Abstract
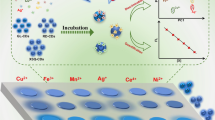
An enzyme-linked immunoassay is described for the fluorometric determination of aflatoxin B1 (AFB1). It is based on the use of carbon dots (CDs) synthesized by using Litchi chinensis as the carbon source via a hydrothermal method. The CDs were modified with MnO2 nanosheets upon which their blue fluorescence (with excitation/emission peaks at 340/425 nm) is quenched. In the presence of AFB1, a competitive immunoreaction takes place between (1) AFB1 conjugated to bovine serum albumin and deposited in the wells of a microplate, and (2) antibody against AFB1 and labeled with alkaline phosphatase (ALP). On subsequent addition of ascorbic acid 2-phosphate, it will be hydrolyzed by ALP to form ascorbic acid and phosphate. The ascorbic acid produced reduces MnO2 nanosheets to Mn2+ ions which then are relased from the CDs. This causes the recovery of fluorescence. Under optimum conditions, fluorescence decreases linearly with increasing AFB1 concentration in the range from 1.0 ng·kg−1 to 10 μg·kg−1, and the limit of detection is 0.69 ng·kg−1. The precision of this method (expressed as RSD) is ±10.3%. The accuracy was tested by analyzing both naturally contaminated and spiked food samples, and the results were in good agreement with those obtained by the established ELISA.

Enzyme hydrolysate-triggered redox reaction of carbon dot-functionalized MnO2 nanosheets was developed for the fluorescence immunoassay of aflatoxin B1 in foodstuff.




Similar content being viewed by others
References
Lim S, Shen W, Gao Z (2015) Carbon quantum dots and their applications. Chem Soc Rev 44:362–381
Fu L, Wang A, Lai G, Lin C, Yu J, Yu A, Liu Z, Xie K, Su W (2018) A glassy carbon electrode modified with N-doped carbon dots for improved detection of hydrogen peroxide and paracetamol. Microchim Acta 185:87
Rong M, Deng X, Chi S, Huang L, Zhou Y, Shen Y, Chen X (2018) Ratiometric fluorometric determination of the anthrax biomarker 2,6-dipicolinic acid by using europium(III)-doped carbon dots in a test stripe. Microchim Acta 185:201
Ma H, Liu X, Wang X, Li X, Yang C, Iqbal A, Liu W, Li J, Qin W (2017) Sensitive fluorescent light-up probe for enzymatic determination of glucose using carbon dots modified with MnO2 nanosheets. Microchim Acta 184:177–185
Sun Y, Wang X, Wang C, Tong D, Wu Q, Jiang K, Jiang Y, Wang C (2018) Red emitting and highly stable carbon dots with dual response to pH values and ferric ions. Microchim Acta 185:83
Hu L, Zhang Q, Gan X, Lin S, Han S, Zhang Z (2018) Fluorometric turn-on determination of the activity of alkaline phosphatase by using WS2 quantum dots and enzymatic cleavage of ascorbic acid 2-phosphate. Microchim Acta 185:390
Mohammadi S, Salimi A (2018) Fluorometric determination of microRNA-155 in cancer cells based on carbon dots and MnO2 nanosheets as a donor acceptor pair. Microchim Acta 185:372
Shu J, Tang D (2017) Current advances in quantum dots-based photoelectrochemical immunoassays. Chem Asian J 12:2780–2789
Das R, Bandyopadhyay R, Pramanik P (2018) Carbon quantum dots from natural resource: a review. Mat Today Chem 8:96–109
Goryacheva I, Sapelkin A, Sukhorukov G (2017) Carbon nanodots: mechanisms of photoluminescence and principles of application. TrAC-Anal Chem 90:27–37
Sun X, Lei Y (2017) Fluorescent carbon dots and their sensing applications. TrAC Anal Chem 89:163–180
Wang Y, Zhu Y, Yu S, Jiang C (2017) Fluorescent carbon dots: rational synthesis, tunable optical properties and analytical applications. RSC Adv 7:40973–40989
Zhou J, Booker C, Li R, Zhou X, Sham T, Sun X, Ding Z (2007) An electrochemical avenue to blue luminescent nanocrystals from multiwalled carbon nanotubers (MWCNTs). J Am Chem Soc 129:744–745
Yang H, He L, Long Y, Li H, Pan S, Liu H, Hu X (2018) Fluorescent carbon dots synthesized by microwave-assisted pyrolysis of chromium(VI) and ascorbic acid sensing and logic gate operation. Spectrochim Acta A 205:12–20
Amin N, Afkhami A, Hosseinzadeh L, Madrakian T (2018) Green and cost-effective synthesis of carbon dots from date kernel and their application as a novel switchable fluorescence probe for sensitive assay of Zoledronic acid in human serum and cellular imaging. Anal Chim Acta 1030:183–193
Ahmadian-Fard-Fini S, Salavati-Niasari M, Ghanbari D (2018) Green synthesis of magnetic Fe3O4-carbon dots by lemon and grape fruit extracts and as a photoluminescescence sensor for detecting of E. Coli bacteria. Spectrochim Acta A 203:481–493
Anmei S, Qingmei Z, Yuye C, Yilin W (2018) Preparation of carbon quantum dots from cigarette filters and its application for fluorescence detection of Sudan I. Anal Chim Acta 1023:115–120
Nutrient contents for Litchis, raw, per 100 g, United States Department of Agriculture (USDA), 2013
Pierre B, Stephane G, Annick B, Laure D, Augustin S, Louise M, Nathalie A, Marie J (2006) Daily polyphenol intake in France from fruit and vegetables. J Nutrition 136:2368–2373
Lin Y, Zhou Q, Zeng Y, Tang D (2018) Liposome-coated mesoporous silica nanoparticles loaded with L-cysteine for photoelectrochemical immunoassay of aflatoxin B1. Microchim Acta 185:311
Zu F, Yan F, Bai Z, Xu J, Wang Y, Huang Y, Zhou X (2017) The quenching of the fluorescence of carbon dots: a review on mechanisms and applications. Microchim Acta 184:1899–1914
Hermanson G, Bioconjugate Techniques, 2nd Ed., Pierce Biotechnology, Thermo Fisher Scientific, Rockford Illiois, USA, pp.798–799
Chaudhary V, Bhowmick A (2015) Green synthesis of fluorescent carbon nanoparticles from lychee (Litchi chinensis) plant. Korean J Chem Eng 32:1707–1711
Lin Y, Zhou Q, Tang D, Niessner R, Knopp D (2017) Signal-on photoelectrochemical immunoassay for aflatoxin B1 based on enzymatic product-etching MnO2 nanosheets for dissociation of carbon dots. Anal Chem 89:5637–5645
Yu Z, Lv S, Ren R, Cai G, Tang D (2017) Photoelectrochemical sensing of hydrogen peroxide at zero working potential using a fluorine-doped tin oxide electrode modified with BiVO4 microrods. Microchim Acta 184:799–806
Wang D, Lin B, Cao Y, Guo M, Yu Y (2016) A highly selective and sensitive fluorescence detection method of glyphosate based on an immune reaction strategy of carbon dot labeled antibody and antigen magnetic beads. J Agric Food Chem 64:6042–6050
Lin Y, Zhou Q, Lin Y, Tang D, Niessner R, Knopp D (2015) Enzymatic hydrolysate-induced displacement reaction with multifunctional silica beads doped with horseradish peroxidase thionine conjugate for ultrasensitive electrochemical immunoassay. Anal Chem 87:8531–8540
Lin Y, Zhou Q, Tang D, Niessner R, Yang H, Knopp D (2016) Silver nanolabels-assisted ion-exchange reaction with CdTe quantum dots mediated exciton trapping for signal-on photoelectrochemical immunoassay of mycotoxins. Anal Chem 88:7858–7866
Taghdisi S, Danesh N, Ramezani M, Abnous K (2018) A new amplified fluorescent aptasensor based on hairpin structure of G-quadruplex oligonucleotide-aptamer chimera and silica nanoparticles for sensitive detection of aflatoxin B1 in the grape juice. Food Chem 268:342–346
Zhang Y, Liao Z, Liu Y, Wang Y, Chang J, Wang H (2017) Flow cytometric immunoassay for aflatoxin B1 using magnetic microspheres encoded with upconverting fluorescent nanocrystals. Microchim Acta 184:1471–1479
Pan D, Li G, Hu H, Xue H, Zhang M, Zhu M, Gong X, Zhang Y, Wan Y, Shen Y (2018) Direct immunoassay for facile and sensitive detection of small molecule aflatoxin B1. Chem Eur J 24:9869–9876
Wang C, Qian J, An K, Ren C, Lu X, Hao N, Liu Q, Li H, Huang X, Wang K (2018) Fabrication of magnetically assembled aptasensing device for label-free determination of aflatoxin B1 based on EIS. Biosens Bioelectron 108:69–75
Lai W, Zeng Q, Tang J, Zhang M, Tang D (2018) A conventional chemical reaction for use in an unconventional assay: a colorimetric immunoassay for aflatoxin B1 by using enzyme-responsive just-in-time generation of a MnO2 based nanocatalyst. Microchim Acta 185:92
Acknowledgements
Support by the National Natural Science Foundation of China (Grant nos.: 21475025, 21675029 and 21874022), and the Chongqing Science & Technology Commission of China (Grant no.: CSTC2015JCYJBX0126 and CSTC2016SHMSZX20001) is gratefully acknowledged.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
The author(s) declare that they have no competing interests.
Electronic supplementary material
ESM 1
(DOCX 149 kb)
Rights and permissions
About this article
Cite this article
Tang, D., Lin, Y. & Zhou, Q. Carbon dots prepared from Litchi chinensis and modified with manganese dioxide nanosheets for use in a competitive fluorometric immunoassay for aflatoxin B1. Microchim Acta 185, 476 (2018). https://doi.org/10.1007/s00604-018-3012-2
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00604-018-3012-2