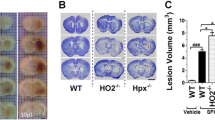
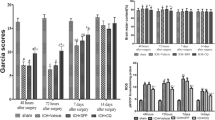
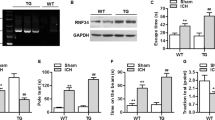
Abstract
Background
Heme oxygenase-1 (HO-1), the rate-limiting enzyme for heme catabolism and iron production, its role in intracerebral hemorrhage (ICH) is controversial. The study was to investigate correlations between brain oxidative injury and HO-1 after experimental ICH.
Method
Sprague–Dawley rats received intra-striatal infusions of 100 μl autologous whole blood as ICH models. HO-1 were examined by immunohistochemical and reverse transcription polymerase chain reaction (RT-PCR) analysis. Brain oxidative stress was quantitated by malondialdehyde (MDA); antioxidation were measured by copper–zinc superoxide dismutase (Cu/Zn-SOD) activity using RT-PCR assay.
Results
The expression of the HO-1 upregulated and reached its peak at days 3 and 7 after ICH (P < 0.01). There was a significant increase of MDA and a top at 3-day post-ICH (P < 0.01); Cu/Zn-SOD was upregulated post-ICH and reached the top at day 7 (P < 0.001); HO-1 was correlated significantly with brain MDA content at days 7 and 14 following ICH (r = 0.435–0.501, P < 0.001) but there is no definite correlation between them on 1 to 3 days (P > 0.05); conversely, HO-1 was correlated significantly with Cu/Zn-SOD on 1 to 3 days after ICH (r = 0.433–0.621, P < 0.001) but there is no definite correlation between them at days 7 and 14 (P > 0.05).
Conclusions
HO-1 has both antioxidant and prooxidant properties in ICH. The early upregulation of HO-1 possibly fit with the events and be protective against oxidative stress, whereas its overexpression in the late stages may result in its dysfunction and be toxic. So it should be prudent to intervene ICH with the inhibitor/activator of HO-1.








Similar content being viewed by others
Abbreviations
- DAB:
-
Diaminobenzidine
- ICH:
-
Intracerebral hemorrhage
- MDA:
-
Malondialdehyde
- Cu/Zn-SOD:
-
Copper–zinc superoxide dismutase
- HO-1:
-
Heme oxygenase-1
- RT-PCR:
-
Reverse transcription polymerase chain reaction
References
Bostanci MO, Bağirici F (2008) Nitric oxide synthesis inhibition attenuates iron-induced neurotoxicity: a stereological study. Neurotoxicology 29:130–135
Chen-Roetling J, Regan RF (2006) Effect of heme oxygenase-1 on the vulnerability of astrocytes and neurons to hemoglobin. Biochem Biophys Res Commun 350:233–237
Davis AS, Zhao H, Sun GH, Sapolsky RM, Steinberg GK (2007) Gene therapy using SOD1 protects striatal neurons from experimental stroke. Neurosci Lett 411:32–36
Dennis MS (2003) Outcome after brain haemorrhage. Cerebrovasc Dis 16:9–13
Dohi K, Mochizuki Y, Satoh K, Jimbo H, Hayashi M, Toyoda I, Ikeda Y, Abe T, Aruga T (2003) Transient elevation of serum bilirubin (a heme oxygenase-1 metabolite) level in hemorrhagic stroke: bilirubin is a marker of oxidant stress. Acta Neurochir Suppl 86:247–249
Ferris CD, Jaffrey SR, Sawa A, Takahashi M, Brady SD, Barrow RK, Tysoe SA, Wolosker H, Barañano DE, Doré S, Poss KD, Snyder SH (1999) Haem oxygenase-1 prevents cell death by regulating cellular iron. Nat Cell Biol 1:152–157
He Y, Hua Y, Song S, Liu W, Keep RF, Xi G (2008) Induction of autophagy in rat hippocampus and cultured neurons by iron. Acta Neurochir Suppl 105:29–32
Hua Y, Keep RF, Hoff JT, Xi G (2008) Deferoxamine therapy for intracerebral hemorrhage. Acta Neurochir Suppl 105:3–6
Hua Y, Keep RF, Hoff JT, Xi G (2003) Thrombin preconditioning attenuates brain edema induced by erythrocytes and iron. J Cereb Blood Flow Metab 23:1448–1454
Kanno H, Ozawa H, Dohi Y, Sekiguchi A, Igarashi K, Itoi E (2009) Genetic ablation of transcription repressor Bach1 reduces neural tissue damage and improves locomotor function after spinal cord injury in mice. J Neurotrauma 26:31–39
Katsu M, Niizuma K, Yoshioka H, Okami N, Sakata H, Chan PH (2010) Hemoglobin-induced oxidative stress contributes to matrix metalloproteinase activation and blood-brain barrier dysfunction in vivo. J Cereb Blood Flow Metab (in press)
Koeppen AH, Dickson AC, Smith J (2004) Heme oxygenase in experimental intracerebral hemorrhage: the benefit of tin-mesoporphyrin. J Neuropathol Exp Neurol 63:587–597
Nakamura T, Keep RF, Hua Y, Nagao S, Hoff JT, Xi G (2006) Iron-induced oxidative brain injury after experimental intracerebral hemorrhage. Acta Neurochir Suppl 96:194–198
Nishio S, Yunoki M, Noguchi Y, Kawauchi M, Asari S, Ohmoto T (1997) Detection of lipid peroxidation and hydroxyl radicals in brain contusion of rats. Acta Neurochir Suppl 70:84–86
Peluffo H, Acarin L, Arís A, González P, Villaverde A, Castellano B, González B (2006) Neuroprotection from NMDA excitotoxic lesion by Cu/Zn superoxide dismutase gene delivery to the postnatal rat brain by a modular protein vector. BMC Neurosci 7:35
Peluffo H, Acarin L, Faiz M, Castellano B, Gonzalez B (2005) Cu/Zn superoxide dismutase expression in the postnatal rat brain following an excitotoxic injury. J Neuroinflammation 2:12
Pyne-Geithman GJ, Morgan CJ, Wagner K, Dulaney EM, Carrozzella J, Kanter DS, Zuccarello M, Clark JF (2005) Bilirubin production and oxidation in CSF of patients with cerebral vasospasm after subarachnoid hemorrhage. J Cereb Blood Flow Metab 25(8):1070–1077
Qing WG, Dong YQ, Ping TQ, Lai LG, Fang LD, Min HW, Xia L, Heng PY (2009) Brain edema after intracerebral hemorrhage in rats: the role of iron overload and AQP4. J Neurosurg 110:462–468
Regan RF, Kumar N, Gao F, Guo Y (2002) Ferritin induction protects cortical astrocytes from heme-mediated oxidative injury. Neuroscience 113:985–994
Ryter WW, Tyrrell RM (2000) The heme synthesis and degradation pathways: role in oxidant sensitivity. Heme oxygenase has both pro- and antioxidant properties. Free Radic Biol Med 28:289–309
Salvador GA (2010) Iron in neuronal function and dysfunction. Biofactors 36(2):103–110
Shimada Y, Tsunoda H, Zang L, Hirano M, Oka T, Tanaka T (2009) Synergistic induction of heme oxygenase-1 by nicaraven after subarachnoid hemorrhage to prevent delayed cerebral vasospasm. Eur J Pharmacol 620(1–3):16–20
Song S, Hua Y, Keep RF, He Y, Wang J, Wu J, Xi G (2008) Deferoxamine reduces brain swelling in a rat model of hippocampal intracerebral hemorrhage. Acta Neurochir Suppl 105:13–18
Suttner DM, Dennery PA (1999) Reversal of HO-1 related cytoprotection with increased expression is due to reactive iron. FASEB J 13:1800–1809
Tang Y, Lu A, Aronow BJ, Wagner KR, Sharp FR (2002) Genomic responses of the brain to ischemic stroke, intracerebral haemorrhage, kainate seizures, hypoglycemia, and hypoxia. Eur J Neurosci 15:1937–1952
True AL, Olive M, Boehm M, San H, Westrick RJ, Raghavachari N, Xu X, Lynn EG, Sack MN, Munson PJ, Gladwin MT, Nabel EG (2007) Heme oxygenase-1 deficiency accelerates formation of arterial thrombosis through oxidative damage to the endothelium, which is rescued by inhaled carbon monoxide. Circ Res 101:893–901
Turner CP, Bergeron M, Matz P, Zegna A, Noble LJ, Panter SS, Sharp FR (1998) Heme oxygenase-1 is induced in glia throughout brain by subarachnoid hemoglobin. J Cereb Blood Flow Metab 18:257–273
Wagner KR, Sharp FR, Ardizzone TD, Lu A, Clark JF (2003) Heme and iron metabolism: role in cerebral hemorrhage. J Cereb Blood Flow Metab 23:629–652
Wang J, Doré S (2007) Heme oxygenase-1 exacerbates early brain injury after intracerebral haemorrhage. Brain 130:1643–1652
Welch KD, Davis TZ, Van Eden ME, Aust SD (2002) Deleterious Iron-mediated oxidation of bio-molecules. Free Radic Biol Med 32:577–583
Wu J, Hua Y, Keep RF, Nakamura T, Hoff JT, Xi G (2003) Iron and iron-handling proteins in the brain after intracerebral hemorrhage. Stroke 34:2964–2969
Wu J, Hua Y, Keep RF, Schallert T, Hoff JT, Xi G (2002) Oxidative brain injury from extravasated erythrocytes after intracerebral hemorrhage. Brain Res 953:45–52
Xi G, Keep RF, Hoff JT (2006) Mechanisms of brain injury after intracerebral haemorrhage. Lancet Neurol 5:53–63
Conflicts of interest
None.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Wang, G., Yang, Q., Li, G. et al. Time course of heme oxygenase-1 and oxidative stress after experimental intracerebral hemorrhage. Acta Neurochir 153, 319–325 (2011). https://doi.org/10.1007/s00701-010-0750-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00701-010-0750-2