Abstract
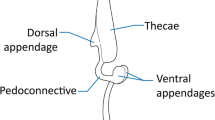
In flowers of Chimonanthus (Calycanthaceae), stamen movements have been known for 150 years. Stamens are initially recurved when flowers open. Subsequently, within approximately 48 h, the stamens gradually turn upright and eventually enclose the gynoecium. However, the exact mechanism involved in this process is still unresolved. In this study, we found that in recurved stamens at the female stage the cell lengths in the epidermal and hypodermal tissue of the abaxial surfaces were significantly smaller than those of the adaxial surfaces. In erect stamens, however, no significant difference in cell length between the hypodermal tissues of both surfaces was found. Although the difference in cell length between the epidermal tissues for erect stamens was significant, it was smaller than that in recurved filaments. We found no variations in cell number between the epidermal tissues or between the hypodermal tissues among recurved and erect stamens. Thus, we conclude that in Chimonanthus the differential cell growth rates between the adaxial and abaxial surfaces of filaments could account for the gradual inward stamen movement following flower opening rather than cell division. Furthermore, application of indole-3-acetic acid (IAA) and gibberellic acid (GA3) to intact flowers in vitro or the wounding of stigmas significantly promoted the stamen erection rate. Surprisingly, we did not observe any effect on this movement following hand-pollination. Different patterns of stamen movement in Calycanthaceae and their implications for reproductive biology are also discussed.




Similar content being viewed by others
References
Azuma H, Toyota M, Asakawa Y (2005) Floral scent chemistry and stamen movement of Chimonanthus praecox (L.) Link (Calycanthaceae). Acta Phytotax Geobot 56:197–201
Bennett AW (1869) On the fertilisation of winter-flowering plants. Nature 1:11–13
Bialczyk J, Lechowski Z (1988) The seismonastic movements of plant organs. Wiad Bot 32:209–226
Blake ST (1972) Idiospermum (Idiospermaceae), a new genus and family for Calycanthus australiensis. Contrib Queensland Herb 12:1–39
Cheng WC, Chang SY (1963) Calycanthus chinensis section Sinocalycanthus. Sci Silvae 8:1–2
Cowan AK, Taylor NJ, van Staden J (2005) Hormone homeostasis and induction of the small-fruit phenotype in ‘Hass’ avocado. Plant Growth Regul 45:11–19
Cruden RW, Hermann-Parker SM (1977) Temporal dioecism: an alternative to dioecism? Evolution 31:863–866
Dickison WC (1990) A study of the floral morphology an anatomy of the Caryocaraceae. Bull Torrey Bot Club 117:123–137
Edwards J, Whitaker D, Klionsky S, Laskowski MJ (2005) A record-breaking pollen catapult. Nature 435:164
Endress PK (1984) The role of inner staminodes in the floral display of some relic Magnoliales. Plant Syst Evol 146:269–282
Endress PK (2010) The evolution of floral biology in basal angiosperms. Phil Trans R Soc B 365:411–421
Grant V (1950) The pollination of Calycanthus occidentalis. Am J Bot 37:294–297
Greyson RI (1994) The development of flowers. Oxford University Press, Oxford
Heywood VH (1978) Flowering plants of the world. Oxford University Press, Oxford
Knuth P, Loew E (1899) Handbuch der Blütenbiologie, vol 2. Engelmann, Leipzig
Koevenig JL (1973) Floral development and stamen filament elongation in Cleome hassleriana. Am J Bot 60:122–129
Koning RE (1983) The role of auxin, ethylene, and acid growth in filament elongation in Gaillardia grandiflora (Asteraceae). Am J Bot 70:602–610
Koning RE, Raab MM (1987) Parameters of filament elongation in Ipomoea nil (Convolvulaceae). Am J Bot 74:510–516
Lechowski Z, Bialczyk J (1992) Effect of external calcium on the control of stamen movement in Berberis vulgaris L. Biol Plantarum 34:121–130
Li J, Ledger J, Ward T, del Tredici P (2004) Phylogenetics of Calycanthaceae based on molecular and morphological data with a special reference to divergent paralogues of the nrDNA ITS region. Harv Pap Bot 9:69–82
Li YJ, Pang XL, Si HQ, Wang HY, Li D (2007) Observing and analyzing the blossom and fragrance habits of Jing Guan’s Chimononthus praecox (Linn.) Link. J Southwest China Norm Univ (Nat Sci) 32:115–119. (In Chinese)
Liu KW, Liu ZJ, Huang LQ, Li LQ, Chen LJ, Tang GD (2006) Self-fertilization strategy in an orchid. Nature 441:945
Lobelo G, Fambrini M, Baraldi R, Lercari B, Pugliesi C (2000) Hormonal influence on photocontrol of the protandry in the genus Helianthus. J Exp Bot 51:1403–1412
Murashige T, Skoog F (1962) A revised medium for rapid growth and bio assays with tobacco tissue cultures. Physiol Plant 15:473–497
Nagy ES, Strong L, Galloway LF (1999) Contribution of delayed autonomous selfing to reproductive success in Mountain Laurel, Kalmia latifolia (Ericaceae). Am Midl Nat 142:39–46
Nicely KA (1965) A monographic study of the Calycanthaceae. Castanea 30:38–81
Prance CT, Da Silva MF (1973) A monograph of the Caryocaraceae. F1. Neotropica Monogr. 12. Hafner Press, New York, pp 1–75
Ren MX (2010) Stamen movements in hermaphroditic flowers: diversity and adaptive significance. Chinese J Plant Ecol 34:867–875 (In Chinese)
Schlindwein C, Wittmann D (1997) Stamen movement in flowers of Opuntia (Cactaceae) favour oligolectic pollinators. Plant Syst Evo 204:179–193
Sheng AW, Guo WM, Sun ZH (1999) Study on dynamics of endogenous hormones and parameters concerned senescence in cut wintersweet flowers. J Beijing For Univ 21:48–53 (In Chinese)
Song JD, Lee DH, Rhew TH, Lee CH (2003) Wound-induced expression of ACC synthase genes in etiolated Mung Bean hypocotyls. J Plant Biol 46:199–203
Staedler YM, Weston PH, Endress PK (2007) Floral phyllotaxis and floral architecture in Calycanthaceae (Laurales). Int J Plant Sci 168:285–306
Staedler YM, Weston PH, Endress PK (2009) Comparative gynoecium structure and development in Calycanthaceae (Laurales). Int J Plant Sci 170:21–41
Sui SZ (2006) Analysis of expressed sequence tags (ESTs) from Chimonanthus praecox Link flower and isolation and function analysis of Cplectin gene [D], Southwest University, Chongqing (In Chinese)
Tashiro S, Tian CE, Watahiki MK, Yamamoto KT (2009) Changes in growth kinetics of stamen filaments cause inefficient pollination in massugu2, an auxin insensitive, dominant mutant of Arabidopsis thaliana. Physiol Plant 137:175–187
Taylor PE, Card G, House J, Dickinson MH, Flagan RC (2006) High-speed pollen release in the white mulberry tree, Morus alba L. Sex Plant Reprod 19:19–24
Wang XF, Tan YY, Chen JH, Lu YT (2006) Pollen tube reallocation in two preanthesis cleistogamous species, Ranalisma rostratum and Sagittaria guyanensis ssp. lappula (Alismataceae). Aquat Bot 85:233–240
Weigend M, Gottschling M (2006) Evolution of funnel-revolver flowers and ornithophily in Nasa (Loasaceae). Plant Biol 8:120–142
Wen J (1999) Evolution of eastern Asian and eastern North American disjunct pattern in flowering plants. Annu Rev Ecol Syst 30:421–455
Wilson CL (1976) Floral anatomy of Idiospermum australiense (Idiospermaceae). Am J Bot 63:987–996
Worboys SJ (1998) Pollination processes and population structure of Idiospermum australiense (Diels) S.T. Blake, a primitive tree of the Queensland wet Tropics. MSc thesis. James Cook University of North Queensland
Worboys SJ, Jackes BR (2005) Pollination processes in Idiospermum australiense (Calycanthaceae), an arborescent basal angiosperm of Australia’s tropical rain forest. Plant Syst Evol 251:107–117
Wu CL, Hu NZ (1995) Studies on the flower form and blooming characteristics of the wintersweet. Acta Horticult Sin 22:277–282 (In Chinese)
Yu YB, Yang SF (1980) Biosynthesis of wound ethylene. Plant Physiol 66:281–285
Yu JQ, Li Y, Qian YR, Zhu ZJ (2001) Changes of endogenous hormone level in pollinated and N-(2-chloropyridyl)-N′–phenylurea (CPPU)-induced parthenocarpic fruits of Lagenaria leucantha. J Hort Sci Biotechnol 76:231–234
Zhou LH, Hao RM, Wu JZ, Mao ZB (2003) Pollination biology of Chimonanthus nitens Oliv. Acta Horticult Sin 30:690–694 (In Chinese)
Zhou LH, Hao RM, Wu JZ (2006a) The pollination biology of Chimonanthus praecox (L.) Link (Calycanthaceae). Acta Horticult Sin 33:323–327 (In Chinese)
Zhou SL, Renner SS, Wen J (2006b) Molecular phylogeny and intra- and inter-continental biogeography of Calycanthaceae. Mol Phylogenet Evol 39:1–15
Acknowledgments
We thank Professor Peter K. Endress (University of Zurich, Switzerland) and two anonymous reviewers for their valuable and inspiring comments on the manuscript. This work is supported by the National Natural Science Foundation of China (grant no. 30970194).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Du, W., Qin, KZ. & Wang, XF. The mechanism of stamen movement in Chimonanthus praecox (Calycanthaceae): differential cell growth rates on the adaxial and abaxial surfaces of filaments after flower opening. Plant Syst Evol 298, 561–567 (2012). https://doi.org/10.1007/s00606-011-0566-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00606-011-0566-4