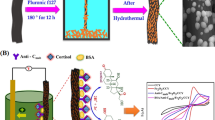
Abstract
Sweat is easily accessible from the human skin’s surface. It is secreted by the eccrine glands and contains a wealth of physiological information, including metabolites and electrolytes like glucose and Na ions. Sweat is a particularly useful biofluid because of its easy and non-invasive access, unlike other biofluids, like blood. On the other hand, nanomaterials have started to show promise operation as a competitive substitute for biosensors and molecular sensors throughout the last 10 years. Among the most synthetic nanomaterials that are studied, applied, and discussed, carbon nanomaterials are special. They are desirable candidates for sensor applications because of their many intrinsic electrical, magnetic, and optical characteristics; their chemical diversity and simplicity of manipulation; their biocompatibility; and their effectiveness as a chemically resistant platform. Carbon nanofibers (CNFs), carbon dots (CDs), carbon nanotubes (CNTs), and graphene have been intensively investigated as molecular sensors or as components that can be integrated into devices. In this review, we summarize recent advances in the use of carbon nanomaterials as sweat sensors and consider how they can be utilized to detect a diverse range of analytes in sweat, such as glucose, ions, lactate, cortisol, uric acid, and pH.
Graphical Abstract











Similar content being viewed by others
Data availability
Data will be made available on request.
References
Gao W et al (2016) Fully integrated wearable sensor arrays for multiplexed in situ perspiration analysis. Nature 529(7587):509–514
Sonner Z et al (2015) The microfluidics of the eccrine sweat gland, including biomarker partitioning, transport, and biosensing implications. Biomicrofluidics 9(3):031301
De Giovanni N, Fucci N (2013) The current status of sweat testing for drugs of abuse: a review. Curr Med Chem 20(4):545–561
Kim J et al (2019) Wearable biosensors for healthcare monitoring. Nat Biotechnol 37(4):389–406
Pirovano P et al (2020) A wearable sensor for the detection of sodium and potassium in human sweat during exercise. Talanta 219:121145
Zhai Q et al (2020) Vertically aligned gold nanowires as stretchable and wearable epidermal ion-selective electrode for noninvasive multiplexed sweat analysis. Anal Chem 92(6):4647–4655
Mitsubayashi K et al (1994) Analysis of metabolites in sweat as a measure of physical condition. Anal Chim Acta 289(1):27–34
Zhang H et al (2022) Integrated solid-state wearable sweat sensor system for sodium and potassium ion concentration detection. Sens Rev 42(1):76–88
Bariya M, Nyein HYY, Javey A (2018) Wearable sweat sensors. Nat Electron 1(3):160–171
Al-Omari M et al (2014) Detection of relative [Na+] and [K+] levels in sweat with optical measurements. J Appl Phys 115(20):203107
Keene ST et al (2019) Wearable organic electrochemical transistor patch for multiplexed sensing of calcium and ammonium ions from human perspiration. Adv Healthcare Mater 8(24):1901321
Schittek B et al (2001) Dermcidin: a novel human antibiotic peptide secreted by sweat glands. Nat Immunol 2(12):1133–1137
Shipway AN, Katz E, Willner I (2000) Nanoparticle arrays on surfaces for electronic, optical, and sensor applications. ChemPhysChem 1(1):18–52
Zhou Y, Xu Z, Yoon J (2011) Fluorescent and colorimetric chemosensors for detection of nucleotides, FAD and NADH: highlighted research during 2004–2010. Chem Soc Rev 40(5):2222–2235
Taniselass S, Arshad MM, Gopinath SC (2019) Graphene-based electrochemical biosensors for monitoring noncommunicable disease biomarkers. Biosens Bioelectron 130:276–292
Bauer M et al (2021) Electrochemical multi-analyte point-of-care perspiration sensors using on-chip three-dimensional graphene electrodes. Anal Bioanal Chem 413:763–777
Jalal AH et al (2018) Prospects and challenges of volatile organic compound sensors in human healthcare. Acs Sensors 3(7):1246–1263
Gao W et al (2016) Wearable microsensor array for multiplexed heavy metal monitoring of body fluids. Acs Sensors 1(7):866–874
Bandodkar AJ, Jeerapan I, Wang J (2016) Wearable chemical sensors: present challenges and future prospects. Acs Sensors 1(5):464–482
Lee H et al (2018) Enzyme-based glucose sensor: from invasive to wearable device. Adv Healthcare Mater 7(8):1701150
Rathi P et al (2017) Real-time, wearable, biomechanical movement capture of both humans and robots with metal-free electrodes. ACS Omega 2(8):4132–4142
Gong S, Cheng W (2017) One-dimensional nanomaterials for soft electronics. Adv Electron Mater 3(3):1600314
Jang H et al (2016) Graphene-based flexible and stretchable electronics. Adv Mater 28(22):4184–4202
Stankovich S et al (2006) Graphene-based composite materials. Nature 442(7100):282–286
Ambrosi A et al (2014) Electrochemistry of graphene and related materials. Chem Rev 114(14):7150–7188
Tiwari JN et al (2016) Engineered carbon-nanomaterial-based electrochemical sensors for biomolecules. ACS Nano 10(1):46–80
Liu Q et al (2007) Direct electrochemistry of glucose oxidase and electrochemical biosensing of glucose on quantum dots/carbon nanotubes electrodes. Biosens Bioelectron 22(12):3203–3209
Goyal RN, Singh SP (2006) Voltammetric quantification of adenine and guanine at C60 modified glassy carbon electrodes. J Nanosci Nanotechnol 6(12):3699–3704
Tucek J et al (2014) Iron-oxide-supported nanocarbon in lithium-ion batteries, medical, catalytic, and environmental applications. ACS Nano 8(8):7571–7612
Balasubramanian K, Burghard M (2006) Biosensors based on carbon nanotubes. Anal Bioanal Chem 385:452–468
di Sant’Agnese PA et al (1953) Abnormal electrolyte composition of sweat in cystic fibrosis of the pancreas: clinical significance and relationship to the disease. Pediatrics 12(5):549–563
Kroto HW et al (1985) C60: Buckminsterfullerene. Nature 318(6042):162–163
Lynch A, Diamond D, Leader M (2000) Point-of-need diagnosis of cystic fibrosis using a potentiometric ion-selective electrode array. Analyst 125(12):2264–2267
Weber J et al (2006) Novel lactate and pH biosensor for skin and sweat analysis based on single walled carbon nanotubes. Sens Actuators, B Chem 117(1):308–313
Lamas-Ardisana PJ et al (2014) Disposable amperometric biosensor based on lactate oxidase immobilised on platinum nanoparticle-decorated carbon nanofiber and poly (diallyldimethylammonium chloride) films. Biosens Bioelectron 56:345–351
Lei Y et al (2019) Laser-scribed graphene electrodes derived from lignin for biochemical sensing. ACS Applied Nano Mater 3(2):1166–1174
Zheng XT et al (2020) Noncovalent fluorescent biodot–protein conjugates with well-preserved native functions for improved sweat glucose detection. Bioconjug Chem 31(3):754–763
Choi J et al (2018) Skin-interfaced systems for sweat collection and analytics. Sci Adv 4(2):eaar3921
Choi J et al (2019) Soft, skin-integrated multifunctional microfluidic systems for accurate colorimetric analysis of sweat biomarkers and temperature. ACS Sensors 4(2):379–388
Sekine Y et al (2018) A fluorometric skin-interfaced microfluidic device and smartphone imaging module for in situ quantitative analysis of sweat chemistry. Lab Chip 18(15):2178–2186
Cui Y et al (2020) Ratiometric fluorescent nanohybrid for noninvasive and visual monitoring of sweat glucose. ACS Sensors 5(7):2096–2105
Bandodkar AJ et al (2019) Battery-free, skin-interfaced microfluidic/electronic systems for simultaneous electrochemical, colorimetric, and volumetric analysis of sweat. Sci Adv 5(1):eaav3294
Zhang Y et al (2019) Passive sweat collection and colorimetric analysis of biomarkers relevant to kidney disorders using a soft microfluidic system. Lab Chip 19(9):1545–1555
Kim SB et al (2020) Soft, skin-interfaced microfluidic systems with integrated enzymatic assays for measuring the concentration of ammonia and ethanol in sweat. Lab Chip 20(1):84–92
Reeder JT et al (2019) Waterproof, electronics-enabled, epidermal microfluidic devices for sweat collection, biomarker analysis, and thermography in aquatic settings. Sci Adv 5(1):eaau6356
Wongkaew N et al (2018) Functional nanomaterials and nanostructures enhancing electrochemical biosensors and lab-on-a-chip performances: recent progress, applications, and future perspective. Chem Rev 119(1):120–194
Ghaffari R, Rogers JA, Ray TR (2021) Recent progress, challenges, and opportunities for wearable biochemical sensors for sweat analysis. Sens Actuators, B Chem 332:129447
Bard AJ, Faulkner LR, White HS (2022) Electrochemical methods: fundamentals and applications. John Wiley & Sons
Wang Z et al (2021) Engineering materials for electrochemical sweat sensing. Adv Func Mater 31(12):2008130
Scozzari A (2008) Electrochemical sensing methods: a brief review. In: Evangelista V, Barsanti L, Frassanito AM, Passarelli V, Gualtieri P (eds) Algal Toxins: Nature, Occurrence, Effect and Detection. NATO Science for Peace and Security Series A: Chemistry and Biology. Springer, Dordrecht
Damiati S, Schuster B (2020) Electrochemical biosensors based on S-layer proteins. Sensors 20(6):1721
Grieshaber D et al (2008) Electrochemical biosensors-sensor principles and architectures. Sensors 8(3):1400–1458
Fanjul-Bolado P et al (2007) Manufacture and evaluation of carbon nanotube modified screen-printed electrodes as electrochemical tools. Talanta 74(3):427–433
Nag A, Mukhopadhyay SC, Kosel J (2017) Wearable flexible sensors: A review. IEEE Sens J 17(13):3949–3960
Huang C-T et al (2002) Uric acid and urea in human sweat. Chin J Physiol 45(3):109–116
Russell E et al (2014) The detection of cortisol in human sweat: implications for measurement of cortisol in hair. Ther Drug Monit 36(1):30–34
Taylor NA, Machado-Moreira CA (2013) Regional variations in transepidermal water loss, eccrine sweat gland density, sweat secretion rates and electrolyte composition in resting and exercising humans. Extreme Physiol Med 2(1):1–30
Yeung KK et al (2021) Recent advances in electrochemical sensors for wearable sweat monitoring: A review. IEEE Sens J 21(13):14522–14539
Baker LB, Wolfe AS (2020) Physiological mechanisms determining eccrine sweat composition. Eur J Appl Physiol 120:719–752
Cao L et al (2020) A novel 3D paper-based microfluidic electrochemical glucose biosensor based on rGO-TEPA/PB sensitive film. Anal Chim Acta 1096:34–43
Yao Y et al (2021) Integration of interstitial fluid extraction and glucose detection in one device for wearable non-invasive blood glucose sensors. Biosens Bioelectron 179:113078
Moyer J et al (2012) Correlation between sweat glucose and blood glucose in subjects with diabetes. Diabetes Technol Ther 14(5):398–402
Noulas C, Tziouvalekas M, Karyotis T (2018) Zinc in soils, water and food crops. J Trace Elem Med Biol 49:252–260
Kim J et al (2015) Wearable temporary tattoo sensor for real-time trace metal monitoring in human sweat. Electrochem Commun 51:41–45
Cordova A, Alvarez-Mon M (1995) Behaviour of zinc in physical exercise: a special reference to immunity and fatigue. Neurosci Biobehav Rev 19(3):439–445
Cordova A, Navas F (1999) Effect of training on zinc metabolism: changes in serum and sweat zinc concentrations in sportsmen. Occup Health Ind Med 1(40):49
Mondal S, Subramaniam C (2019) Point-of-care, cable-type electrochemical Zn2+ sensor with ultrahigh sensitivity and wide detection range for soil and sweat analysis. ACS Sustain Chem Eng 7(17):14569–14579
Luetkemeier MJ, Coles MG, Askew EW (1997) Dietary sodium and plasma volume levels with exercise. Sports Med 23:279–286
Rosner MH, Kirven J (2007) Exercise-associated hyponatremia. Clin J Am Soc Nephrol 2(1):151–161
Maughan R, Shirreffs S (2010) Dehydration and rehydration in competative sport. Scand J Med Sci Sports 20:40–47
Eichner ER (2008) Genetic and other determinants of sweat sodium. Curr Sports Med Rep 7(4):S36–S40
Parrilla M et al (2016) Wearable potentiometric sensors based on commercial carbon fibres for monitoring sodium in sweat. Electroanalysis 28(6):1267–1275
Coppedè N et al (2020) Ion selective textile organic electrochemical transistor for wearable sweat monitoring. Org Electron 78:105579
He W et al (2019) Integrated textile sensor patch for real-time and multiplex sweat analysis. Sci Adv 5(11):eaax0649
Lu Y et al (2019) Wearable sweat monitoring system with integrated micro-supercapacitors. Nano Energy 58:624–632
Kardalas E et al (2018) Hypokalemia: a clinical update. Endocr Connect 7(4):R135
Valdivielso JM et al (2021) Hyperkalemia in chronic kidney disease in the new era of kidney protection therapies. Drugs 81(13):1467–1489
Mo L et al (2023) Weavable, large-scaled, rapid response, long-term stable electrochemical fabric sensor integrated into clothing for monitoring potassium ions in sweat. Chem Eng J 454:140473
Kim JP et al (2017) Citrate-based fluorescent materials for low-cost chloride sensing in the diagnosis of cystic fibrosis. Chem Sci 8(1):550–558
Farrell PM et al (2001) Early diagnosis of cystic fibrosis through neonatal screening prevents severe malnutrition and improves long-term growth. Pediatrics 107(1):1–13
LeGrys VA (2001) Assessment of sweat-testing practices for the diagnosis of cystic fibrosis. Arch Pathol Lab Med 125(11):1420–1424
Lezana JL et al (2003) Sweat conductivity and chloride titration for cystic fibrosis diagnosis in 3834 subjects. J Cyst Fibros 2(1):1–7
Pagaduan JV et al (2018) Revisiting sweat chloride test results based on recent guidelines for diagnosis of cystic fibrosis. Pract Lab Med 10:34–37
Fellmann N, Fabry R, Coudert J (1989) Calf sweat lactate in peripheral arterial occlusive disease. Am J Physiol-Heart Circ Physiol 257(2):H395–H398
Artiss JD et al (2000) A liquid-stable reagent for lactic acid lsevels: application to the Hitachi 911 and Beckman CX7. Am J Clin Pathol 114(1):139–143
Frost M, Meyerhoff ME (2006) In vivo chemical sensors: tackling biocompatibility. Anal Chem 78(21):7370–7377
Pfeiffer D et al (1997) Amperometric lactate oxidase catheter for real-time lactate monitoring based on thin film technology. Biosens Bioelectron 12(6):539–550
Faude O, Kindermann W, Meyer T (2009) Lactate threshold concepts: how valid are they? Sports Med 39:469–490
Polliack A, Taylor R, Bader D (1997) Sweat analysis following pressure ischaemia in a group of debilitated subjects. J Rehabil Res Dev 34:303–308
Pérez S, Sánchez S, Fabregas E (2012) Enzymatic strategies to construct L-lactate biosensors based on polysulfone/carbon nanotubes membranes. Electroanalysis 24(4):967–974
Piano M et al (2010) Amperometric lactate biosensor for flow injection analysis based on a screen-printed carbon electrode containing Meldola’s Blue-Reinecke salt, coated with lactate dehydrogenase and NAD+. Talanta 82(1):34–37
Zorn JV et al (2017) Cortisol stress reactivity across psychiatric disorders: a systematic review and meta-analysis. Psychoneuroendocrinology 77:25–36
Corbalán-Tutau D et al (2014) Daily profile in two circadian markers “melatonin and cortisol” and associations with metabolic syndrome components. Physiol Behav 123:231–235
Arya SK et al (2012) Recent advances in ZnO nanostructures and thin films for biosensor applications. Anal Chim Acta 737:1–21
Kang N et al (2016) Nanoparticle–nanofibrous membranes as scaffolds for flexible sweat sensors. ACS Sensors 1(8):1060–1069
Sakihara S et al (2010) Evaluation of plasma, salivary, and urinary cortisol levels for diagnosis of Cushing’s syndrome. Endocr J 57(4):331–337
Kazory A (2010) Emergence of blood urea nitrogen as a biomarker of neurohormonal activation in heart failure. Am J Cardiol 106(5):694–700
D’Apolito M et al (2015) Urea-induced ROS cause endothelial dysfunction in chronic renal failure. Atherosclerosis 239(2):393–400
Wang Q, Wen X, Kong J (2020) Recent progress on uric acid detection: a review. Crit Rev Anal Chem 50(4):359–375
Wang Y-X et al (2022) Boron-doped graphene quantum dots anchored to carbon nanotubes as noble metal-free electrocatalysts of uric acid for a wearable sweat sensor. ACS Applied Nano Materials 5(8):11100–11110
Ehtesabi H, Kalji S-O, Movsesian L (2022) Smartphone-based wound dressings: a mini-review. Heliyon 8(7):e09876
Ye X et al (2019) A red emissive two-photon fluorescence probe based on carbon dots for intracellular pH detection. Small 15(48):1901673
Yang P et al (2019) Orange-emissive carbon quantum dots: toward application in wound pH monitoring based on colorimetric and fluorescent changing. Small 15(44):1902823
Laysandra L et al (2021) Synergistic effect in a graphene quantum dot-enabled luminescent skinlike copolymer for long-term pH detection. ACS Appl Mater Interfaces 13(50):60413–60424
Yang W et al (2010) Carbon nanomaterials in biosensors: should you use nanotubes or graphene? Angew Chem Int Ed 49(12):2114–2138
Asadian E, Ghalkhani M, Shahrokhian S (2019) Electrochemical sensing based on carbon nanoparticles: a review. Sens Actuators, B Chem 293:183–209
Ray S et al (2009) Fluorescent carbon nanoparticles: synthesis, characterization, and bioimaging application. J Phys Chem C 113(43):18546–18551
Zhao A et al (2015) Recent advances in bioapplications of C-dots. Carbon 85:309–327
Ehtesabi H, Kalji S-O, Ahadian MM (2023) Carbon nanomaterials in prodrug-based therapeutics. J Drug Deliv Sci Technol 88:104930
Taherpour AA, Mousavi F (2018) Carbon nanomaterials for electroanalysis in pharmaceutical applications. Fullerens, Graphenes and Nanotubes. Elsevier, pp 169–225
Wang HG et al (2017) Flexible electrodes for sodium-ion batteries: recent progress and perspectives. Adv Mater 29(45):1703012
Manickam P et al (2017) Fabric based wearable biosensor for continuous monitoring of steroids. ECS Trans 77(11):1841
Allen JA, Murugesan D, Viswanathan C (2019) Circumferential growth of zinc oxide nanostructure anchored over carbon fabric and its photocatalytic performance towards p-nitrophenol. Superlattice Microstruct 125:159–167
Georgakilas V et al (2015) Broad family of carbon nanoallotropes: classification, chemistry, and applications of fullerenes, carbon dots, nanotubes, graphene, nanodiamonds, and combined superstructures. Chem Rev 115(11):4744–4822
Ming H et al (2012) Large scale electrochemical synthesis of high quality carbon nanodots and their photocatalytic property. Dalton Trans 41(31):9526–9531
Huo F et al (2020) Preparation and biomedical applications of multicolor carbon dots: recent advances and future challenges. Part Part Syst Charact 37(4):1900489
Hola K et al (2014) Carbon dots—Emerging light emitters for bioimaging, cancer therapy and optoelectronics. Nano Today 9(5):590–603
Bao X et al (2018) In vivo theranostics with near-infrared-emitting carbon dots—highly efficient photothermal therapy based on passive targeting after intravenous administration. Light: Sci Appl 7(1):91
Ehtesabi H (2020) Carbon nanomaterials for salivary-based biosensors: a review. Mater Today Chem 17:100342
Ehtesabi H (2020) Application of carbon nanomaterials in human virus detection. J Sci: Adv Mater Devices 5(4):436–450
Roushani M et al (2017) Application of graphene quantum dots as green homogenous nanophotocatalyst in the visible-light-driven photolytic process. J Mater Sci: Mater Electron 28:5135–5143
Roushani M, Mavaei M, Rajabi HR (2015) Graphene quantum dots as novel and green nano-materials for the visible-light-driven photocatalytic degradation of cationic dye. J Mol Catal A: Chem 409:102–109
Fakhraie S et al (2021) In situ simultaneous chemical activation and exfoliation of carbon quantum dots for atmospheric adsorption of H2S and CO2 at room temperature. Appl Surf Sci 559:149892
Hu C et al (2019) Design and fabrication of carbon dots for energy conversion and storage. Chem Soc Rev 48(8):2315–2337
Kipnusu WK et al (2020) Nonlinear optics to glucose sensing: multifunctional nitrogen and boron doped carbon dots with solid-state fluorescence in nanoporous silica films. Part Part Syst Charact 37(6):2000093
Rezaei A, Ehtesabi H (2022) Fabrication of alginate/chitosan nanocomposite sponges using green synthesized carbon dots as potential wound dressing. Mater Today Chem 24:100910
Ehtesabi H, Nasri R (2021) Carbon dot-based materials for wound healing applications. Adv Natl Sci: Nanosci Nanotechnol 12(2):025006
Shakiba-Marani R, Ehtesabi H (2023) A flexible and hemostatic chitosan, polyvinyl alcohol, carbon dot nanocomposite sponge for wound dressing application. Int J Biol Macromol 224:831–839
Bariya M et al (2018) Roll-to-roll gravure printed electrochemical sensors for wearable and medical devices. ACS Nano 12(7):6978–6987
Kurniawan D, Chiang W-H (2020) Microplasma-enabled colloidal nitrogen-doped graphene quantum dots for broad-range fluorescent pH sensors. Carbon 167:675–684
Ehtesabi H et al (2021) Smartphone-based portable device for rapid and sensitive pH detection by fluorescent carbon dots. Sens Actuators, A 332:113057
Azad LM, Ehtesabi H, Rezaei A (2021) Smartphone-based fluorometer for pH detection using green synthesized carbon dots. Nano-Struct Nano-Objects 26:100722
Hu Y et al (2017) Green preparation of S and N Co-doped carbon dots from water chestnut and onion as well as their use as an off–on fluorescent probe for the quantification and imaging of coenzyme A. ACS Sustain Chem Eng 5(6):4992–5000
Li L-S, Xu L (2020) Highly fluorescent N, S, P tri-doped carbon dots for Cl− detection and their assistance of TiO2 as the catalyst in the degradation of methylene blue. J Photochem Photobiol, A 401:112772
Iijima S (1991) Helical microtubules of graphitic carbon. Nature 354(6348):56–58
Robertson J (2004) Realistic applications of CNTs. Mater Today 7(10):46–52
Sherigara BS, Kutner W, D’Souza F (2003) Electrocatalytic properties and sensor applications of fullerenes and carbon nanotubes. Electroanalysis: Int J Devoted Fundam Pract Aspects Electroanalysis 15(9):753–772
Xing Z et al (2018) Highly flexible printed carbon nanotube thin film transistors using cross-linked poly (4-vinylphenol) as the gate dielectric and application for photosenstive light-emitting diode circuit. Carbon 133:390–397
Javey A et al (2003) Ballistic carbon nanotube field-effect transistors. Nature 424(6949):654–657
Besteman K et al (2003) Enzyme-coated carbon nanotubes as single-molecule biosensors. Nano Lett 3(6):727–730
Geier ML et al (2015) Solution-processed carbon nanotube thin-film complementary static random access memory. Nat Nanotechnol 10(11):944–948
Maji D et al (2015) Buckling assisted and lithographically micropatterned fully flexible sensors for conformal integration applications. Sci Rep 5(1):17776
Liu Q et al (2021) Preparation of nanostructured PDMS film as flexible immunosensor for cortisol analysis in human sweat. Anal Chim Acta 1184:339010
Claussen JC et al (2009) Electrochemical biosensor of nanocube-augmented carbon nanotube networks. ACS Nano 3(1):37–44
Park S-C et al (2021) Carbon nanotube-based ion-sensitive field-effect transistors with an on-chip reference electrode toward wearable sodium sensing. ACS Appl Electron Mater 3(6):2580–2588
Bollella P, Gorton L (2018) Enzyme based amperometric biosensors. Curr Opin Electrochem 10:157–173
Kim J et al (2018) Wearable bioelectronics: Enzyme-based body-worn electronic devices. Acc Chem Res 51(11):2820–2828
Milton RD, Minteer SD (2017) Direct enzymatic bioelectrocatalysis: differentiating between myth and reality. J R Soc Interface 14(131):20170253
Monteiro T, Almeida MG (2019) Electrochemical enzyme biosensors revisited: Old solutions for new problems. Crit Rev Anal Chem 49(1):44–66
Prévoteau A, Mano N (2012) Oxygen reduction on redox mediators may affect glucose biosensors based on “wired” enzymes. Electrochim Acta 68:128–133
Ito K et al (2019) Designer fungus FAD glucose dehydrogenase capable of direct electron transfer. Biosens Bioelectron 123:114–123
Mazurenko I et al (2018) Pore size effect of MgO-templated carbon on enzymatic H2 oxidation by the hyperthermophilic hydrogenase from Aquifex aeolicus. J Electroanal Chem 812:221–226
Sakai K et al (2018) Assembly of direct-electron-transfer-type bioelectrodes with high performance. Electrochim Acta 271:305–311
Xia H-Q et al (2020) Mediator-free electron-transfer on patternable hierarchical meso/macro porous bienzyme interface for highly-sensitive sweat glucose and surface electromyography monitoring. Sens Actuators, B Chem 312:127962
Hao J et al (2022) Photosensitive-stamp-inspired scalable fabrication strategy of wearable sensing arrays for noninvasive real-time sweat analysis. Anal Chem 94(10):4547–4555
Oh SY et al (2018) Skin-attachable, stretchable electrochemical sweat sensor for glucose and pH detection. ACS Appl Mater Interfaces 10(16):13729–13740
Kalji O et al (2020) Colloidal graphene oxide enhances the activity of a lipase and protects it from oxidative damage: insights from physicochemical and molecular dynamics investigations. J Colloid Interface Sci 567:285–299
Novoselov KS et al (2004) Electric field effect in atomically thin carbon films. Science 306(5696):666–669
Li W et al (2011) Electrochemistry of individual monolayer graphene sheets. ACS Nano 5(3):2264–2270
Fenzl C et al (2017) Laser-scribed graphene electrodes for aptamer-based biosensing. Acs Sensors 2(5):616–620
Mao C et al (2021) Separating boundary potential changes at thin solid contact ion transfer voltammetric membrane electrodes. J Electroanal Chem 880:114800
Zdrachek E, Bakker E (2021) Ion-to-electron capacitance of single-walled carbon nanotube layers before and after ion-selective membrane deposition. Microchim Acta 188:1–10
Lyu Y et al (2020) Solid-contact ion-selective electrodes: response mechanisms, transducer materials and wearable sensors. Membranes 10(6):128
Yeung KK et al (2022) Utilizing gradient porous graphene substrate as the solid-contact layer to enhance wearable electrochemical sweat sensor sensitivity. Nano Lett 22(16):6647–6654
Prabhakaran A, Nayak P (2019) Surface engineering of laser-scribed graphene sensor enables non-enzymatic glucose detection in human body fluids. ACS Appl Nano Mater 3(1):391–398
Sharma S et al (2020) Laser induced flexible graphene electrodes for electrochemical sensing of hydrazine. Mater Lett 262:127150
Marques AC et al (2020) Laser-induced graphene-based platforms for dual biorecognition of molecules. ACS Appl Nano Mater 3(3):2795–2803
Choudhury S et al (2021) Potentiometric ion-selective sensors based on UV-ozone irradiated laser-induced graphene electrode. Electrochim Acta 387:138341
Khan A, Winder M, Hossain G (2022) Modified graphene-based nanocomposite material for smart textile biosensor to detect lactate from human sweat. Biosens Bioelectron: X 10:100103
Xu M et al (2021) Reduced graphene oxide-coated silica nanospheres as flexible enzymatic biosensors for detection of glucose in sweat. ACS Appl Nano Mater 4(11):12442–12452
Lu Q et al (2021) Flexible paper-based Ni-MOF composite/AuNPs/CNTs film electrode for HIV DNA detection. Biosens Bioelectron 184:113229
Shu Y et al (2021) Highly stretchable wearable electrochemical sensor based on Ni-Co MOF nanosheet-decorated Ag/rGO/PU fiber for continuous sweat glucose detection. Anal Chem 93(48):16222–16230
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Ehtesabi, H., Kalji, SO. Carbon nanomaterials for sweat-based sensors: a review. Microchim Acta 191, 77 (2024). https://doi.org/10.1007/s00604-023-06162-7
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00604-023-06162-7