Abstract
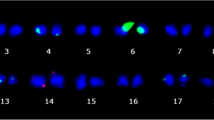
Species in the genus Curcuma (Zingiberaceae) that are cultivated widely in Thailand for their phytoestrogen-producing rhizomes are called wan-chak-motluk. Five cultivars belonging to Curcuma comosa (cultivars with 2n = 42 and 63) and Curcuma elata (2n = 63) were examined using the molecular cytogenetic method of fluorescence in situ hybridisation (FISH) in order to identify genetic relationships among these cultivars based on chromosomal maps of the 18S–25S ribosomal loci. The results revealed hybrid features in this Curcuma species group and a significant similarity among wan-chak-motluk cultivars. The main features included: (1) the presence of the single largest ribosomal site, assigned the Cc1 marker site, in the somatic 2n complement of all cultivars, and (2) the odd numbers of ribosomal sites in the complements, most often in sets of three. We therefore propose that the cultivar with 2n = 42 (C. comosa) is a homoploid hybrid species comprised of two different ancestral genomes and has a diploid status with the basic chromosome number x = 21. The cultivars with 2n = 63 (C. comosa and C. elata) are most probably triploids arising within the 2n = 42 diploid species/cultivars via a meiotic modification, rather than from hybridisation between diploid and tetraploid plants. The knowledge about genetic and genomic relationships among wan-chak-motluk cultivars will be important in the research projects that aim to explore and promote new plant materials for cultivation.

Similar content being viewed by others
References
Anamthawat-Jónsson K (2003) Preparation of chromosomes from plant leaf meristems for karyotype analysis and in situ hybridization. Met Cell Sci 25:91–95
Anamthawat-Jónsson K, Bödvarsdóttir SK (2001) Genomic and genetic relationships among species of Leymus (Poaceae: Triticeae) inferred from 18S.26S ribosomal genes. Amer J Bot 88:553–559
Anamthawat-Jónsson K, Heslop-Harrison JS (1995) Molecular cytogenetics of Icelandic birch species: physical mapping by in situ hybridization and rDNA polymorphism. Can J Forest Res 25:101–108
Anamthawat-Jónsson K, Schwarzacher T, Leitch AR, Bennett MD, Heslop-Harrison JS (1990) Discrimination between closely related Triticeae species using genomic DNA as a probe. Theor Appl Genet 79:721–728
Anamthawat-Jónsson K, Wenke T, ÆTh Thórsson, Sveinsson S, Zakrzewski F, Schmidt T (2009) Evolutionary diversification of satellite DNA sequences from Leymus (Poaceae: Triticeae). Genome 54:381–390
Arnold ML (1992) Natural hybridization as an evolutionary process. Annu Rev Ecol Syst 23:237–261
Begum R, Alam SS, Menzel G, Schmidt T (2009) Comparative molecular cytogenetics of major repetitive sequence families of three Dendrobium species (Orchidaceae) from Bangladesh. Ann Bot 104:863–872
Buerkle CA, Rieseberg LH (2008) The rate of genome stabilization in homoploid hybrid species. Evolution 62:266–275
Chokchaichamnankit P, Anamthawat-Jónsson K, Chulalaksananukul W (2008) Chromosomal mapping of 18S–25S and 5S ribosomal genes on 15 species of Fagaceae from northern Thailand. Silvae Genet 57:5–13
Contento A, Heslop-Harrison JS, Schwarzacher T (2005) Diversity of major repetitive DNA sequence in diploid and polyploidy Triticeae. Cytogenet Genome Res 109:34–42
Gerlach WL, Bedbrook JR (1979) Cloning and characterization of ribosomal RNA genes from wheat and barley. Nucleic Acids Res 7:1869–1885
Grant V (1982) Periodicities in the chromosome numbers of the angiosperms. Bot Gaz 143:379–389
Gross BL, Rieseberg LH (2005) The ecological genetics of homoploid hybrid speciation. J Hered 96:241–252
Guerra M (2008) Chromosome numbers in plant cytotaxonomy: concepts and implications. Cytogenet Genome Res 120:339–350
Howarth DG, Baum DA (2005) Genealogical evidence of homoploid hybrid speciation in an adaptive radiation of Scaevola (Goodeniaceae) in the Hawaiian Islands. Evolution 59:948–961
Islam MA, Meister A, Schubert V, Kloppstech K, Esch E (2007) Genetic diversity and cytogenetic analysis in Curcuma zedoaria (Christm.) Roscue from Bangladesh. Genet Resources Crop Evol 54:149–156
Jurgens TM, Frazier EG, Schaeffer JM, Jones TE, Zink DL, Borris RP, Nanakorn W, Beck HT, Balick MJ (1994) Novel nematocidal agents from Curcuma comosa. J Nat Prod 57:230–235
Leong-Skornickova J, Sida O, Jarolimova V, Sabu M, Fer T, Travnicek P, Suda J (2007) Chromosome numbers and genome size variation in Indian species of Curcuma (Zingiberaceae). Ann Bot 100:505–526
Mir C, Toumi L, Jarne P, Sarda V, Di Giusto F, Lumaret R (2006) Endemic North African Quercus afares Pomel originates from hybridisation between two genetically very distant oak species (Q. suber L. and Q. canariensis Willd.): evidence from nuclear and cytoplasmic markers. Heredity 96:175–184
Nahar L, Sarker SD (2007) Phytochemistry of the genus Curcuma. In: Ravindran PN, Babu KN, Sivaraman K (eds) Turmeric: the genus Curcuma. CRC Press, USA, pp 71–106
Nakamura S, Qu Y, Xu F, Matsuda H, Yoshikawa M (2008) Structures of new monoterpenes from Thai herbal medicine Curcuma comosa. Chem Pharm Bull 56:1604–1606
Orgaard M, Jacobsen N, Heslop-Harrison JS (1995) The hybrid origin of two cultivars of Crocus (Iridaceae) analysed by molecular cytogenetics including genomic Southern and in situ hybridizaiton. Ann Bot 76:253–262
Piyachaturawat P, Charoenpiboonsin J, Toskulkao C, Suksamrarn A (1999) Reduction of plasma cholesterol by Curcuma comosa extract in hypercholesteromic hamsters. J Ethnopharm 66:199–204
Prana MS, Sastrapradja S, Hawkes JG, Lubis I (1978) A cytological study of some Indonesian Curcuma species. J Root Crops 4:31–35
Qu Y, Xu F, Nakamura S, Matsuda H, Pongpiriyadacha Y, Wu L, Yoshikawa M (2009) Sesquiterpenes from Curcuma comosa. J Nat Med 63:102–104
Ramachandran K (1961) Chromosome numbers in the genus Curcuma Linn. Curr Sci 30:194–196
Rieseberg LH (1997) Hybrid origins of plant species. Annu Rev Ecol Syst 28:359–389
Rieseberg LH, Willis JH (2007) Plant Speciation. Science 317:910–914
Sharma G, Gohil RN (2008) Intrapopulation karyotypic variability in Allium roylei Stearn—a threatened species. Bot J Linn Soc 158:242–248
Siroky B (2008) Chromosome landmarks as tools to study the genome of Arabidopsis thaliana. Cytogenet Genome Res 120:202–209
Sodsai A, Piyachaturawat P, Sophasan S, Suksamrarn A, Vongsakul M (2007) Suppression by Curcuma comosa Roxb. of pro-inflammatory cytokine secretion in phorbol-12-myristate-13-acetate stimulated human mononuclear cells. Int Immunopharm 7:524–531
Soontornchainaksaeng P, Jenjittikul T (2010) Chromosome number variation of phytoestrogen-producing Curcuma (Zingiberaceae) from Thailand. J Nat Med 64:370–377
Soontornchainaksaeng P, Chantaranothai P, Senakun C (2003) Genetic diversity of Croton L. (Euphorbiaceae) in Thailand. Cytologia 68:379–382
Stebbins GL (1971) Chromosome evolution in higher plants. Addison–Wesley, Massachusetts
Stevens JP, Bougourd SM (1991) The frequency and meiotic behaviour of structural chromosome variants in natural populations of Allium schoenoprasum L. (wild chives) in Europe. Heredity 66:391–401
Suksamrarn A, Ponglikitmongkol M, Wongkrajang K, Chindaduang A, Kittidanairak S, Jankam A, Yingyongnarongkul B, Kittikanumat N, Chokchaisiri R, Khetkam P, Piyachaturawat P (2008) Diarylheptanoids, new phytoestrogens from the rhizomes of Curcuma comosa: isolation, chemical modification and estrogenic activity evaluation. Bioorg Med Chem 16:6891–6902
Takano A, Okada H (2002) Multiple occurrence of triploid formation in Globba (Zingiberaceae) from molecular evidence. Plant Syst Evol 230:143–159
Thampithak A, Jaisin Y, Meesarapee B, Chongthammakun S, Piyachaturawat P, Govitrapong P, Supavilai P, Sanvarinda Y (2009) Transcriptional regulation of iNOS and COX-2 by a novel compound from Curcuma comosa in lipopolysaccharide-induced microglial activation. Neurosci Lett 462:171–175
Winuthayanon W, Piyachaturawat P, Suksamrarn A, Ponglikitmongkol M, Arao Y, Hewitt S, Korach KS (2009) Diarylheptanoid phytoestrogens isolated from the medicinal plant Curcuma comosa: biologic actions in vitro and in vivo indicate estrogen receptor-dependent mechanisms. Environ Health Perspect 117:1155–1161
Xu F, Nakamura S, Qu Y, Matsuda H, Pongpiriyadacha Y, Wu L, Yoshikawa M (2009) Structures of new sesquiterpenes from Curcuma comosa. Chem Pharm Bull 56:1710–1716
Acknowledgments
This work was supported by Mahidol University, the National Research Council of Thailand, the Office of the Higher Education Commission of Thailand and the University of Iceland. We are grateful to Prof. Pawinee Piyachaturawat of Mahidol University for valuable comments on the project. We thank Dr Thaya Jenjittikul of Mahidol University for her contribution in sample collection and taxonomic identification of plant materials and Dr Ploenpit Chokchaichamnankit of Chulalongkorn University for assistance with the optimization of molecular cytogenetic protocols. We also thank the staff of the Cytogenetic Research Laboratory of the Botany Department, Mahidol University, for their assistance, especially Jatuporn Chandrmai and Tidarat Puangpairote.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Soontornchainaksaeng, P., Anamthawat-Jónsson, K. Ribosomal FISH mapping reveals hybridity in phytoestrogen producing Curcuma species from Thailand. Plant Syst Evol 292, 41–49 (2011). https://doi.org/10.1007/s00606-010-0408-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00606-010-0408-9