Abstract
It is found that the fluorescence of carbon dots (CD) with an emission peak at 459 nm is strongly quenched by silver nanoparticles (AgNPs) with their absorption peak at 430 nm. The finding was applied in a fluorescence quenchometric lateral flow immunochromatographic assay for detection of zearalenone (ZEN) with CDs conjugated to ovalbumin (OVA) as donor signal probe and AgNP-Ab as acceptor signal probe. The assay has an LOD of 0.1 μg·L−1 for ZEN. This is 10 times better than the respective “turn-off” AgNP-based LFIA. In case of cereal samples and their products, the LODs range from 1 to 2.5 μg·kg−1. Only minor cross reactivity is found for fusarium toxins, and no cross-sensitivity for aflatoxin B1, T-2 mycotoxin, ochratoxin A, deoxynivalenol, and fumonisin B1. The assay represents a simple, sensitive, and rapid tool for determination of ZEN in cereal samples and their products.

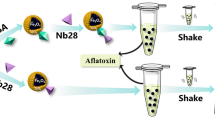
Schematic presentation of fluorescence quenching lateral flow immunochromatographic assay (FLFIA) based on carbon dots (CD) and silver nanoparticle (AgNP) fluorescence resonance energy transfer (FRET) system for the rapid high sensitive detection of zearalenone (ZEN) in cereal samples.



Similar content being viewed by others
References
Bennett JW, Klich M (2003) Mycotoxins. Clin Microbiol Rev 16(3):497–516. https://doi.org/10.1128/cmr.16.3.497-516.2003
Hussein HS, Brasel JM (2001) Toxicity, metabolism, and impact of mycotoxins on humans and animals. Toxicology 167(2):101
Pang J, Zhou Q, Sun X, Li L, Zhou B, Zeng F, Zhao Y, Shen W, Sun Z (2017) Effect of low-dose zearalenone exposure on reproductive capacity of male mice. Toxicol Appl Pharmacol 333:60–67. https://doi.org/10.1016/j.taap.2017.08.011
Wozny M, Obremski K, Zalewski T, Mommens M, Lakomiak A, Brzuzan P (2017) Transfer of zearalenone to the reproductive system of female rainbow trout spawners: a potential risk for aquaculture and fish consumers? Food Chem Toxicol 107(Pt A):386–394. https://doi.org/10.1016/j.fct.2017.07.010
Abassi H, Ayed-Boussema I, Shirley S, Abid S, Bacha H, Micheau O (2016) The mycotoxin zearalenone enhances cell proliferation, colony formation and promotes cell migration in the human colon carcinoma cell line HCT116. Toxicol Lett 254:1–7. https://doi.org/10.1016/j.toxlet.2016.04.012
Gajecka M, Zielonka L, Gajecki M (2016) Activity of Zearalenone in the porcine intestinal tract. Molecules 22(1). https://doi.org/10.3390/molecules22010018
Smith MC, Madec S, Pawtowski A, Coton E, Hymery N (2017) Individual and combined toxicological effects of deoxynivalenol and zearalenone on human hepatocytes in in vitro chronic exposure conditions. Toxicol Lett 280:238–246. https://doi.org/10.1016/j.toxlet.2017.08.080
Vogelgsang S, Musa T, Banziger I, Kagi A, Bucheli TD, Wettstein FE, Pasquali M, Forrer HR (2017) Fusarium mycotoxins in Swiss wheat: a survey of Growers' samples between 2007 and 2014 shows strong year and minor geographic effects. Toxins 9(8). https://doi.org/10.3390/toxins9080246
Chen F, Luan C, Wang L, Wang S, Shao L (2017) Simultaneous determination of six mycotoxins in peanut by high-performance liquid chromatography with a fluorescence detector. J Sci Food Agric 97(6):1805–1810. https://doi.org/10.1002/jsfa.7978
De Santis B, Debegnach F, Gregori E, Russo S, Marchegiani F, Moracci G, Brera C (2017) Development of a LC-MS/MS method for the multi-mycotoxin determination in composite cereal-based samples. Toxins 9(5). https://doi.org/10.3390/toxins9050169
Toda K, Kokushi E, Uno S, Shiiba A, Hasunuma H, Fushimi Y, Wijayagunawardane MPB, Zhang C, Yamato O, Taniguchi M, Fink-Gremmels J, Takagi M (2017) Gas chromatography-mass spectrometry for metabolite profiling of Japanese black cattle naturally contaminated with Zearalenone and Sterigmatocystin. Toxins 9(10). https://doi.org/10.3390/toxins9100294
Maragos CM, Appell M (2007) Capillary electrophoresis of the mycotoxin zearalenone using cyclodextrin-enhanced fluorescence. J Chromatogr A 1143(1-2):252–257. https://doi.org/10.1016/j.chroma.2006.12.085
Chen Y, Fu Q, Li D, Xie J, Ke D, Song Q, Tang Y, Wang H (2017) A smartphone colorimetric reader integrated with an ambient light sensor and a 3D printed attachment for on-site detection of zearalenone. Anal Bioanal Chem 409(28):6567–6574
Lv Y, Wu R, Feng K, Li J, Mao Q, Yuan H, Shen H, Chai X, Li LS (2017) Highly sensitive and accurate detection of C-reactive protein by CdSe/ZnS quantum dot-based fluorescence-linked immunosorbent assay. J Nanobiotechnol 15(1):35
Liu N, Nie D, Tan Y, Zhao Z, Liao Y, Wang H, Sun C, Wu A (2016) An ultrasensitive amperometric immunosensor for zearalenones based on oriented antibody immobilization on a glassy carbon electrode modified with MWCNTs and AuPt nanoparticles. Microchim Acta 184(1):147–153
Xu W, Qing Y, Chen S, Chen J, Qin Z, Qiu J, Li C (2017) Electrochemical indirect competitive immunoassay for ultrasensitive detection of zearalenone based on a glassy carbon electrode modified with carboxylated multi-walled carbon nanotubes and chitosan. Microchim Acta 184(9):3339–3347
Zhang X, Yu X, Wen K, Li C, Mujtaba Mari G, Jiang H, Shi W, Shen J, Wang Z (2017) Multiplex Lateral Flow Immunoassays Based on Amorphous Carbon Nanoparticles for Detecting Three Fusarium Mycotoxins in Maize J Agric Food Chem 65(36): 8063-8071
Goryacheva OA, Beloglazova NV, Vostrikova AM, Pozharov MV, Sobolev AM, Goryacheva IY (2017) Lanthanide-to-quantum dot Forster resonance energy transfer (FRET): application for immunoassay. Talanta 164:377–385. https://doi.org/10.1016/j.talanta.2016.11.054
Fu Q, Liang J, Lan C, Zhou K, Shi C, Tang Y (2014) Development of a novel dual-functional lateral-flow sensor for on-site detection of small molecule analytes. Sensors Actuators B Chem 203:683–689. https://doi.org/10.1016/j.snb.2014.06.043
Shi CY, Deng N, Liang JJ, Zhou KN, Fu QQ, Tang Y (2015) A fluorescent polymer dots positive readout fluorescent quenching lateral flow sensor for ractopamine rapid detection. Anal Chim Acta 854:202–208. https://doi.org/10.1016/j.aca.2014.11.005
Zhang G, Chen M, Liu D, Xiong Y, Feng R, Zhong P, Lai W (2016) Quantitative detection of β2-adrenergic agonists using fluorescence quenching by immunochromatographic assay. Anal Methods 8(3):627–631. https://doi.org/10.1039/c5ay02585k
Wang J, Cao F, He S, Xia Y, Liu X, Jiang W, Yu Y, Zhang H, Chen W (2018) FRET on lateral flow test strip to enhance sensitivity for detecting cancer biomarker. Talanta 176:444–449. https://doi.org/10.1016/j.talanta.2017.07.096
Shen, H., Xu, F., Xiao, M., Fu, Q., Cheng, Z., & Zhang, S., et al. (2017). A new lateral-flow immunochromatographic strip combined with quantum dot nanobeads and gold nanoflowers for rapid detection of tetrodotoxin. Analyst,142.https://doi.org/10.1039/C7AN01227F
Hu G, Sheng W, Li S, Zhang Y, Wang J, Wang S (2017) Quantum dot based multiplex fluorescence quenching immune chromatographic strips for the simultaneous determination of sulfonamide and fluoroquinolone residues in chicken samples. RSC Adv 7(49):31123–31128. https://doi.org/10.1039/c7ra01753g
Hu G, Sheng W, Li J, Zhang Y, Wang J, Wang S (2017) Fluorescent quenching immune chromatographic strips with quantum dots and upconversion nanoparticles as fluorescent donors for visual detection of sulfaquinoxaline in foods of animal origin. Anal Chim Acta 982:185–192. https://doi.org/10.1016/j.aca.2017.06.013
Shamsipur M, Barati A, Karami S (2017) Long-wavelength, multicolor, and white-light emitting carbon-based dots: achievements made, challenges remaining, and applications. Carbon 124:429–472. https://doi.org/10.1016/j.carbon.2017.08.072
Bu D, Zhuang H, Yang G, Ping X (2014) An immunosensor designed for polybrominated biphenyl detection based on fluorescence resonance energy transfer (FRET) between carbon dots and gold nanoparticles. Sensors Actuators B Chem 195:540–548. https://doi.org/10.1016/j.snb.2014.01.079
Thouvenot D, Morfin RF (1983) Radioimmunoassay for zearalenone and zearalanol in human serum: production, properties, and use of porcine antibodies. Appl Environ Microbiol 45(1):16
Zhu S, Meng Q, Wang L, Zhang J, Song Y, Jin H, Zhang K, Sun H, Wang H, Yang B (2013) Highly photoluminescent carbon dots for multicolor patterning, sensors, and bioimaging. Angew Chem 52(14):3953–3957. https://doi.org/10.1002/anie.201300519
Cao Y, Zheng R, Ji X, Liu H, Xie R, Yang W (2014) Syntheses and characterization of nearly monodispersed, size-tunable silver nanoparticles over a wide size range of 7-200 nm by tannic acid reduction. Langmuir the Acs Journal of Surfaces & Colloids 30(13):3876–3882
Bao L, Zhang Z-L, Tian Z-Q, Zhang L, Liu C, Lin Y, Qi B, Pang D-W (2011) Electrochemical tuning of luminescent carbon Nanodots: from preparation to luminescence mechanism. Adv Mater 23(48):5801–5806. https://doi.org/10.1002/adma.201102866
Afzali D, Fathirad F (2016) Determination of zearalenone with a glassy carbon electrode modified with nanocomposite consisting of palladium nanoparticles and a conductive polymeric ionic liquid. Microchim Acta 183(9):2633–2638. https://doi.org/10.1007/s00604-016-1907-3
Li T, Kim BB, Shim W-B, Byun J-Y, Chung D-H, Shin Y-B, Kim M-G (2014) Homogeneous fluorescence resonance energy transfer immunoassay for the determination of Zearalenone. Anal Lett 47(3):453–464. https://doi.org/10.1080/00032719.2013.843186
Wu S, Liu L, Duan N, Li Q, Zhou Y, Wang Z (2018) Aptamer-based lateral flow test strip for rapid detection of Zearalenone in corn samples. J Agric Food Chem 66(8):1949–1954. https://doi.org/10.1021/acs.jafc.7b05326
Li SJ, Sheng W, Wen W, Gu Y, Wang JP, Wang S (2018) Three kinds of lateral flow immunochromatographic assays based on the use of nanoparticle labels for fluorometric determination of zearalenone. Mikrochimica acta 185(4):238. https://doi.org/10.1007/s00604-018-2778-6
Acknowledgments
We would like to thank The National Key R&D Program of China (No. 2016YFD0401202), Special Project of Tianjin Innovation Platform (No.17PTGCCX00230).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
The author(s) declare that they have no competing interests.
Electronic supplementary material
ESM 1
(DOCX 1293 kb)
Rights and permissions
About this article
Cite this article
Li, S., Wang, J., Sheng, W. et al. Fluorometric lateral flow immunochromatographic zearalenone assay by exploiting a quencher system composed of carbon dots and silver nanoparticles. Microchim Acta 185, 388 (2018). https://doi.org/10.1007/s00604-018-2916-1
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00604-018-2916-1