Abstract
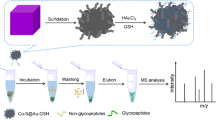
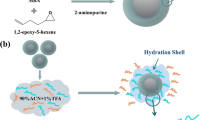
A new polymeric (methyl methacrylate/ethylene glycol dimethacrylate/1,2-epoxy-5-hexene) base/matrix has been fabricated and decorated with zwitterionic hydrophilic cysteic acid (Cya) for the enrichment of intact N-glycopeptides from standards and biological samples. Terpolymer-Cya provides good enrichment efficiency, improved hydrophilicity, and selectivity by virtue of better surface area (2.09 × 102 m2/g) provided by terpolymer and the zwitterionic property offered by cysteic acid. Cysteic acid-functionalized polymeric hydrophilic interaction liquid chromatography (HILIC) sorbent enriches 35 and 24 N-linked glycopeptides via SPE (solid phase extraction) mode from tryptic digests of model glycoproteins, i.e., immunoglobulin G (IgG) and horseradish peroxidase (HRP), respectively. Zwitterionic chemistry of cysteine helps in achieving higher selectivity with BSA digest (1:200), and lower detection limit down to 100 attomoles with a complete glycosylation profile of each standard digest. The recovery of 81% and good reproducibility define the application of terpolymer-Cya for complex samples like a serum. Analysis of human serum provides a profile of 807 intact N-linked glycopeptides via nano-liquid chromatography-tandem mass spectrometry (nLC-MS/MS). To the best of our knowledge, this is the highest number of glycopeptides enriched by any HILIC sorbent. Selected glycoproteins are evaluated in link to various cancers including the breast, lung, uterine, and melanoma using single-nucleotide variances (BioMuta). This study represents the complete idea of using an in-house developed strategy as a successful tool to help analyze, relate, and answer glycoprotein-based clinical issues regarding cancers.
Graphical abstract






Similar content being viewed by others
Data availability
Data is available via the Proteome Xchange with identifier PXD030890.
References
Gilgunn S, Conroy PJ, Saldova R, Rudd PM, O’kennedy RJ (2013) Aberrant PSA glycosylation—a sweet predictor of prostate cancer. Nat Rev Urol 10(2):99–107. https://doi.org/10.1038/nrurol.2012.258
Ohtsubo K, Chen MZ, Olefsky JM, Marth JD (2011) Pathway to diabetes through attenuation of pancreatic beta cell glycosylation and glucose transport. Nat Med 17(9):1067–1075. https://doi.org/10.1038/nm.2414
Wang AC, Jensen EH, Rexach JE, Vinters HV, Hsieh-Wilson LC (2016) Loss of O-GlcNAc glycosylation in forebrain excitatory neurons induces neurodegeneration. Proc Nat Acad Sci 113(52):15120–15125. https://doi.org/10.1073/pnas.1606899113
Hafkenscheid L, Bondt A, Scherer HU, Huizinga TW, Wuhrer M, Toes RE, Rombouts Y (2017) Structural analysis of variable domain glycosylation of anti-citrullinated protein antibodies in rheumatoid arthritis reveals the presence of highly sialylated glycans. Mol Cell Proteom 16(2):278–287. https://doi.org/10.1074/mcp.M116.062919
Ghesquière B, Van Damme J, Martens L, Vandekerckhove J, Gevaert K (2006) Proteome-wide characterization of N-glycosylation events by diagonal chromatography. J Proteome Res 5(9):2438–2447. https://doi.org/10.1021/pr060186m
Ruhaak LR, Xu G, Li Q, Goonatilleke E, Lebrilla CB (2018) Mass spectrometry approaches to glycomic and glycoproteomic analyses. Chem Rev 118(17):7886–7930. https://doi.org/10.1021/acs.chemrev.7b00732
Xiao H, Suttapitugsakul S, Sun F, Wu R (2018) Mass spectrometry-based chemical and enzymatic methods for global analysis of protein glycosylation. Acc Chem Res 51(8):1796–1806. https://doi.org/10.1021/acs.accounts.8b00200
Liu Q, Deng CH, Sun N (2018) Hydrophilic tripeptide-functionalized magnetic metal–organic frameworks for the highly efficient enrichment of N-linked glycopeptides. Nanoscale 10(25):12149–12155. https://doi.org/10.1039/C8NR03174F
Kailasa SK, D’souza S, Wu HF (2014) Analytical applications of nanoparticles in MALDI-MS for bioanalysis. Bioanalysis 7(17):2265–2276. https://doi.org/10.4155/bio.15.149
Kailasa S, Wu HF (2014) Recent developments in nanoparticle-based MALDI mass spectrometric analysis of phosphoproteomes. Microchim Acta 181:853–864. https://doi.org/10.1007/s00604-014-1191-z
Kailasa S, Koduru JK, Baek SH, Wu HF, Hussain CM, Park TJ (2020) Review on matrix-assisted laser desorption/ionization time-of-flight mass spectrometry for the rapid screening of microbial species: a promising bioanalytical tool. Microchem J 159:105387. https://doi.org/10.1016/j.microc.2020.105387
Liu L, Yu M, Zhang Y, Wang C, Lu H (2014) Hydrazide functionalized core–shell magnetic nanocomposites for highly specific enrichment of N-glycopeptides. ACS Appl Mater Interfaces 6(10):7823–7832. https://doi.org/10.1021/am501110e
Boersema PJ, Mohammed S, Heck AJ (2008) Hydrophilic interaction liquid chromatography (HILIC) in proteomics. Anal Bioanal Chem 391(1):151–159. https://doi.org/10.1007/s00216-008-1865-7
Tang J, Liu Y, Qi D, Yao G, Deng C, Zhang X (2009) On-plate-selective enrichment of glycopeptides using boronic acid-modified gold nanoparticles for direct MALDI-QIT-TOF MS analysis. Proteomics 9(22):5046–5055. https://doi.org/10.1002/pmic.200900033
Madera M, Mechref Y, Novotny MV (2005) Combining lectin microcolumns with high-resolution separation techniques for enrichment of glycoproteins and glycopeptides. Anal Chem 77(13):4081–4090. https://doi.org/10.1021/ac050222l
Dong L, Feng S, Li S, Song P, Wang J (2015) Preparation of concanavalin A-chelating magnetic nanoparticles for selective enrichment of glycoproteins. Anal Chem 87(13):6849–6853. https://doi.org/10.1021/acs.analchem.5b01184
Liang Y, Wu C, Zhao Q, Wu Q, Jiang B, Weng Y, Liang Z, Zhang L, Zhang Y (2015) Gold nanoparticles immobilized hydrophilic monoliths with variable functional modification for highly selective enrichment and on-line deglycosylation of glycopeptides. Anal Chim Acta 900:83–89. https://doi.org/10.1016/j.aca.2015.10.024
Qu Y, Sun L, Zhang Z, Dovichi NJ (2018) Site-specific glycan heterogeneity characterization by hydrophilic interaction liquid chromatography solid-phase extraction, reversed-phase liquid chromatography fractionation, and capillary zone electrophoresis-electrospray ionization-tandem mass spectrometry. Anal Chem 90(2):1223–1233. https://doi.org/10.1021/acs.analchem.7b03912
Saleem S, Sajid MS, Hussain D, Jabeen F, Najam-ul-Haq M, Saeed A (2020) Boronic acid functionalized MOFs as HILIC material for N-linked glycopeptide enrichment. Anal Bioanal Chem 412(7):1509–1520. https://doi.org/10.1007/s00216-020-02427-9
Zhang YW, Li Z, Zhao Q, Zhou YL, Liu HW, Zhang XX (2014) A facilely synthesized amino-functionalized metal–organic framework for highly specific and efficient enrichment of glycopeptides. Chem Commun 50(78):11504–11516. https://doi.org/10.1039/C4CC05179C
Qing G, Yan J, He X, Li X, Liang X (2020) Recent advances in hydrophilic interaction liquid interaction chromatography materials for glycopeptide enrichment and glycan separation. TrAC Trends Anal Chem 124:115570. https://doi.org/10.1016/j.trac.2019.06.020
Liu L, Zhang Y, Zhang L, Yan G, Yao J, Yang P, Lu H (2012) Highly specific revelation of rat serum glycopeptidome by boronic acid-functionalized mesoporous silica. Anal Chim Acta 753:64–72. https://doi.org/10.1016/j.aca.2012.10.002
Yu L, Li X, Guo Z, Zhang X, Liang X (2009) Hydrophilic interaction chromatography-based enrichment of glycopeptides by using click maltose: a matrix with high selectivity and glycosylation heterogeneity coverage. Chem Eur J 15(46):12618–12626. https://doi.org/10.1002/chem.200902370
Xiong Z, Qin H, Wan H, Huang G, Zhang Z, Dong J, Zhang L, Zhang W, Zou H (2013) Layer-by-layer assembly of multilayer polysaccharide coated magnetic nanoparticles for the selective enrichment of glycopeptides. Chem Commun 49(81):9284–9296. https://doi.org/10.1039/C3CC45008B
Bai H, Zhang B, Cheng X, Liu J, Wang X, Qin W, Zhang M (2022) Synthesis of zwitterionic polymer modified graphene oxide for hydrophilic enrichment of N-glycopeptides from urine of healthy subjects and patients with lung adenocarcinoma. Talanta 237:122938. https://doi.org/10.1016/j.talanta.2021.122938
Ma W, Xu L, Li X, Shen S, Wu M, Bai Y, Liu H (2017) Cysteine-functionalized metal–organic framework: facile synthesis and high efficient enrichment of n-linked glycopeptides in cell lysate. ACS Appl Mater Interfaces 9:19562–19568. https://doi.org/10.1021/acsami.7b02853
He Y, Zheng Q, Huang H, Ji Y, Lin Z (2022) Synergistic synthesis of hydrophilic hollow zirconium organic frameworks for simultaneous recognition and capture of phosphorylated and glycosylated peptides. Anal Chim Acta 1198:339552. https://doi.org/10.1016/j.aca.2022.339552
Sajid MS, Jovcevski B, Pukala TL, Jabeen F, Najam-ul-Haq M (2019) Fabrication of piperazine functionalized polymeric monolithic tip for rapid enrichment of glycopeptides/glycans. Anal Chem 92(1):683–689. https://doi.org/10.1021/acs.analchem.9b02068
Shaipulizan NS, Md Jamil SN, Kamaruzaman S, Subri NN, Adeyi AA, Abdullah AH, Abdullah LC (2020) Preparation of ethylene glycol dimethacrylate (EGDMA)-based terpolymer as potential sorbents for pharmaceuticals adsorption. Polymers 12(2):423. https://doi.org/10.3390/polym12020423
Demirci S, Khiev D, Can M, Sahiner M, Biswal MR, Ayyala RS, Sahiner N (2021) Chemically cross-linked poly (β-cyclodextrin) particles as promising drug delivery materials. ACS Appl Poly Mater 3:6238–6251. https://doi.org/10.1021/acsapm.1c01058
Podlesnyuk VV, Hradil J, Marutovskii RM, Klimenko NA, Fridman LE (1997) Sorption of organic compounds from aqueous solutions by glycidyl methacrylate-styrene-ethylene dimethacrylate terpolymers. React Funct Polym 33(2–3):275–288. https://doi.org/10.1016/S1381-5148(97)00065-5
Miletic N, Vukovic Z, Nastasovic A, Loos K (2009) Macroporous poly(glycidyl methacrylate-co-ethylene glycol dimethacrylate) resins—versatile immobilization supports for biocatalysts. J Mol Catal B 56:196–201. https://doi.org/10.1016/j.molcatb.2008.04.012
Qu JB, Lin YY, Liu Y, Zhu BQ, Sun YJ, Peng WS, Li J (2021) Synthesis of bimodal open-porous polystyrene monoliths with glycopolymer surfaces for high-speed protein chromatography. ACS Appl Polym Mater 3:2657–2666. https://doi.org/10.1021/acsapm.1c00232
Mompo RO, Vergara BM, Lerma GJ, Simó EF, Herrero M (2021) Boronate affinity sorbents based on thiol-functionalized polysiloxane-polymethacrylate composite materials in syringe format for selective extraction of glycopeptides. Microchem J 164:106018. https://doi.org/10.1016/j.microc.2021.106018
Furukawa H, Ko N, Go YB, Aratani N, Choi SB, Choi E, Yazaydin AÖ, Snurr RQ, O’Keeffe M, Kim J, Yaghi OM (2010) Ultrahigh porosity in metal-organic frameworks. Science 329(5990):424–428. https://doi.org/10.1126/science.1192160
Diggle B, Jiang Z, Li RW, Connal LA (2021) Self-healing polymer network with high strength, tunable properties, and biocompatibility. Chem Mater 33:3712–3720. https://doi.org/10.1021/acs.chemmater.1c00707
Ma YF, Yuan F, Zhang XH, Zhou YL, Zhang XX (2017) Highly efficient enrichment of N-linked glycopeptides using a hydrophilic covalent-organic framework. Analyst 142(17):3212–3218. https://doi.org/10.1039/C7AN01027C
Zhang W, Jiang L, Fu L, Jia Q (2019) Selective enrichment of glycopeptides based on copper tetra (N-carbonylacrylic) aminephthalocyanine and iminodiacetic acid functionalized polymer monolith. J Sep Sci 42(5):1037–1044. https://doi.org/10.1002/jssc.201801030
Bi C, Liang Y, Shen L, Tian S, Zhang K, Li Y, He X, Chen L, Zhang Y (2018) Maltose-functionalized hydrophilic magnetic nanoparticles with polymer brushes for highly selective enrichment of N-linked glycopeptides. ACS Omega 3(2):1572–1580. https://doi.org/10.1021/acsomega.7b01788
Ying LL, Wang DY, Yang HP, Deng XY, Peng C, Zheng C, Xu B, Dong LY, Wang X, Xu L, Zhang YW (2018) Synthesis of boronate-decorated polyethyleneimine-grafted porous layer open tubular capillaries for enrichment of polyphenols in fruit juices. J Chromatogr A 1544:23–32. https://doi.org/10.1016/j.chroma.2018.02.044
Wang YF, Jin HX, Wang YG, Yang LY, OuYang XK, Wu WJ (2016) Synthesis and characterization of magnetic molecularly imprinted polymer for the enrichment of ofloxacin enantiomers in fish samples. Molecules 21(7):915. https://doi.org/10.3390/molecules21070915
Zou X, Zhong L, Liu D, Yang B, Lou Y, Peng J, Rainer M, Feuerstein I, Muhammad NUH, Huck CW, Bonn GK (2011) Novel multifunctional chitosan-GMA-IDA-Cu (II) nanospheres for high dynamic range characterization of the human plasma proteome. Anal Bioanal Chem 400(3):747–756. https://doi.org/10.1007/s00216-011-4812-y
Zhao Q, Fang F, Wu C, Wu Q, Liang Y, Liang Z, Zhang L, Zhang Y (2016) imFASP: An integrated approach combining in-situ filter-aided sample pretreatment with microwave-assisted protein digestion for fast and efficient proteome sample preparation. Anal Chim Acta 912:58–64. https://doi.org/10.1016/j.aca.2016.01.049
Peng J, Hu Y, Zhang H, Wan L, Wang L, Liang Z, Zhang L, Wu RA (2019) High anti-interfering profiling of endogenous glycopeptides for human plasma by the dual-hydrophilic metal–organic framework. Anal Chem 91(7):4852–4859. https://doi.org/10.1021/acs.analchem.9b00542
Cao Z, Jiang S (2012) Super-hydrophilic zwitterionic poly (carboxybetaine) and amphiphilic non-ionic poly (ethylene glycol) for stealth nanoparticles. Nano Today 7(5):404–413. https://doi.org/10.1016/j.nantod.2012.08.001
Sun N, Wu H, Chen H, Shen X, Deng C (2019) Advances in hydrophilic nanomaterials for glycoproteomics. Chem Commun 55(70):10359–10375. https://doi.org/10.1039/C9CC04124A
Roy R, Ang E, Komatsu E, Domalaon R, Bosseboeuf A, Harb J, Hermouet S, Krokhin O, Schweizer F, Perreault H (2018) Absolute quantitation of glycoforms of two human IgG subclasses using synthetic fc peptides and glycopeptides. J Am Soc Mass Spectrom 29(6):1086–1098. https://doi.org/10.1007/s13361-018-1900-7
Krokhin O, Ens W, Standing KG, Wilkins J, Perreault H (2004) Site-specific N-glycosylation analysis: matrix-assisted laser desorption/ionization quadrupole-quadrupole time-of-flight tandem mass spectral signatures for recognition and identification of glycopeptides. Rapid Commun Mass Spectrom 18(18):2020–2030. https://doi.org/10.1002/rcm.1585
Wang H, Jiao F, Gao F, Huang J, Zhao Y, Shen Y, Zhang Y, Qian X (2017) Facile synthesis of magnetic covalent organic frameworks for the hydrophilic enrichment of N-glycopeptides. J Mater Chem B 5(22):4052–4059. https://doi.org/10.1039/C7TB00700K
Ji Y, Xiong Z, Huang G, Liu J, Zhang Z, Liu Z, Ou J, Ye M, Zou H (2014) Efficient enrichment of glycopeptides using metal–organic frameworks by hydrophilic interaction chromatography. Analyst 139(19):4987–4993. https://doi.org/10.1039/C4AN00971A
Li Y, Wang H, You X, Ma S, Dong J, Wei Y, Ou J, Ye M (2018) Facile preparation of microporous organic polymers functionalized macroporous hydrophilic resin for selective enrichment of glycopeptides. Anal Chim Acta 1030:96–104. https://doi.org/10.1016/j.aca.2018.05.040
Jiang B, Wu Q, Zhang L, Zhang Y (2017) Preparation and application of silver nanoparticle-functionalized magnetic graphene oxide nanocomposites. Nanoscale 9(4):1607–1615. https://doi.org/10.1039/C6NR09260H
Huang G, Xiong Z, Qin H, Zhu J, Sun Z, Zhang Y, Peng X, Zou H (2014) Synthesis of zwitterionic polymer brushes hybrid silica nanoparticles via controlled polymerization for highly efficient enrichment of glycopeptides. Anal Chim Acta 809:61–68. https://doi.org/10.1016/j.aca.2013.11.049
Zhou J, Liang Y, He X, Chen L, Zhang Y (2017) Dual-functionalized magnetic metal–organic framework for highly specific enrichment of phosphopeptides. ACS Sustain Chem Eng 5(12):11413–11421. https://doi.org/10.1021/acssuschemeng.7b02521
Stewart II, Thomson T, Figeys D (2001) 18O labeling: a tool for proteomics. Rapid Commun Mass Spectrom 15(24):2456–2465. https://doi.org/10.1002/rcm.525
Funding
This work is supported by the National Institute of General Medical Sciences and the National Cancer Institute of the National Institutes of Health under Award Numbers R35GM141944 and U01CA185188.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Ethics approval
Sera from subjects recruited at Howard University Hospital (HUH) through IRB Protocol # 2014–0059.
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Additional information includes the Digestion protocols; Sample preparations; Characterizations (IR, and EDX); Selectivity, sensitivity, and reproducibility MALDI-MS spectra; MS/MS spectra and tables.
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Sajid, M.S., Saleem, S., Jabeen, F. et al. Terpolymeric platform with enhanced hydrophilicity via cysteic acid for serum intact glycopeptide analysis. Microchim Acta 189, 277 (2022). https://doi.org/10.1007/s00604-022-05343-0
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00604-022-05343-0