Abstract
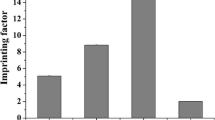
A comparative study was performed on different polymeric formats for targeting corticosteroids, focusing on the use of bulk monolith and precipitation polymerisation strategies. Hydrocortisone-17-butyrate was selected as the template and methacrylic acid as the functional monomer, following 1H NMR investigation of the pre-polymerisation mixture. Three different cross-linkers were tested, ranging from moderate to highly hydrophobic. The synthesised bulk and precipitated imprinted polymers were physically characterised by nitrogen sorption and evaluated by means of HPLC and frontal chromatography against a range of template analogues. While some degree of selectivity for the template was achieved for all tested polymers, the ones based on the tri-functional cross-linking monomer trimethylolpropane trimethacrylate exhibited the longest retention for all corticosteroids, especially in the precipitated format, which suggested broader group selectivity.








Similar content being viewed by others
References
Sellergren B (2001) Molecularly imprinted polymers. Man made mimics of antibodies and their applications in analytical chemistry, vol 23. Techniques and instrumentation in analytical chemistry. Elsevier Science B.V, Amsterdam
Steinke J, Sherrington D, Dunkin I (1995) Imprinting of synthetic polymers using molecular templates. In: Advances in polymer science, vol 123. p 81
Zhu T, Li SN, Row KH (2011) Molecularly imprinted monolithic material for the extraction of three organic acids from salicornia herbacea L. J Appl Polym Sci 121:1691
Manesiotis P, Osmani Q, McLoughlin P (2012) An enantio-selective chromatographic stationary phase for S-ibuprofen prepared by stoichiometric molecular imprinting. Journal of Materials Chemistry 22:11201
Yu ZR, Yang JJ, Zhong JF, Wu SQ, Xu ZG, Tang YW (2012) Emodin voltammetric sensor based on molecularly imprinted polymer membrane-modified electrode using a multiple hydrogen bonds strategy. J Appl Polym Sci 126:1344
Li SJ, Ge Y, Turner APF (2011) A catalytic and positively thermosensitive molecularly imprinted polymer. Adv Funct Mater 21:1194
Javanbakht M, Mohammadi S, Esfandyari-Manesh M, Abdouss M (2011) Molecularly imprinted polymer microspheres with nanopore cavities prepared by precipitation polymerization as new carriers for the sustained release of dipyridamole. J Appl Polym Sci 119:1586
Frois C, Wu EQ, Ray S, Colice GL (2009) Inhaled corticosteroids or long-acting [beta]-agonists alone or in fixed-dose combinations in asthma treatment: a systematic review of fluticasone/budesonide and formoterol/salmeterol. Clin Ther 31(12):2779
Rivkin A (2009) Certolizumab pegol for the management of Crohn’s disease in adults. Clin Ther 31:1158
Kim J, Peng C-C, Chauhan A (2010) Extended release of dexamethasone from silicone-hydrogel contact lenses containing vitamin E. J Control Release 148:110
MacMahon PJ, Eustace SJ, Kavanagh EC (2009) Injectable corticosteroid and local anesthetic preparations: a review for radiologists. Radiology 252:647
Yao R, Wang Y, Lemon WJ, Lubet RA, You M (2004) Budesonide exerts its chemopreventive efficacy during mouse lung tumorigenesis by modulating gene expressions. Oncogene 23:7746
Donihi AC, Raval D, Saul M, Korytkowski MT, DeVita MA (2006) Prevalence and predictors of corticosteroid-related hyperglycemia in hospitalized patients. Endocr Pract 12:358
Hougardy DM, Peterson GM, Bleasel MD, Randall CT (2000) Is enough attention being given to the adverse effects of corticosteroid therapy? J Clin Pharm Ther 25:227
96/22/EC CD (1996) Off J Eur Commun, vol L125
Ramstrom O, Ye L, Mosbach K (1996) Artificial antibodies to corticosteroids prepared by molecular imprinting. Chem Biol 3:471
Ramstrom O, Ye L, Krook M, Mosbach K (1998) Screening of a combinatorial steroid library using molecularly imprinted polymers. Anal Commun 35:9
Sreenivasan K (1997) Application of molecularly imprinted polymer as a drug retaining matrix. Angew Makromol Chem 246:65–69
Sreenivasan K (1999) On the application of molecularly imprinted poly(HEMA) as a template responsive release system. J Appl Polym Sci 71:1819
Baggiani C, Giraudi G, Trotta F, Giovannoli C, Vanni A (2000) Chromatographic characterization of a molecular imprinted polymer binding cortisol. Talanta 51:71
Baggiani C, Baravalle P, Giovannoli C, Anfossi L, Giraudi G (2010) Molecularly imprinted polymers for corticosteroids: analysis of binding selectivity. Biosens Bioelectron 26:590
Diaz-Bao M, Barreiro R, Regal P, Cepeda A, Fente C (2012) Evaluation of molecularly imprinted polymers for the simultaneous SPE of six corticosteroids in milk. Chromatographia 75:223
Hall AJ, Quaglia M, Manesiotis P, De Lorenzi E, Sellergren B (2006) Polymeric receptors for the recognition of folic acid and related compounds via substructure imprinting. Anal Chem 78:8362
Rampey AM, Umpleby RJ, Rushton GT, Iseman JC, Shah RN, Shimizu KD (2004) Characterization of the imprint effect and the influence of imprinting conditions on affinity, capacity, and heterogeneity in molecularly imprinted polymers using the Freundlich isotherm-affinity distribution analysis. Anal Chem 76:1123
Wang JF, Cormack PAG, Sherrington DC, Khoshdel E (2007) Synthesis and characterization of micrometer-sized molecularly imprinted spherical polymer particulates prepared via precipitation polymerization. Pure Appl Chem 79:1505
Yoshimatsu K, Reimhult K, Krozer A, Mosbach K, Sode K, Ye L (2007) Uniform molecularly imprinted microspheres and nanoparticles prepared by precipitation polymerization: the control of particle size suitable for different analytical applications. Anal Chim Acta 584:112
Li K, Stover HDH (1993) Synthesis of monodisperse poly(Divinylbenzene) microspheres. J Polym Sci Pol Chem 31(13):3257–3263
Li WH, Stover HDH (1998) Porous monodisperse poly(divinylbenzene) microspheres by precipitation polymerization. J Polym Sci Pol Chem 36(10):1543–1551
Oxelbark J, Legido-Quigley C, Aureliano CSA, Titirici M-M, Schillinger E, Sellergren B, Courtois J, Irgum K, Dambies L, Cormack PAG, Sherrington DC, De Lorenzi E (2007) Chromatographic comparison of bupivacaine imprinted polymers prepared in crushed monolith, microsphere, silica-based composite and capillary monolith formats. J Chromatogr A 1160:215
Urraca JL, Carbajo MC, Torralvo MJ, González-Vázquez J, Orellana G, Moreno-Bondi MC (2008) Effect of the template and functional monomer on the textural properties of molecularly imprinted polymers. Biosens Bioelectron 24:155
Spivak DA, Shea KJ (2001) Investigation into the scope and limitations of molecular imprinting with DNA molecules. Anal Chim Acta 435:65
Sing KSW (1985) Reporting physisorption data for gas/solid systems with special reference to the determination of surface area and porosity (Recommendations 1984). Pure Appl Chem 57:603
Li WH, Stover HDH (1998) Porous monodisperse poly(divinylbenzene) microspheres by precipitation polymerization. J Polym Sci Pol Chem 36:1543
Kempe M, Mosbach K (1995) Receptor binding mimetics: a novel molecularly imprinted polymer. Tet Lett 36:3563
Acknowledgments
The authors gratefully acknowledge the funding provided by Enterprise Ireland, under the Technology Gateway Programme, the Irish Research Council for Science, Engineering and Technology, under the Embark Initiative and the Technological Sector Research under the Strand III programme.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
ESM 1
(PDF 36 kb)
Rights and permissions
About this article
Cite this article
Fitzhenry, L., Manesiotis, P., Duggan, P. et al. Molecularly imprinted polymers for corticosteroids: impact of polymer format on recognition behaviour. Microchim Acta 180, 1421–1431 (2013). https://doi.org/10.1007/s00604-013-1034-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00604-013-1034-3