Abstract
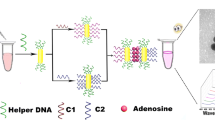
The authors describe a fluorescence based aptasensor for adenosine (AD), a conceivable biomarker for cancer. The assay is based on the immobilization of capture DNA on newly synthesized quaternary CuInZnS quantum dots (QDs) and the conjugation of probe DNA on gold nanoparticles (AuNPs). The capture DNA is an adenosine-specific aptamer that is partly complementary to the probe DNA. Once the capture aptamer hybridizes probe DNA, the fluorescence of the QDs (measured at excitation/emission wavelengths of 522/650 nm) is quenched by the AuNPs. However, when AD is added, it will bind to the aptamer and restrain the hybridization between capture DNA and probe DNA. Therefore, the fluorescence of the QDs will increase with increasing AD concentration. Under optimal conditions, fluorescence is linearly related to the AD concentration in the range from 50 to 400 μM, the detection limit being 1.1 μM. This assay is sensitive, selective, reproducible and acceptably stable. It was applied to the determination of AD in spiked human serum samples where it gave satisfactory results.

Aptamer based fluorescent assay of adenosine using quaternary CuInZnS quantum dots and gold nanoparticles






Similar content being viewed by others
References
Yan XL, Cao ZJ, Kai M, Lua JZ (2009) Label-free aptamer-based chemiluminescence detection of adenosine. Talanta 79:383–387
Dunwiddie TV, Masino SA (2001) The role and regulation of adenosine in the central nervous system. Annu Rev Neurosci 24:31–55
Huang HL, Shi S, Gao X, Gao RR, Zhu Y, Wu XW, Zang RM, Yao TM (2016) A universal label-free fluorescent aptasensor based on Ru complex and quantum dots for adenosine, dopamine and 17β-estradiol detection. Biosens Bioelectron 79:198–204
Yang TX, Guo XY, Wu YP, Wang H, Fu SY, Wen Y, Yang HF (2014) Facile and label-free detection of lung cancer biomarker in urine by magnetically assisted surface-enhanced Raman scattering. Appl Mater Interfaces 6:20985–20993
Zheng YF, Xu GW, Liu DY, Xiong JH, Zhang PD, Zhang C, Yang Q, Lv S (2002) Study of urinary nucleosides as biological marker in cancer patients analyzed by micellar electrokinetic capillary chromatography. Electrophoresis 23:4104–4109
Xue XF, Wang F, Zhou JH, Chen F, Li Y, Zhao J (2009) Online cleanup of accelerated solvent extractions for determination of adenosine 5-triphosphate (ATP), adenosine 5'-diphosphate (ADP), and adenosine 5-monophosphate (AMP) in royal jelly using high-performance liquid chromatography. J Agric Food Chem 57:4500–4505
Tzeng HF, Hung CH, Wang JY, Chou CH, Hung HP (2006) Simultaneous determination of adenosine and its metabolites by capillary electrophoresis as a rapid monitoring tool for 5′-nucleotidase activity. J. Chromatogr A 1129:149–152
Chen JW, Liu XP, Feng KJ, Liang Y, Jiang JH, Shen GL, Yu RQ (2008) Detection of adenosine using surface-enhanced Raman scattering based on structure-switching signaling aptamer. Biosens Bioelectron 24:66–71
Zhang JQ, Wang YS, He Y, Jiang T, Yang HM, Tan X, Kang RH, Yuan YK, Shi LF (2010) Determination of urinary adenosine using resonance light scattering of gold nanoparticles modified structure-switching aptamer. Anal Biochem 397:212–217
Jayasena SD (1999) Aptamers: an emerging class of molecules that rival antibodies in diagnostics. Clin Chem 45:1628–1650
Liu F, Wang SM, Zhang M, Wang YH, Ge SG, Yu JH, Yan M (2014) Aptamer based test stripe for ultrasensitive detection of mercury(II) using a phenylene-ethynylene reagent on nanoporous silver as a chemiluminescence reagent. Microchim Acta 181:663–670
Xing H, Wong NY, Xiang Y, Lu Y (2012) DNA aptamer functionalized nanomaterials for intracellular analysis, cancer cell imaging and drug delivery. Curr Opin Chem Biol 16:429–435
Famulok M, MAYER G (2011) Aptamer modules as sensors and detectors. Acc Chem Res 44:1349–1358
Song PS, Xiang Y, Xing H, Zhou ZJ, Tong AJ, Lu Y (2012) Label-free catalytic and molecular beacon containing an abasic site for sensitive fluorescent detection of small inorganic and organic molecules. Anal Chem 84:2916–2922
Huo Y, Qi L, Lv XJ, Lai T, Zhang J, Zhang ZQ (2016) A sensitive aptasensor for colorimetric detection of adenosine triphosphate based on the protective effect of ATP-aptamer complexes on unmodified gold nanoparticles. Biosens Bioelectron 78:315–320
Wang XZ, Dong SS, Gai PP, Duan R, Li F (2016) Highly sensitive homogeneous electrochemical aptasensor for antibiotic residues detection based on dual recycling amplification strategy. Biosens Bioelectron 82:49–54
Xu WC, Lu Y (2011) A smart magnetic resonance imaging contrast agent responsive to adenosine based on a DNA aptamer-conjugated gadolinium complex. Chem Commun 47:4998–5000
Ma Q, Su XG (2011) Recent advances and applications in QDs-based sensors. Analyst 136:4883–2893
Gill R, Zayats M, Willner I (2008) Semiconductor quantum dots for bioanalysis. Angew Chem Int Ed 47:7602–7625
Liu SY, Shi FP, Zhao XJ, Chen L, Su XG (2013) 3-Aminophenyl boronic acid-functionalized CuInS2 quantum dots as a near-infrared fluorescence probe for the determination of dopamine. Biosens Bioelectron 47:379–384
Liu ZP, Ma Q, Wang XY, Lin ZH, Zhang H, Liu LL, Su XG (2014) A novel fluorescent nanosensor for detection of heparin and heparinase based on CuInS quantum dots. Biosens Bioelectron 54:617–622
Huizenga DE, Szostak JW (1995) A DNA aptamer that binds adenosine and ATP. Biochemistry 34:656–665
Liu LL, Wang XY, Ma Q, Lin ZH, Chen SF, Li Y, Lu LH, Qu HP, Su XG (2016) Multiplex electrochemiluminescence DNA sensor for determination of hepatitis B virus and hepatitis C virus based on multicolor quantum dots and Au nanoparticles. Anal Chim Acta 916:92–101
Liu LL, Ma Q, Li Y, Liu ZP, Su XG (2015) A novel signal-off electrochemiluminescence biosensor for the determination of glucose based on double nanoparticles. Biosens Bioelectron 63:519–524
Saha K, Agasti SS, Kim C, Li X, Rotello VM (2012) Gold nanoparticles in chemical and biological sensing. Chem Rev 112:2739–2779
Sekiguchi S, Niikura K, Iyo N, Matsuo Y, Eguchi A, Nakabayashi T, Ohta N, Ijiro K (2011) pH-dependent network formation of quantum dots and fluorescent quenching by Au nanoparticle embedding. Appl Mater Interfaces 3:4169–4173
Chen GW, Song F, Xiong XQ, Peng XJ (2013) Fluorescent nanosensors based on fluorescence resonance energy transfer (FRET). Ind Eng Chem Res 52:11228–11245
Shi JY, Chan CY, Pang YT, Ye WW, Tian F, Lyu J, Zhang Y, Yang M (2015) A fluorescence resonance energy transfer (FRET) biosensor based on graphene quantum dots (GQDs) and gold nanoparticles (AuNPs) for the detection of mecA gene sequence of Staphylococcus aureus. Biosens Bioelectron 67:595–600
Zhu X, Zhang YS, Yang WQ, Liu QD, Lin ZY, Qiu B, Chen GN (2011) Highly sensitive electrochemiluminescent biosensor for adenosine based on structure-switching of aptamer. Anal Chim Acta 684:121–125
Zhu Y, Hu XC, Shi S, Gao RR, Huang HL, Zhu YY, Lv XY, Yao TM (2016) Ultrasensitive and universal fluorescent aptasensor for the detection of biomolecules (ATP, adenosine and thrombin) based on DNA/Ag nanoclusters fluorescence light-up system. Biosens Bioelectron 79:205–212
Huang YF, Chang HT (2007) Analysis of adenosine triphosphate and glutathione through gold nanoparticles assisted laser desorption/ionization mass spectrometry. Anal Chem 79:4852–4859
Li F, Zhang J, Cao XN, Wang LH, Li D, Song SP, Ye BC, Fan CH (2009) Adenosine detection by using gold nanoparticles and designed aptamer sequences. Analyst 134:1355–1360
Chen Z, Li G, Zhang L, Jiang JF, Li Z, Peng ZH, Deng L (2008) A new method for the detection of ATP using a quantum-dot-tagged aptamer. Anal Bioanal Chem 392:1185–1188
Gao H, Duan Y, Xi M, Sun W (2011) Voltammetric detection of guanosine and adenosine using a carbon paste electrode modified with 1-ethyl-3-thylimidazolium ethylsulfate. Microchim Acta 172:57–64
Acknowledgements
The authors gratefully acknowledge financial support from the National Natural Science Foundation of China (Nos. 21275063 and 21005029) and Youth Science Fund of Jilin Province (20140520081JH).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
The author(s) declare that they have no competing interests.
Electronic supplementary material
ESM 1
(DOCX 2.25 mb)
Rights and permissions
About this article
Cite this article
Chen, X., Chen, S., Hu, T. et al. Fluorescent aptasensor for adenosine based on the use of quaternary CuInZnS quantum dots and gold nanoparticles. Microchim Acta 184, 1361–1367 (2017). https://doi.org/10.1007/s00604-017-2128-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00604-017-2128-0