Abstract
A facile and versatile competitive electrochemical aptasensor for tobramycin (TOB) detection is described using electrochemical-deposited AuNPs coordinated with PEI-functionalized Fe-based metal-organic framework (AuNPs/P-MOF) as signal-amplification platform and a DNA probe labeled with methylene blue (MB) at the 3′-end (MB-Probe) as a signal producer. First, F-Probe (short complementary DNA strands of both the aptamer and the MB-Probe label with a sulfhydryl group at the 5′-end) was immobilized on the AuNPs/P-MOF modified electrode as detection probes, which competed with TOB in binding to the aptamer. TOB-aptamer binding resulted in F-Probe remaining unhybridized on the electrode surface, so that a significant current response was generated by hybridizing with MB-Probe instead. The developed strategy showed favorable repeatability, with a relative standard deviation (RSD) of 4.3% computed over five independent assays, and high stability, with only 6.8% degradation after 15 days of storage. Under optimal conditions, the proposed aptamer strategy exhibited a linear detection range from 100 pM to 500 nM with a limit of detection (LOD) of 56 pM (S/N = 3). The electrochemical aptasensor demonstrated remarkable selectivity, and its feasibility for accurate and quantitative detection of TOB in milk samples was confirmed (RSD < 4.5%). Due to its simple design, easy operation, and high sensitivity and selectivity, the proposed method could expect to detect other antibiotics by replacing the aptamers. In summary, this study provides a simple and effective new strategy for electrochemical aptasening based on MOF-based sensing interface.
Graphical abstract

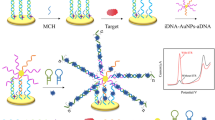
Scheme illustration of label-free competitive electrochemical aptamer-based detection of tobramycin based on electrochemically deposited AuNPs coordinated with PEI-functionalized Fe-based metal-organic framework as signal-amplification platform.






Similar content being viewed by others
Data availability
The data and material are available within this paper.
References
Cháfer-Pericás C, Maquieira Á, Puchades R (2010) Fast screening methods to detect antibiotic residues in food samples. TrAC Trends Anal Chem 29:1038–1049. https://doi.org/10.1016/j.trac.2010.06.004
Van Boeckel TP, Glennon EE, Chen D, Gilbert M, Robinson TP, Grenfell BT, Levin SA, Bonhoeffer S, Laxminarayan R (2017) Reducing antimicrobial use in food animals consider user fees and regulatory caps on veterinary use. Science 357:1350–1352. https://doi.org/10.1126/science.aao1495
Ma Q, Wang Y, Jia J, Xiang Y (2018) Colorimetric aptasensors for determination of tobramycin in milk and chicken eggs based on DNA and gold nanoparticles. Food Chem 249:98–103. https://doi.org/10.1016/j.foodchem.2018.01.022
Barco S, Mesini A, Barbagallo L, Maffia A, Tripodi G, Pea F, Saffioti C, Castagnola E, Cangemi G (2020) A liquid chromatography-tandem mass spectrometry platform for the routine therapeutic drug monitoring of 14 antibiotics: application to critically ill pediatric patients. J Pharmaceut Biomed Anal 186:113273. https://doi.org/10.1016/j.jpba.2020.113273
Orizondo RA, Fabiilli ML, Morales MA, Cook KE (2016) Effects of emulsion composition on pulmonary tobramycin delivery during antibacterial perfluorocarbon ventilation. J Aerosol Med Pulm D 29:251–259. https://doi.org/10.1089/jamp.2015.1235
Jiang L, Wei DL, Zeng K, Shao J, Zhu F, Du DL (2018) An enhanced direct competitive immunoassay for the detection of kanamycin and tobramycin in milk using multienzyme-particle amplification. Food Anal Method 11:2066–2075. https://doi.org/10.1007/s12161-018-1185-2
Kaufmann A, Butcher P, Maden K (2012) Determination of aminoglycoside residues by liquid chromatography and tandem mass spectrometry in a variety of matrices. Anal Chim Acta 711:46–53. https://doi.org/10.1016/j.aca.2011.10.042
Liu X, Jiang YN, Luo J, Guo XY, Ying Y, Wen Y, Yang HF, Wu YP (2021) A SnO2/Bi2S3- based photoelectrochemical aptasensor for sensitive detection of tobramycin in milk. Food Chem 344:128716. https://doi.org/10.1016/j.foodchem.2020.128716
Yan S, Lai X, Wang Y, Ye N, Xiang Y (2019) Label free aptasensor for ultrasensitive detection of tobramycin residue in pasteurized cow’s milk based on resonance scattering spectra and nanogold catalytic amplification. Food Chem 295:36–41. https://doi.org/10.1016/j.foodchem.2019.05.110
Zhang Z, Zhang M, Xu Y, Wen Z, Ding C, Guo Y, Hao N, Wang K (2020) Bi3+ engineered black anatase titania coupled with graphene for effective tobramycin photoelectrochemical detection. Sensor Actuators B-Chem 321:128464. https://doi.org/10.1016/j.snb.2020.128464
Garcia-Guzman JJ, Perez-Rafols C, Cuartero M, Crespo GA (2021) Microneedle based electrochemical (bio)sensing: towards decentralized and continuous health status monitoring. Trac-Trends Anal Chem 135:116148. https://doi.org/10.1016/j.trac.2020.116148
Du H, Xie Y, Wang J (2021) Nanomaterial-sensors for herbicides detection using electrochemical techniques and prospect applications. Trac-Trends Anal Chem 135:116178. https://doi.org/10.1016/j.trac.2020.116178
Lin Z, Liu X, Li Y, Li C, Yang L, Ma K, Zhang Z, Huang H (2021) Electrochemical aptasensor based on Mo2C/Mo2N and gold nanoparticles for determination of chlorpyrifos. Microchim Acta 188:170. https://doi.org/10.1007/s00604-021-04830-0
Walther BK, Dinu CZ, Guldi DM, Sergeyev VG, Creager SE, Cooke JP, Guiseppi-Elie A (2020) Nanobiosensing with graphene and carbon quantum dots: recent advances. Mater Today 39:23–46. https://doi.org/10.1016/j.mattod.2020.04.008
Negahdary M (2020) Electrochemical aptasensors based on the gold nanostructures. Talanta 216. https://doi.org/10.1016/j.talanta.2020.120999
Kang M, Li Z, Hu M, Oderinde O, Hu B, He L, Wang M, Fu G, Zhang Z, Du M (2020) Bimetallic MnCo oxide nanohybrids prepared from Prussian blue analogue for application as impedimetric aptasensor carrier to detect myoglobin. Chem Eng J 395. https://doi.org/10.1016/j.cej.2020.125117
Wang SJ, Li ZZ, Duan FH, Hu B, He LH, Wang MH, Zhou N, Jia QJ, Zhang ZH (2019) Bimetallic cerium/copper organic framework-derived cerium and copper oxides embedded by mesoporous carbon: label-free aptasensor for ultrasensitive tobramycin detection. Anal Chim Acta 1047:150–162. https://doi.org/10.1016/j.aca.2018.09.064
Xue Y, Zheng S, Xue H, Pang H (2019) Metal-organic framework composites and their electrochemical applications. J Mater Chem A 7:7301–7327. https://doi.org/10.1039/c8ta12178h
Li CW, Sun XJ, Yao YG, Hong G (2021) Recent advances of electrically conductive metal-organic frameworks in electrochemical applications. MaterToday Nano 13:100105. https://doi.org/10.1016/j.mtnano.2020.100105
Cruz-Navarro JA, Hernandez-Garcia F, Romero GAA (2020) Novel applications of metal-organic frameworks (MOFs) as redox-active materials for elaboration of carbon-based electrodes with electroanalytical uses. Coord Chem Rev 412:213263. https://doi.org/10.1016/j.ccr.2020.213263
Song Y, Xu M, Liu X, Li Z, Wang C, Jia Q, Zhang Z, Du M (2021) A label-free enrofloxacin electrochemical aptasensor constructed by a semiconducting CoNi-based metal-organic framework (MOF). Electrochimica Acta 368:137609. https://doi.org/10.1016/j.electacta.2020.137609
Li J, Liu L, Ai Y, Liu Y, Sun H, Liang Q (2020) Self-polymerized dopamine-decorated au NPs and coordinated with Fe-MOF as a dual binding sites and dual signal-amplifying electrochemical aptasensor for the detection of CEA. ACS Appl Mater Inter 12:5500–5510. https://doi.org/10.1021/acsami.9b19161
Petit C, Bandosz TJ (2011) Synthesis, characterization, and ammonia adsorption properties of mesoporous metal-organic framework (MIL(Fe))-graphite oxide composites: exploring the limits of materials fabrication. Adv Funct Mater 21:2108–2117. https://doi.org/10.1002/adfm.201002517
Liu X-W, Sun T-J, Hu J-L, Wang S-D (2016) Composites of metal-organic frameworks and carbon-based materials: preparations, functionalities and applications. J Mater Chem A 4:3584–3616. https://doi.org/10.1039/c5ta09924b
Lu J, Hu Y, Wang P, Liu P, Chen Z, Sun D (2020) Electrochemical biosensor based on gold nanoflowers-encapsulated magnetic metal-organic framework nanozymes for drug evaluation with in-situ monitoring of H2O2 released from H9C2 cardiac cells. Sensor Actuators B-Chem 311:127909. https://doi.org/10.1016/j.snb.2020.127909
Zhang J, Chen S, Ruo Y, Zhong X, Wu X (2015) An ultrasensitive electrochemiluminescent biosensor for the detection of concanavalin A based on poly(ethylenimine) reduced graphene oxide and hollow gold nanoparticles. Anal Bioanal Chem 407:447–453. https://doi.org/10.1007/s00216-014-8290-x
Rowe AA, Miller EA, Plaxco KW (2010) Reagentless measurement of aminoglycoside antibiotics in blood serum via an electrochemical, ribonucleic acid aptamer-based biosensor. Anal Chem 82:7090–7095. https://doi.org/10.1021/ac101491d
Liu YL, Fu WL, Li CM, Huang CZ, Li YF (2015) Gold nanoparticles immobilized on metal–organic frameworks with enhanced catalytic performance for DNA detection. Anal Chim Acta 861:55–61. https://doi.org/10.1016/j.aca.2014.12.032
Tan H, Chen Y (2012) Silver nanoparticle enhanced fluorescence of europium (III) for detection of tetracycline in milk. Sensors Actuators B-Chem 173:262–267. https://doi.org/10.1016/j.snb.2012.06.090
Cui L, Hu J, C-c L, Wang C-m, C-y Z (2018) An electrochemical biosensor based on the enhanced quasi-reversible redox signal of Prussian blue generated by self-sacrificial label of iron metal-organic framework. Biosens Bioelectron 122:168–174. https://doi.org/10.1016/j.bios.2018.09.061
Zhou L, He B, Huang J (2013) One-step synthesis of robust amine- and vinyl-capped magnetic iron oxide nanoparticles for polymer grafting, dye adsorption, and catalysis. ACS Appl Mater Inter 5:8678–8685. https://doi.org/10.1021/am402334f
Li Z, Yin J, Gao C, Qiu G, Meng A, Li Q (2019) The construction of electrochemical aptasensor based on coral-like poly-aniline and Au nano-particles for the sensitive detection of prostate specific antigen. Sensors Actuators B-Chem 295:93–100. https://doi.org/10.1016/j.snb.2019.05.070
Gao W, Xia X-H, Xu J-J, Chen H-Y (2007) Three-dimensionally ordered macroporous gold structure as an efficient matrix for solid-state electrochemiluminescence of Ru(bpy)32+/TPA system with high sensitivity. J Phys Chem C 111:12213–12219. https://doi.org/10.1021/jp0722814
Gao Z, Li Y, Zhang X, Feng J, Kong L, Wang P, Chen Z, Dong Y, Wei Q (2018) Ultrasensitive electrochemical immunosensor for quantitative detection of HBeAg using Au@Pd/MoS2@ MWCNTs nanocomposite as enzyme-mimetic labels. Biosens Bioelectron 102:189–195. https://doi.org/10.1016/j.bios.2017.11.032
Hajian R, Tayebi Z, Shams N (2017) Fabrication of an electrochemical sensor for determination of doxorubicin in human plasma and its interaction with DNA. J Pharmaceut Anal 7:27–33. https://doi.org/10.1016/j.jpha.2016.07.005
Code availability
Not applicable.
Funding
This work was supported by the National Natural Science Foundation of China (31901783); Postdoctoral Science Foundation of China (2019M652835); National Key R&D Program of China (2017YFC1600403), Guangdong Provincial Key Laboratory (2020B121201009) and GDAS’ Project of Science and Technology Development (2019GDASYL0103006, 2020GDASYL-0301002).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Ethics approval
Not applicable.
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
ESM 1
(DOCX 3.05 mb)
Rights and permissions
About this article
Cite this article
Zhang, Y., Li, B., Wei, X. et al. Amplified electrochemical antibiotic aptasensing based on electrochemically deposited AuNPs coordinated with PEI-functionalized Fe-based metal-organic framework. Microchim Acta 188, 286 (2021). https://doi.org/10.1007/s00604-021-04912-z
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00604-021-04912-z