Abstract
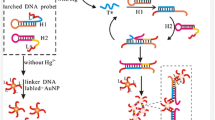
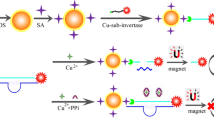
We report on a simple and sensitive protocol for quantitative assay of Hg(II) ions. The process involves the following steps: A thymine (T)-rich single-stranded DNA1 is used to modify the surface of a well of a microplate by the use of biotin-avidin interface chemistry. In parallel, clusters of gold microspheres with a core containing the enzyme invertase were prepared and modified, via thiol chemistry, with a second T-rich oligomer (DNA2). If the gold clusters incorporating invertase and carrying DNA2 are added to the DNA1 immobilized in the wells of the microplate, no interaction will occur and the gold cluster will be removed in the subsequent washing step. If, however, Hg(II) is present in the sample, the DNA on the gold clusters incorporating the enzyme invertase will bind to the DNA in the wells due to the formation of strong T-Hg(II)-T links between the two DNA strands. The encapsulated invertase will not be removed in the following washing step. Sucrose is added in the next step along with invertase which will hydrolyze it to form glucose and fructose. The quantity of glucose formed increases with the quantity of Hg(II) ions present in the sample. The glucose generated is then quantified by using a commercial personal glucometer. Under optimal conditions, the signal for glucose increases with Hg(II) concentration in the range from 0.05 to 80 nM, and the detection limit is as low as 10 pM. The assay has good repeatability and shows an intermediate precision of down to 7.5 %. The method is highly specific for Hg(II) over other metal ions. It was applied to the determination of Hg(II) in naturally contaminated sewage and in spiked samples of drinking water. This approach has a wide scope of application in that it may be extended to numerous other kinds of nanoparticles, oligomer interactions, enzymes and ions.

A simple and sensitive sensing protocol was designed for the quantitative monitoring of mercury(II) using a low-cost microplate with a glucometer readout using invertase encapsulated in a nanogold microsphere for signal amplification.




Similar content being viewed by others
References
Jiang Y, Tian J, Hu K, Zhao Y, Zhao S (2014) Sensitive aptamer-based fluorescence polarization assay for mercury(II) ions and cysteine using silver nanoparticles as a signal amplifier. Microchim Acta 181:1423–1430
Guo Y, Zhang L, Zhang S, Yang Y, Chen X, Zhang M (2015) Fluorescent carbon nanoparticles for the fluorescent detection of metal ions. Biosens Bioelectron 63:61–71
Kang Y, Wu T, Liu B, Wang X, Du Y (2014) Selective determination of mercury(II) by self-referenced surface-enhanced Raman scattering using dialkyne-modified silver nanoparticles. Microchim Acta 181:1333–1339
Botasini S, Heijo G, Mendez E (2013) Toward decentralized analysis of mercury (II) in real samples. A critical review on nanotechnology-based methodologies. Anal Chim Acta 800:1–11
Ma P, Liang F, Yang Q, Wang D, Sun Y, Wang X, Gao D, Song D (2014) Highly sensitive SERS probe for mercury(II) using cyclodextrin-protected silver nanoparticles functionalized with methimazole. Microchim Acta 181:975–981
Liu F, Wang S, Zhang M, Wang Y, Ge S, Yu J, Yan M (2014) Aptamer based test stripe for ultrasensitive detection of mercury(II) using a phenylene-ethynylene reagent on nanoporous silver as a chemiluminescence reagent. Microchim Acta 181:663–670
Ding H, Li H, Liu P, Hitunen J, Wu Y, Chen Z, Shen J (2014) Templated in-situ synthesis of gold nanoclusters conjugated to drug target bacterial enoyl-ACP reductase, and their application to the detection of mercury ions using a test stripe. Microchim Acta 181:1029–1034
Wan J, Ma X, Xing L (2013) Highly sensitive fluorescence detection of mercury (II) ions based on DNA machine amplification. Sens Actu B 178:615–620
Liu H, Xiang Y, Lu Y, Crooks R (2012) Aptamer-based origami paper analytical device for electrochemical detection of adenosine. Angew Chem Int Ed 51:6925–6928
Meier R, Simbuurger J, Soukka T, Schauferling M (2014) Background-free referenced luminescence sensing and imaging of pH using upconverting phosphors and color camera read-out. Anal Chem 86:5535–5540
Wang Y, Lu M, Zhu J, Tian S (2014) Wrapping DNA-gated mesoporous silica nanoparticles for quantitative monitoring of telomerase activity with glucometer readout. J Mater Chem B 2:5847–5853
Chen J, Wu W, Zeng L (2014) A universal glucometer-based biosensor for portable and quantitative detection of transcription factors. Anal Methods 6:4840–4844
Fu X, Feng X, Xu K, Huang R (2014) A portable and quantitative enzyme immunoassay of neuron-specific enolase with a glucometer readout. Anal Methods 6:2233–2238
Hou L, Zhu C, Wu X, Chen G, Tang D (2014) Bioresponsive controlled release from mesoporous silica nanocontainers with glucometer readout. Chem Commun 50:1441–1443
Xuan F, Luo X, Hsing I (2013) Conformation-dependent exonuclease III activity mediated by metal ions reshuffling on thymine-rich DNA duplexes for an ultrasensitive electrochemical method for Hg2+ detection. Anal Chem 85:4586–4593
Gao L, Lian C, Zhou Y, Yan L, Li Q, Zhang C, Chen L, Chen K (2014) Graphene oxide-DNA based sensors. Biosens Bioelectron 60:22–29
Chen G, Chen W, Yen Y, Wang C, Chang H, Chen C (2014) Detection of mercury(II) ions using colorimetric gold nanoparticles on paper-based analytical devices. Anal Chem 86:6843–6849
Cui Y, Chen H, Tang D, Yang H, Chen G (2012) Au(III)-promoted polyaniline gold nanospheres with electrocatalytic recycling of self-produced reactants for signal amplification. Chem Commun 48:10307–10309
Huang J, Cheng Y, Liu Y, Li K, Xian Y, Jin L (2009) Gold nanolabels for enhanced electrochemical immunoanalysis of Escherichia coli. Acta Chim Sin 67:2329–2334
Ambrosi A, Castaneda M, Killard A, Smyth M, Alegret S, Merkoci A (2007) Double-codified gold nanolabels for enhanced immunoanalysis. Anal Chem 79:5232–5240
Tang D, Ren J (2008) In situ amplified electrochemical immunoassay for carcinoembryonic antigen using horseradish peroxidase-encapsulated nanogold hollow microspheres as labels. Anal Chem 80:8064–8070
Tang J, Tang D, Niessner R, Chen G, Knopp D (2011) Magneto-controlled graphene immunosensing platform for simultaneous multiplexed electrochemical immunoassay using distinguishable signal tags. Anal Chem 83:5407–5414
Zargoosh K, Babadi F (2015) Highly selective and sensitive optical sensor for determination of Pb2+ and Hg2+ ions based on the covalent immobilization of dithizone on agarose membrane. Spectrochim Acta A 137:105–110
Tan H, Li Q, Ma C, Song Y, Xu F, Chen S, Wang L (2015) Lanthanide based dual-emission fluorescent probe for detection of mercury (II) in milk. Biosens Bioelectron 63:566–571
Cui X, Zhu L, Wu J, Hou Y, Wang P, Wang Z, Yang M (2015) A fluorescent biosensor based on carbon dots-labeled oligodeoxyribonucleotide and graphene oxide for mercury (II) detection. Biosens Bioelectron 63:506–512
Nidya M, Umadevi M, Rajkumar B (2014) Structural, morphological and optical studies of L-cysteine modified silver nanoparticles and its application as a probe for the selective colorimetric detection of Hg2+. Spectrochim Acta A 133:265–271
Triathi R, Gupta R, Singh P, Bhadwal A, Shrivastav A, Kumar N, Shrivastav B (2014) Ultra-sensitive detection of mercury (II) ions in water sample using gold nanoparticles synthesized by Trichoderma harzianum and their mechanistric approach. Sens Actu B 204:637–646
Zhu M, Wang Y, Deng Y, Yao L, Adeloju S, Pan D, Xue F, Wu Y, Zheng L, Chen W (2014) Ultrasensitive detection of mercury with a novel one-step signal amplified lateral flow strip based on gold nanoparticle-labeled ssDNA recognition and enhancement probes. Biosens Bioelectron 61:14–20
Zhou Q, Xing A, Zhao K (2014) Simultaneous determination of nickel, cobalt and mercury ions in water samples by solid phase extraction using multiwalled carbon nanotubes as adsorbent after chelating with sodium diethyldithiocarbamate prior to high performance liquid chromatography. J Chromatogr A 1360:76–81
Tang S, Tong P, Liu W, Chen J, Yang Z, Zhang L (2014) A novel label-free electrochemical sensor for Hg2+ based on the catalytic formation of metal nanoparticle. Biosens Bioelectron 59:1–5
Ksu K, Lee C, Tseng W, Chao Y, Huang Y (2014) Selective and eco-friendly method for determination of mercury(II) ions in aqueous samples using an on-line AuNPs-PDMS composite microfluidic device/ICP-MS system. Talanta 128:408–413
Acknowledgments
Support by the National Natural Science Foundation of China (grant nos. 41176079 & 21475025), the National Science Foundation of Fujian Province (grant no. 2014 J0105), and the Program for Changjiang Scholars and Innovative Research Team in University (grant no. IRT1116) is gratefully acknowledged.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Zhang, J., Tang, Y., Lv, J. et al. Glucometer-based quantitative determination of Hg(II) using gold particle encapsulated invertase and strong thymine-Hg(II)-thymine interaction for signal amplification. Microchim Acta 182, 1153–1159 (2015). https://doi.org/10.1007/s00604-014-1437-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00604-014-1437-9