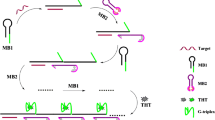
Abstract
A dual-targeting nanobiosensor has been developed for the simultaneous detection of AMELX and AMELY genes based on the different fluorescence signals emitted from gold and silver nanoclusters, AuNCs and AgNCs respectively. In our design, both catalytic hairpin assembly (CHA) and hybridization chain reaction (HCR) have been used as isothermal, enzyme-free and simple methods for signal’s amplification. The working principle is based on the initiation of a cascade of CHA-HCR reactions when AMELX is present, in which AuNCs, synthesized on the third hairpin, are aggregated on the surface of the dsDNA product, performing the phenomenon of aggregation induced emission (AIE) and enhancing their fluorescence signal. On the other hand, the presence of the second target, AMELY, is responsible for the enhancement of the fluorescence signal corresponding to AgNCs by the same phenomenon, via hybridizing to the free end of the dsDNA formed and at the same time to the probe of silver nanoclusters fixing it closer to the surface of the dsDNA product. Such a unique design has the merits of being simple, inexpensive, specific and stable and presents rapid results. The detection limits of this assay for AMELX and AMELY are as low as 3.16 fM and 23.6 fM respectively. Moreover, this platform showed great performance in real samples. The design has great promise for the application of dual-targeting nanobiosensors to other biomarkers.
Graphical Abstract







Similar content being viewed by others
Data Availability
The authors confirm that the data supporting the findings of this study are available within the article and its supplementary materials.
References
Clark LC Jr, Lyons C (1968) Electrode systems for continuous monitoring in cardiovascular surgery. Ann New York Acad Sci 102(1):29–45. https://doi.org/10.1111/j.1749-6632.1962.tb13623.x
Chen X, Yao C, Li Z (2023) Microarray-based chemical sensors and biosensors: Fundamentals and food safety applications. TrAC Trends in Analytical Chem 158:116785
Hassan RYA (2022) Advances in Electrochemical Nano-Biosensors for Biomedical and Environmental Applications: From Current Work to Future Perspectives. Sensors 22(19):7539. https://doi.org/10.3390/s22197539
Kang S, Zhao K, Yu D-G, Zheng X, Huang C (2022) Advances in Biosensing and Environmental Monitoring Based on Electrospun Nanofibers. Adv Fiber Mater 4(3):404–435
Bazzi F, Ebrahimi-Hoseinzadeh B, Sangachin EA, Hosseini M (2023) The integration of hybridization chain reaction (HCR) with fluorogenic silver nanoclusters (AgNCs) in an aggregation induced emission (AIE)-based nanosensor for sex determination and its forensic application. Microchemical Journal 185:108188
Saiki RK et al (1985) Enzymatic Amplification of β-Globin Genomic Sequences and Restriction Site Analysis for Diagnosis of Sickle Cell Anemia,". Science 230(4732):1350–1354
Banér J, Nilsson M, Mendel-Hartvig M, Landegren U (1998) Signal amplification of padlock probes by rolling circle replication. Nucleic Acids Res 26(22):5073–5078
Piepenburg O, Williams CH, Stemple DL, Armes NA (2006) DNA Detection Using Recombination Proteins. PLoS Biol 4(7):e204
Dirks RM, Pierce NA (2004) Triggered amplification by hybridization chain reaction. Proc Nat Acad Sci 101(43):15275–15278
Yin P, Choi HMT, Calvert CR, Pierce NA (2008) Programming biomolecular self-assembly pathways. Nature 451(7176):318–322
Chen P et al (2019) Multimode MicroRNA Sensing via Multiple Enzyme-Free Signal Amplification and Cation-Exchange Reaction. ACS Appl Mater Interfaces 11(40):36476–36484
Tao Y, Li M, Ren J, Qu X (2015) Metal nanoclusters: novel probes for diagnostic and therapeutic applications. Chem Soc Rev 44(23):8636–8663. https://doi.org/10.1039/C5CS00607D
Huang H, Li H, Feng J-J, Wang A-J (2016) One-step green synthesis of fluorescent bimetallic Au/Ag nanoclusters for temperature sensing and in vitro detection of Fe3+,. Sensors and Actuators B: Chemical 223:550–556
Li H, Chen J, Huang H, Feng J-J, Wang A-J, Shao L-X (2016) Green and facile synthesis of l-carnosine protected fluorescent gold nanoclusters for cellular imaging. Sensors and Actuators B: Chemical 223:40–44
Hosseini M, Ahmadi E, Borghei Y-S, Reza Ganjali M (2017) A new fluorescence turn-on nanobiosensor for the detection of micro-RNA-21 based on a DNA–gold nanocluster. Methods App Fluoresc 5(1):015005
Nakahori Y, Takenaka O, Nakagome Y (1991) A human X-Y homologous region encodes “amelogenin.” Genomics 9(2):264–269
Wang C et al (2015) Elaborately designed diblock nanoprobes for simultaneous multicolor detection of microRNAs. Nanoscale 7(38):15822–15829. https://doi.org/10.1039/C5NR04618A
Ma Q et al (2023) Target controlled alternative hybridization chain reaction for fluorescent detection of dual mycotoxins. Analytica Chimica Acta 1237:340595
Fu P, Xing S, Xu M, Zhao Y, Zhao C (2020) Peptide nucleic acid-based electrochemical biosensor for simultaneous detection of multiple microRNAs from cancer cells with catalytic hairpin assembly amplification. Sensors and Actuators B: Chemical 305:127545
Xue Y, Wang Y, Feng S, Yan M, Huang J, Yang X (2022) A dual-amplification mode and Cu-based metal-organic frameworks mediated electrochemical biosensor for sensitive detection of microRNA. Biosensors and Bioelectronics 202:113992
Li T et al (2018) One-Step Synthesis of DNA Templated Water-Soluble Au–Ag Bimetallic Nanoclusters for Ratiometric Fluorescence Detection of DNA. J Biomed Nanotechnol 14(1):150–160
Fan C, Li Q (2019) Advances in DNA Nanotechnology. Small 15(26):1902586. https://doi.org/10.1002/smll.201902586
Yeh H-C, Sharma J, Han JJ, Martinez JS, Werner JH (2010) A DNA−Silver Nanocluster Probe That Fluoresces upon Hybridization. Nano Letters 10(8):3106–3110
Wang H-B, Bai H-Y, Dong G-L, Liu Y-M (2019) DNA-templated Au nanoclusters coupled with proximity-dependent hybridization and guanine-rich DNA induced quenching: a sensitive fluorescent biosensing platform for DNA detection. Nanoscale Advances 1(4):1482–1488. https://doi.org/10.1039/C8NA00278A
Wang J, Lin X, Shu T, Su L, Liang F, Zhang X (2019) Self-Assembly of Metal Nanoclusters for Aggregation-Induced Emission. Int J Mol Sci 20(8):1891. https://doi.org/10.3390/ijms20081891
Shiang Y-C, Huang C-C, Chen W-Y, Chen P-C, Chang H-T (2012) Fluorescent gold and silver nanoclusters for the analysis of biopolymers and cell imaging. J Mater Chem 22(26):12972–12982. https://doi.org/10.1039/C2JM30563A
Bautista-Sánchez D et al (2020) The Promising Role of miR-21 as a Cancer Biomarker and Its Importance in RNA-Based Therapeutics. Mol Therapy - Nucleic Acids 20:409–420
Cao X et al (2021) Rapid and sensitive detection of dual lung cancer-associated miRNA biomarkers by a novel SERS-LFA strip coupling with catalytic hairpin assembly signal amplification. J Mater Chem C 9(10):3661–3671. https://doi.org/10.1039/D0TC05737A
Zhang Y et al (2020) Label-Free Analysis of H5N1 Virus Based on Three-Segment Branched DNA-Templated Fluorescent Silver Nanoclusters. ACS Appl Mater Interfaces 12(43):48357–48362
Li Z-Y, Wu Y-T, Tseng W-L (2015) UV-Light-Induced Improvement of Fluorescence Quantum Yield of DNA-Templated Gold Nanoclusters: Application to Ratiometric Fluorescent Sensing of Nucleic Acids. ACS Appl Mater Interfaces 7(42):23708–23716
Li H et al (2021) Intracellular CircRNA imaging and signal amplification strategy based on the graphene oxide-DNA system. Analytica Chimica Acta 1183:338966
Huang J, Ye L, Gao X, Li H, Xu J, Li Z (2015) Molybdenum disulfide-based amplified fluorescence DNA detection using hybridization chain reactions. J Mater Chem B 3(11):2395–2401. https://doi.org/10.1039/C4TB01986E
Hao N, Dai P-P, Yu T, Xu J-J, Chen H-Y (2015) A dual target-recycling amplification strategy for sensitive detection of microRNAs based on duplex-specific nuclease and catalytic hairpin assembly. Chem Commun 51(70):13504–13507. https://doi.org/10.1039/C5CC05350A
Cai W, Xie S, Zhang J, Tang D, Tang Y (2017) An electrochemical impedance biosensor for Hg2+ detection based on DNA hydrogel by coupling with DNAzyme-assisted target recycling and hybridization chain reaction. Biosensors and Bioelectronics 98:466–472
Zhang W, Hao W, Liu X, Sun X, Yan J, Wang Y (2020) Visual detection of miRNAs using enzyme-free amplification reactions and ratiometric fluorescent probes. Talanta 219:121332
Acknowledgements
Gratefully, this study was financially supported by the research Council of University of Tehran.
Author information
Authors and Affiliations
Contributions
Fatima Bazzi: Formal analysis, visualization, data curation, writing-original, draft, review and editing., Morteza Hosseini: Conceptualization, methodology, designing the analysis, visualization, writing-review and editing. Bahman Ebrahimi-Hoseinzadeh: Project administration, visualization, designing the analysis, Haider A.J. Al Lawati: writing-review and editing, Project consultant and Mohammad Reza Ganjali: Writing-review and editing.
Corresponding authors
Ethics declarations
Conflicts of interest
The authors have no financial or non-financial conflicts of interest to disclose.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
AMELX, Amelogenin gene X; AMELY, Amelogenin gene Y; AuNCs, Gold nanoclusters; AgNCs, silver nanoclusters; CHA, catalytic hairpin assembly; HCR, hybridization chain reaction; AIE, aggregation induced emission.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Bazzi, F., Hosseini, M., Ebrahimi-Hoseinzadeh, B. et al. A dual-targeting nanobiosensor for Gender Determination applying Signal Amplification Methods and integrating Fluorometric Gold and Silver Nanoclusters. Microchim Acta 190, 368 (2023). https://doi.org/10.1007/s00604-023-05947-0
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00604-023-05947-0