Abstract
A glassy carbon (GC) electrode was modified with poly(1,8-diaminonaphthalene) (p-1,8-DAN) that was coated with silver nanoparticles (Ag NPs) (size: 10.0–60.0 nm by TEM) by electrodeposition process using cyclic voltammetry (CV) technique. The resulting nanocomposite was characterized by FE-SEM, AFM, EDX, XPS, TEM and XRD. The surface area and the electrochemical characteristics of the electrode were investigated by CV and square wave voltammetry (SWV) techniques, and the probe preparation conditions were optimized. The electrode was used for individual and simultaneous determination of the heavy metal ions cadmium(II) (Cd2+), lead(II) (Pb2+) and copper(II) (Cu2+) in water samples by square wave anodic stripping voltammetry (ASV) using scan rate 0.005 V. s−1. The probe showed well separated anodic stripping peaks for Cd2+, Pb2+, and Cu2+. Attractive features of the method include (a) peak voltages of −1.02, −0.78 and − 0.32 V (vs. Ag/AgCl) for the three ions, and (b) low limits of detection (19, 30 and 6 ng.L−1, respectively. The electrode can also detect zinc(II) (Zn2+) and mercury(II) (Hg2+), typically at −1.36 V and + 0.9, respectively.

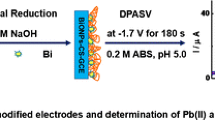
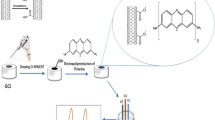
Schematic presentation of simultaneous electrochemical determination of Pb2+, Cd2+, and Cu2+ at a poly(1,8-diaminonaphthalene)-modified glassy carbon electrode coated with silver nanoparticles.







Similar content being viewed by others
References
El Mhammedi MA, Achak M, Bakasse M (2013) Evaluation of a platinum electrode modified with hydroxyapatite in the lead (II) determination in a square wave voltammetric procedure. Arab J Chem 6(3):299–305. https://doi.org/10.1016/j.arabjc.2010.10.010
Pohl P (2009) Determination of metal content in honey by atomic absorption and emission spectrometries. Trends Anal Chem 28(1):117–128. https://doi.org/10.1016/j.trac.2008.09.015
Wang J (2005) Stripping analysis at bismuth electrodes: a review. Electroanal 17(15–16):1341–1346. https://doi.org/10.1002/elan.200403270
Su Z, Liu Y, Zhang Y, Xie Q, Chen L, Huang Y, Fu Y, Meng Y, Li X, Ma M, Yao S (2013) Thiol–ene chemistry guided preparation of thiolated polymeric nanocomposite for anodic stripping voltammetric analysis of Cd2+ and Pb2+. Analyst 138(4):1180–1186. https://doi.org/10.1039/C2AN36114K
Tang J, Li Z, Xia Q, Williams RS (2009) Fractal structure formation from Ag nanoparticle films on insulating substrates. Langmuir 25(13):7222–7225. https://doi.org/10.1021/la9010532
March G, Nguyen T, Piro B (2015) Modified electrodes used for electrochemical detection of metal ions in environmental analysis. Biosens 5(2):241–275. https://doi.org/10.3390/bios5020241
Wang Z, Ma L (2009) Gold nanoparticle probes. Coord Chem Rev 253(11–12):1607–1618. https://doi.org/10.1016/j.ccr.2009.01.005
Cheon J, Lee J-H (2008) Synergistically integrated nanoparticles as multimodal probes for Nanobiotechnology. Acc Chem Res 41(12):1630–1640. https://doi.org/10.1021/ar800045c
Berlina AN, Zherdev AV, Dzantiev BB (2019) Progress in rapid optical assays for heavy metal ions based on the use of nanoparticles and receptor molecules. Microchim Acta 186(3):172. https://doi.org/10.1007/s00604-018-3168-9
Hahm J-I (2013) Biomedical detection via macro- and nano-sensors fabricated with metallic and semiconducting oxides. J BIiomed Nanotechnol 9(1):1–25. http://www.ncbi.nlm.nih.gov/pmc/articles/PMC3766318/
Nishijo J, Oishi O, Judai K, Nishi N (2007) Facile and mass-producible fabrication of one-dimensional ag nanoparticle arrays. Chem Mater 19(19):4627–4629. https://doi.org/10.1021/cm071688i
Zheng J, Li X, Gu R, Lu T (2002) Comparison of the surface properties of the assembled silver nanoparticle electrode and roughened silver electrode. J Phys Chem B 106(5):1019–1023. https://doi.org/10.1021/jp012083r
Sandmann G, Dietz H, Plieth W (2000) Preparation of silver nanoparticles on ITO surfaces by a double-pulse method. J Electroanal Chem 491(1–2):78–86. https://doi.org/10.1016/S0022-0728(00)00301-6
Xing S, Xu H, Chen J, Shi G, Jin L (2011) Nafion stabilized silver nanoparticles modified electrode and its application to Cr (VI) detection. J Electroanal Chem 652(1–2):60–65. https://doi.org/10.1016/j.jelechem.2010.03.035
Hassan K, Abdel Azzem M (2015) Electrocatalytic oxidation of ascorbic acid, uric acid, and glucose at nickel nanoparticles/poly (1-amino-2-methyl-9,10-anthraquinone) modified electrode in basic medium. J Appl Electrochem 45(6):567–575. https://doi.org/10.1007/s10800-015-0805-4
Ng KH, Liu H, Penner RM (2000) Subnanometer silver clusters exhibiting unexpected electrochemical Metastability on graphite. Langmuir 16(8):4016–4023. https://doi.org/10.1021/la9914716
Shameli K, Ahmad MB, Jazayeri SD, Shabanzadeh P, Sangpour P, Jahangirian H, Gharayebi Y (2012) Investigation of antibacterial properties silver nanoparticles prepared via green method. Chem Cent J 6(1):73. https://doi.org/10.1186/1752-153x-6-73
Bard AJ, Faulkner LR (2001) Fundamentals and applications, vol 2. Electrochemical Methods
Salih FE, Ouarzane A, El Rhazi M (2017) Electrochemical detection of lead (II) at bismuth/poly(1,8-diaminonaphthalene) modified carbon paste electrode. Arab J Chem 10(5):596–603. https://doi.org/10.1016/j.arabjc.2015.08.021
Hutton LA, Newton ME, Unwin PR, Macpherson JV (2011) Factors controlling stripping voltammetry of Lead at polycrystalline boron doped diamond electrodes: new insights from high-resolution microscopy. Anal Chem 83(3):735–745. https://doi.org/10.1021/ac101626s
Hassan KM, Elsaid Gaber S, Fatehy M, Abdel Azzem M (2018) Novel sensor based on poly (1,2-Diaminoanthraquinone) for individual and simultaneous anodic stripping voltammetry of Cd2+, Pb2+, Cu2+ and Hg2+. J Electroanal 30(6):1155–1162. https://doi.org/10.1002/elan.201800097
Vu HD, Nguyen L-H, Nguyen TD, Nguyen HB, Nguyen TL, Tran DL (2015) Anodic stripping voltammetric determination of Cd2+ and Pb2+ using interpenetrated MWCNT/P1,5-DAN as an enhanced sensing interface. Ionics 21(2):571–578. https://doi.org/10.1007/s11581-014-1199-8
Guo Z, Li D-d, Luo X-k, Li Y-h, Zhao Q-N, Li M-m, Zhao Y-t, Sun T-s, Ma C (2017) Simultaneous determination of trace Cd (II), Pb (II) and Cu (II) by differential pulse anodic stripping voltammetry using a reduced graphene oxide-chitosan/poly-l-lysine nanocomposite modified glassy carbon electrode. J Colloid Interface Sci 490:11–22. https://doi.org/10.1016/j.jcis.2016.11.006
Hwang GH, Han WK, Park JS, Kang SG (2008) Determination of trace metals by anodic stripping voltammetry using a bismuth-modified carbon nanotube electrode. Talanta 76(2):301–308. https://doi.org/10.1016/j.talanta.2008.02.039
Wang Z, Wang H, Zhang Z, Liu G (2014) Electrochemical determination of lead and cadmium in rice by a disposable bismuth/electrochemically reduced graphene/ionic liquid composite modified screen-printed electrode. Sensors Actuators B Chem 199:7–14. https://doi.org/10.1016/j.snb.2014.03.092
Ouyang R, Zhu Z, Tatum CE, Chambers JQ, Xue Z-L (2011) Simultaneous stripping detection of Zn (II), Cd (II) and Pb (II) using a bimetallic Hg–Bi/single-walled carbon nanotubes composite electrode. J Electroanal Chem 656(1–2):78–84. https://doi.org/10.1016/j.jelechem.2011.01.006
Huang H, Chen T, Liu X, Ma H (2014) Ultrasensitive and simultaneous detection of heavy metal ions based on three-dimensional graphene-carbon nanotubes hybrid electrode materials. Anal Chim Acta 852:45–54. https://doi.org/10.1016/j.aca.2014.09.010
Li J, Guo S, Zhai Y, Wang E (2009) High-sensitivity determination of lead and cadmium based on the Nafion-graphene composite film. Anal Chim Acta 649(2):196–201. https://doi.org/10.1016/j.aca.2009.07.030
Pokpas K, Zbeda S, Jahed N, Mohamed N, Baker PG, Iwuoha EI (2014) Electrochemically reduced graphene oxide pencil-graphite in situ plated bismuth-film electrode for the determination of trace metals by anodic stripping voltammetry. Int J Electrochem Sci 9:736–759. http://hdl.handle.net/10566/3290
Chamjangali MA, Kouhestani H, Masdarolomoor F, Daneshinejad H (2015) A voltammetric sensor based on the glassy carbon electrode modified with multi-walled carbon nanotube/poly(pyrocatechol violet)/bismuth film for determination of cadmium and lead as environmental pollutants. Sensors Actuators B Chem 216:384–393. https://doi.org/10.1016/j.snb.2015.04.058
Serrano N, González-Calabuig A, del Valle M (2015) Crown ether-modified electrodes for the simultaneous stripping voltammetric determination of Cd (II), Pb (II) and Cu (II). Talanta 138:130–137. https://doi.org/10.1016/j.talanta.2015.01.044
Sahoo PK, Panigrahy B, Sahoo S, Satpati AK, Li D, Bahadur D (2013) In situ synthesis and properties of reduced graphene oxide/Bi nanocomposites: As an electroactive material for analysis of heavy metals. Biosens Bioelectron 43:293–296. https://doi.org/10.1016/j.bios.2012.12.031
Silva ACO, Oliveira LCF, Delfino AV, Meneghetti MR, Abreu FC (2016) Electrochemical study of carbon nanotubes/Nanohybrids for determination of metal species Cu2+ and Pb2+ in water samples. J AnalL Methods Chem 2016:1–12. https://doi.org/10.1155/2016/9802738
Chira A, Bucur B, Bucur MP, Radu GL (2014) Electrode-modified with nanoparticles composed of 4,4[prime or minute]-bipyridine-silver coordination polymer for sensitive determination of Hg (II), Cu (II) and Pb (II). New J Chem 38(11):5641–5646. https://doi.org/10.1039/C4NJ01245C
Sang S, Li D, Zhang H, Sun Y, Jian A, Zhang Q, Zhang W (2017) Facile synthesis of AgNPs on reduced graphene oxide for highly sensitive simultaneous detection of heavy metal ions. RSC Adv 7(35):21618–21624. https://doi.org/10.1039/C7RA02267K
Acknowledgements
The authors are appreciative to Alexander von Humboldt Foundation for providing some electrodes and accessories.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
The author(s) declare that they have no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
ESM 1
(DOCX 147 kb)
Rights and permissions
About this article
Cite this article
Hassan, K.M., Elhaddad, G.M. & AbdelAzzem, M. Voltammetric determination of cadmium(II), lead(II) and copper(II) with a glassy carbon electrode modified with silver nanoparticles deposited on poly(1,8-diaminonaphthalene). Microchim Acta 186, 440 (2019). https://doi.org/10.1007/s00604-019-3552-0
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00604-019-3552-0