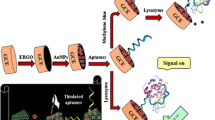
Abstract
The authors describe the fabrication of an electrochemical immunosensor for the determination of the activity of protein kinase A (PKA). The method involves (a) electrochemical deposition of gold nanoparticles (AuNPs) on a glassy carbon electrode, (b) PKA-induced catalytic phosphorylation of serine, and (c) the use of phosphoserine antibody and horseradish peroxidase conjugated to IgG on gold nanoparticles (HRP-IgG-AuNPs). The use of AuNPs and HRP-IgG-AuNPs results in large amplification so that the method, at a typical working potential as low as 0.08 V (vs. SCE), has a linear range that extends from 0.1 to 50 activity units per mL, and the detection limit is 0.026 units per mL (at an S/N ratio of 3). The assay is selective (not the least due to a rather low working potential) and well reproducible. It may also be applied to screening for PKA inhibitors and to quantify the PKA activity in human cell lysates.

ᅟ




Similar content being viewed by others
References
López-Otín C, Hunter T (2010) The regulatory crosstalk between kinases and proteases in cancer. Nat rev Cancer 10(4):278–292. doi:10.1038/nrc2823
Griner EM, Kazanietz MG (2007) Protein kinase C and other diacylglycerol effectors in cancer. Nat rev Cancer 7(4):281–294. doi:10.1038/nrc2110
Flajolet M, He G, Heiman M, Lin A, Nairn AC, Greengard P (2007) Regulation of Alzheimer's disease amyloid-β formation by casein kinase I. Proc Natl Acad Sci 104(10):4159–4164. doi:10.1073/pnas.0611236104
Turk BE, Hutti JE, Cantley LC (2006) Determining protein kinase substrate specificity by parallel solution-phase assay of large numbers of peptide substrates. Nat Protoc 1(1):375. doi:10.1038/nprot.2006.57
Wang Z, Lee J, Cossins AR, Brust M (2005) Microarray-based detection of protein binding and functionality by gold nanoparticle probes. Anal Chem 77(17):5770–5774. doi:10.1021/ac050679v
Wang Z, Lévy R, Fernig DG, Brust M (2006) Kinase-catalyzed modification of gold nanoparticles: a new approach to colorimetric kinase activity screening. J Am Chem Soc 128(7):2214–2215. doi:10.1021/ja058135y
Shen C, Zhang K, Gao N, Wei S, Liu G, Chai Y, Yang M (2016) Colorimetric and electrochemical determination of the activity of protein kinase based on retarded particle growth due to binding of phosphorylated peptides to DNA-capped silver nanoclusters. Microchim Acta 183(11):2933–2939. doi:10.1007/s00604-016-1944-y
Zhou J, Xu X, Liu W, Liu X, Nie Z, Qing M, Nie L, Yao S (2013) Graphene oxide-peptide nanocomplex as a versatile fluorescence probe of protein kinase activity based on phosphorylation protection against carboxypeptidase digestion. Anal Chem 85(12):5746–5754. doi:10.1021/ac400336u
Ji L, Wu J-H, Luo Q, Li X, Zheng W, Zhai G, Wang F, Lu S, Feng Y-Q, Liu J (2012) Quantitative mass spectrometry combined with separation and enrichment of phosphopeptides by titania coated magnetic mesoporous silica microspheres for screening of protein kinase inhibitors. Anal Chem 84(5):2284–2291. doi:10.1021/ac202897u
Li T, Liu D, Wang Z (2009) Microarray-based Raman spectroscopic assay for kinase inhibition by gold nanoparticle probes. Biosens Bioelectron 24(11):3335–3339. doi:10.1016/j.bios.2009.04.033
Inamori K, Kyo M, Nishiya Y, Inoue Y, Sonoda T, Kinoshita E, Koike T, Katayama Y (2005) Detection and quantification of on-chip phosphorylated peptides by surface plasmon resonance imaging techniques using a phosphate capture molecule. Anal Chem 77(13):3979–3985. doi:10.1021/ac050135t
Shan J, Ma Z (2017) A review on amperometric immunoassays for tumor markers based on the use of hybrid materials consisting of conducting polymers and noble metal nanomaterials. Microchim Acta. doi:10.1007/s00604-00017-02146-y
Kavosi B, Hallaj R, Teymourian H, Salimi A (2014) Au nanoparticles/PAMAM dendrimer functionalized wired ethyleneamine-viologen as highly efficient interface for ultra-sensitive α-fetoprotein electrochemical immunosensor. Biosens Bioelectron 59:389–396. doi:10.1016/j.bios.2014.03.049
Yin H, Xu Z, Zhou Y, Wang M, Ai S (2013) An ultrasensitive electrochemical immunosensor platform with double signal amplification for indole-3-acetic acid determinations in plant seeds. Analyst 138(6):1851–1857. doi:10.1039/c3an36526c
Arduini F, Cinti S, Scognamiglio V, Moscone D (2016) Nanomaterials in electrochemical biosensors for pesticide detection: advances and challenges in food analysis. Microchim Acta 183(7):2063–2083. doi:10.1007/s00604-016-1858-8
Amouzadeh Tabrizi M, Shamsipur M, Saber R, Sarkar S, Zolfaghari N (2017) An ultrasensitive sandwich-type electrochemical immunosensor for the determination of SKBR-3 breast cancer cell using rGO-TPA/FeHCFnano labeled anti-HCT as a signal tag. Sensors Actuators B Chem 243:823–830. doi:10.1016/j.snb.2016.12.061
Haji-Hashemi H, Norouzi P, Safarnejad MR, Ganjali MR (2017) Label-free electrochemical immunosensor for direct detection of Citrus tristeza virus using modified gold electrode. Sensors Actuators B Chem 244:211–216. doi:10.1016/j.snb.2016.12.135
Boujday S, Briandet R, Salmain M, Herry J-M, Marnet P-G, Gautier M, Pradier C-M (2008) Detection of pathogenic Staphylococcus aureus bacteria by gold based immunosensors. Microchim Acta 163(3):203. doi:10.1007/s00604-008-0024-3
Fei J, Dou W, Zhao G (2016) Amperometric immunoassay for the detection of Salmonella pullorum using a screen - printed carbon electrode modified with gold nanoparticle-coated reduced graphene oxide and immunomagnetic beads. Microchim Acta 183(2):757–764. doi:10.1007/s00604-015-1721-3
Hasanzadeh M, Shadjou N (2017) Advanced nanomaterials for use in electrochemical and optical immunoassays of carcinoembryonic antigen. A Rreview. Microchim Acta 184(2):389–414. doi:10.1007/s00604-016-2066-2
Liu X, Li Y, Xu X, Li P, Nie Z, Huang Y, Yao S (2014) Nanomaterial-based tools for protein kinase bioanalysis. TrAC Trends Anal Chem 58:40–53. doi:10.1016/j.trac.2014.01.009
Saha K, Agasti SS, Kim C, Li X, Rotello VM (2012) Gold nanoparticles in chemical and biological sensing. Chem rev 112(5):2739–2779. doi:10.1021/cr2001178
Wang J (2012) Electrochemical biosensing based on noble metal nanoparticles. Microchim Acta 177(3):245–270. doi:10.1007/s00604-011-0758-1
Zhang S, Huang N, Lu Q, Liu M, Li H, Zhang Y, Yao S (2016) A double signal electrochemical human immunoglobulin G immunosensor based on gold nanoparticles-polydopamine functionalized reduced graphene oxide as a sensor platform and AgNPs/carbon nanocomposite as signal probe and catalytic substrate. Biosens Bioelectron 77:1078–1085. doi:10.1016/j.bios.2015.10.089
Liang R-P, Xiang C-Y, Zhao H-F, Qiu J-D (2014) Highly sensitive electrogenerated chemiluminescence biosensor in profiling protein kinase activity and inhibition using a multifunctional nanoprobe. Anal Chim Acta 812:33–40. doi:10.1016/j.aca.2013.12.037
Liu J, He X, Wang K, He D, Wang Y, Mao Y, Shi H, Wen L (2015) A highly sensitive electrochemiluminescence assay for protein kinase based on double-quenching of graphene quantum dots by G-quadruplex-hemin and gold nanoparticles. Biosens Bioelectron 70:54–60. doi:10.1016/j.bios.2015.03.026
Wang Z, Yan Z, Sun N, Liu Y (2015) Multiple signal amplification electrogenerated chemiluminescence biosensors for sensitive protein kinase activity analysis and inhibition. Biosens Bioelectron 68:771–776. doi:10.1016/j.bios.2015.02.006
Xu S, Liu Y, Wang T, Li J (2010) Highly sensitive electrogenerated chemiluminescence biosensor in profiling protein kinase activity and inhibition using gold nanoparticle as signal transduction probes. Anal Chem 82(22):9566–9572
Yin H, Sun B, Dong L, Li B, Zhou Y, Ai S (2015) A signal "on" photoelectrochemical biosensor for assay of protein kinase activity and its inhibitor based on graphite-like carbon nitride, Phos-tag and alkaline phosphatase. Biosens Bioelectron 64:462–468. doi:10.1016/j.bios.2014.09.070
Li X, Zhou Y, Xu Y, Xu H, Wang M, Yin H, Ai S (2016) A novel photoelectrochemical biosensor for protein kinase activity assay based on phosphorylated graphite-like carbon nitride. Anal Chim Acta 934:36–43. doi:10.1016/j.aca.2016.06.024
Yin H, Wang M, Li B, Yang Z, Zhou Y, Ai S (2015) A sensitive electrochemical biosensor for detection of protein kinase a activity and inhibitors based on Phos-tag and enzymatic signal amplification. Biosens Bioelectron 63:26–32. doi:10.1016/j.bios.2014.07.016
Wang Z, Sun N, He Y, Liu Y, Li J (2014) DNA assembled gold nanoparticles polymeric network blocks modular highly sensitive electrochemical biosensors for protein kinase activity analysis and inhibition. Anal Chem 86(12):6153–6159. doi:10.1021/ac501375s
Liu J, He X, Wang K, Wang Y, Yan G, Mao Y (2014) Amplified electrochemical detection of protein kinase activity based on gold nanoparticles/multi-walled carbon nanotubes nanohybrids. Talanta 129:328–335. doi:10.1016/j.talanta.2014.05.043
Shen C, Xia X, Hu S, Yang M, Wang J (2015) Silver nanoclusters-based fluorescence assay of protein kinase activity and inhibition. Anal Chem 87(1):693–698. doi:10.1021/ac503492k
Zhou J, Xu X, Liu X, Li H, Nie Z, Qing M, Huang Y, Yao S (2014) A gold nanoparticles colorimetric assay for label-free detection of protein kinase activity based on phosphorylation protection against exopeptidase cleavage. Biosens Bioelectron 53:295–300. doi:10.1016/j.bios.2013.09.070
Acknowledgements
This work was supported by China Postdoctoral Science Foundation (No. 2014 M550369), the National Natural Science Foundation of China (No. 21375079), the Natural Science Foundation of Shandong province, China (No. ZR2014BQ029), and the Project of Development of Science and Technology of Shandong Province, China (No. 2013GZX20109).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
The author(s) declare that they have no competing interests.
Electronic supplementary material
ESM 1
(DOC 2.83 mb)
Rights and permissions
About this article
Cite this article
Zhou, Y., Wang, M., Yin, H. et al. Amperometric determination of the activity of protein kinase a using a glassy carbon electrode modified with IgG functionalized gold nanoparticles conjugated to horseradish peroxidase. Microchim Acta 184, 3301–3308 (2017). https://doi.org/10.1007/s00604-017-2341-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00604-017-2341-x