disruption in seven TSCI patients at the first follow-up, five TSCI patients at the second follow-up, and two patients at the third follow-up.
Conclusions
Our results suggest that TSCI mainly triggers innate inflammatory responses that resolves over time, although with some degree of non-resolving inflammation, particularly in CSF. Our results cannot confirm BSCB disruption in all TSCI patients.



Similar content being viewed by others
References
Alizadeh A, Dyck SM, Karimi-Abdolrezaee S (2019) Traumatic Spinal Cord Injury: An Overview of Pathophysiology, Models and Acute Injury Mechanisms. Front Neurol 10:1–25
Anwar MA, Al Shehabi TS, Eid AH (2016) Inflammogenesis of secondary spinal cord injury. Front Cell Neurosci 10:98. https://doi.org/10.3389/fncel.2016.00098
Bartanusz V, Jezova D, Alajajian B, Digicaylioglu M (2011) The blood-spinal cord barrier: morphology and clinical implications. Ann Neurol 70(2):194–206
Biglari B, Swing T, Child C, Büchler A, Westhauser F, Bruckner T, Ferbert T, Jürgen Gerner H, Moghaddam A (2015) A pilot study on temporal changes in IL-1β and TNF-α serum levels after spinal cord injury: the serum level of TNF-α in acute SCI patients as a possible marker for neurological remission. Spinal Cord 53(7):510–514
Brodin P, Davis MM (2017) Human immune system variation. Nat Rev Immunol 17(1):21–29
Carman CV, Martinelli R (2015) T lymphocyte-endothelial interactions: emerging understanding of trafficking and antigen-specific immunity. Front Immunol. https://doi.org/10.3389/fimmu.2015.00603
Carpenter RS, Jiang RR, Brennan FH, Hall JCE, Gottipati MK, Niewiesk S, Popovich PG (2019) Human immune cells infiltrate the spinal cord and impair recovery after spinal cord injury in humanized mice. Sci Rep 9(1):1–16
Casha S, Rice T, Stirling DP, Silva C, Gnanapavan S, Giovannoni G, John Hurlbert R, Wee Yong V (2018) Cerebrospinal fluid biomarkers in human spinal cord injury from a phase II minocycline trial. J Neurotrauma 35(16):1918–1928
Dalkilic T, Fallah N, Noonan VK et al (2018) Predicting injury severity and neurological recovery after acute cervical spinal cord injury: a comparison of cerebrospinal fluid and magnetic resonance imaging biomarkers. J Neurotrauma 35(3):435–445
de Mello RM, Oses JP, Kutchak FM et al (2019) Serum biomarkers and clinical outcomes in traumatic spinal cord injury: prospective cohort study. World Neurosurg 122:e1028–e1036
Davies AL, Hayes KC, Dekaban GA (2007) Clinical correlates of elevated serum concentrations of cytokines and autoantibodies in patients with spinal cord injury. Arch Phys Med Rehabil 88(11):1384–1393
Elizei SS, Kwon BK (2017) The translational importance of establishing biomarkers of human spinal cord injury. Neural Regen Res 12(3):385–388
Fernández M, Baldassarro VA, Capirossi R et al (2020) Possible strategies to optimize a biomarker discovery approach to correlate with neurological outcome in patients with spinal cord injury: a pilot study. J Neurotrauma 37(3):431–440
Fleming JC, Norenberg MD, Ramsay DA, Dekaban GA, Marcillo AE, Saenz AD, Pasquale-Styles M, Dietrich WD, Weaver LC (2006) The cellular inflammatory response in human spinal cords after injury. Brain 129(12):3249–3269
Gao K, Niu J, Dang X (2020) Neuroprotection of netrin-1 on neurological recovery via Wnt/β-catenin signaling pathway after spinal cord injury. NeuroReport 31(7):537–543
Gaojian T, Dingfei Q, Linwei L et al (2020) Parthenolide promotes the repair of spinal cord injury by modulating M1/M2 polarization via the NF-κB and STAT 1/3 signaling pathway. Cell Death Discovery. https://doi.org/10.1038/s41420-020-00333-8
Hayes KC, Hull TC, Delaney GA, Potter PJ, Sequeira KA, Campbell K, Popovich PG (2002) Elevated serum titers of proinflammatory cytokines and CNS autoantibodies in patients with chronic spinal cord injury. J Neurotrauma 19(6):753–761
Heller RA, Raven TF, Swing T et al (2017) CCL-2 as a possible early marker for remission after traumatic spinal cord injury. Spinal Cord 55(11):1002–1009
Huang W, Vodovotz Y, Kusturiss MB, Barclay D, Greenwald K, Boninger ML, Coen PM, Brienza D, Sowa G (2014) Identification of distinct monocyte phenotypes and correlation with circulating cytokine profiles in acute response to spinal cord injury: a pilot study. PM and R 6(4):332–341
Jin L-Y, Li J, Wang K-F, Xia W-W, Zhu Z-Q, Wang C-R, Li X-F, Liu H-Y (2021) Blood–spinal cord barrier in spinal cord injury: a review. J Neurotrauma 38:1–22
Kenward MG, Roger JH (1997) Small sample inference for fixed effects from restricted maximum likelihood. Biometrics 53(3):983–997
Kigerl KA, Gensel JC, Ankeny DP, Alexander JK, Donnelly DJ, Popovich PG (2009) Identification of two distinct macrophage subsets with divergent effects causing either neurotoxicity or regeneration in the injured mouse spinal cord. J Neurosci 29(43):13435–13444
Kwon BK, Streijger F, Fallah N et al (2017) Cerebrospinal fluid biomarkers to stratify injury severity and predict outcome in human traumatic spinal cord injury. J Neurotrauma 34(3):567–580
Kwon BK, Stammers AMT, Belanger LM et al (2010) Cerebrospinal fluid inflammatory cytokines and biomarkers of injury severity in acute human spinal cord injury. J Neurotrauma 27(4):669–682
Lin W, Chen W, Liu W, Xu Z, Zhang L (2019) Sirtuin4 suppresses the anti-neuroinflammatory activity of infiltrating regulatory T cells in the traumatically injured spinal cord. Immunology 158(4):362–374
Lindblad C, Nelson DW, Zeiler FA et al (2020) Influence of blood-brain barrier integrity on brain protein biomarker clearance in severe traumatic brain injury: a longitudinal prospective study. J Neurotrauma 37(12):1381–1391
Moghaddam A, Child C, Bruckner T, Gerner HJ (2015) Posttraumatic inflammation as a key to neuroregeneration after traumatic spinal cord injury. Int J Mol Sci 16:7900–7916
Monahan R, Stein A, Gibbs K, Bank M, Bloom O (2015) Circulating T cell subsets are altered in individuals with chronic spinal cord injury. Immunol Res 63(1–3):3–10
Riegger T, Conrad S, Schluesener HJ, Kaps HP, Badke A, Baron C, Gerstein J, Dietz K, Abdizahdeh M, Schwab JM (2009) Immune depression syndrome following human spinal cord injury (SCI): A pilot study. Neuroscience 158(3):1194–1199
Roberts TT, Leonard GR, Cepela DJ (2017) Classifications In Brief: American Spinal Injury Association (ASIA) Impairment Scale. Clin Orthop Relat Res 475(5):1499–1504
Schwab JM, Zhang Y, Kopp MA, Brommer B, Popovich PG (2014) The paradox of chronic neuroinflammation, systemic immune suppression, autoimmunity after traumatic chronic spinal cord injury. Exp Neurol 258:121–129
Sroga JM, Jones TB, Kigerl KA, McGaughy VM, Popovich PG (2003) Rats and mice exhibit distinct inflammatory reactions after spinal cord injury. J Comp Neurol 462(2):223–240
Stanley AC, Lacy P (2010) Pathways for cytokine secretion. Physiology 25(4):218–229
Sun G, Yang S, Cao G et al (2018) γδ T cells provide the early source of IFN-γ to aggravate lesions in spinal cord injury. J Exp Med 215(2):521–535
Svenningsson A, Andersen O, Edsbagge M, Stemme S (1995) Lymphocyte phenotype and subset distribution in normal cerebrospinal fluid. J Neuroimmunol 63:39–46
Vangansewinkel T, Lemmens S, Geurts N, Quanten K, Dooley D, Pejler G, Hendrix S (2019) Mouse mast cell protease 4 suppresses scar formation after traumatic spinal cord injury. Sci Rep 9(1):1–11
Vangansewinkel T, Geurts N, Quanten K, Nelissen S, Lemmens S, Geboes L, Dooley D, Vidal PM, Pejler G, Hendrix S (2016) Mast cells promote scar remodeling and functional recovery after spinal cord injury via mouse mast cell protease 6. FASEB J 30(5):2040–2057
Whetstone W, Hsu J, Eisenberg M, Werb Z, Noble-Haeusslein L (2003) Blood-spinal cord barrier after spinal cord injury: relation to revascularization and wound healing. J Neurosci Res 74(2):227–239
Yang L, Blumbergs PC, Jones NR, Manavis J, Sarvestani GT, Ghabriel MN (2004) Early expression and cellular localization of proinflammatory cytokines interleukin-1β, interleukin-6, and tumor necrosis factor-α in human traumatic spinal cord injury. Spine 29(9):966–971
Yu YA, Hotten DF, Malakhau Y, Volker E, Ghio AJ, Noble PW, Kraft M, Hollingsworth JW, Gunn MD, Tighe RM (2016) Flow cytometric analysis of myeloid cells in human blood, bronchoalveolar lavage, and lung tissues. Am J Respir Cell Mol Biol 54:13–24
Zrzavy T, Schwaiger C, Wimmer I, Berger T, Bauer J, Butovsky O, Schwab JM, Lassmann H, Höftberger R (2021) Acute and non-resolving inflammation associate with oxidative injury after human spinal cord injury. Brain : a J neurol 144(1):144–161
Acknowledgements
We sincerely thank all the patients who volunteered to be included in this study and the staff at Aarhus University Hospital for their participation. We thank Anne-Marie Toft for her support and assistance in performing the Conventional Flow Cytometry analyses. The Imaging Flow Cytometry analyses were performed at the FACS Core Facility at Aarhus University. We thank for their assistance in performing the analyses and interpretation of the results.
Funding
This research was funded by Lundbeckfonden; Aase og Ejnar Danielsens Fond; Grosserer L.F. Foghts Fond; Dagmar Marshalls Fond; A.P. Møller Fonden; Grosserer A.V. Lykfeldt og Hustrus Legat; Jascha Fonden; and UlykkesPatientForeningen.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Ethical approval
This research was approved by the Central Denmark Region Committees on Health Research Ethics (1–10-72–382-17) and the Danish Data Protection Agency (1–16-02–754-17) and registered on ClinicalTrials.gov (NCT03505463).
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
This article is part of the Topical Collection on Spine trauma
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Wichmann, T.O., Kasch, H., Dyrskog, S. et al. The inflammatory response and blood-spinal cord barrier integrity in traumatic spinal cord injury: a prospective pilot study. Acta Neurochir 164, 3143–3153 (2022). https://doi.org/10.1007/s00701-022-05369-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00701-022-05369-6