Abstract
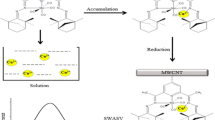
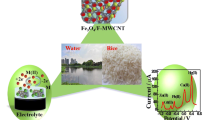
A new composite electrode is described for anodic stripping voltammetry determination of Pb(II) at trace level in aqueous solution. The electrode is based on the use of multiwalled carbon nanotubes and Amberlite IR-120. The anodic stripping voltammograms depend, to a large extent, on the composition of the modified electrode and the preconcentration conditions. Under optimum conditions, the anodic peak current at around −0.57 V is linearly related to the concentration of Pb(II) in the range from 9.6 × 10−8 to 1.7 × 10−6 mol L−1 (R = 0.998). The detection limit is 2.1 × 10−8 mol L−1, and the relative standard deviation (RSD) at 0.24 × 10−6 mol L−1 is 1.7% (n = 6). The modified electrode was applied to the determination of Pb(II) using the standard addition method; the results showed average relative recoveries of 95% for the samples analysed.

A new composite electrode is described for anodic stripping voltammetry determination of Pb(II) at trace level in aqueous solution. The electrode is based on the use of MWCNT and Amberlite IR-120. The method showed a good linearity for 9.6 × 10−8 - 1.7 × 10−6 mol L−1 and detection limit of 2.1 × 10−8 mol L−1.




Similar content being viewed by others
References
Yáñez-Sedeño P, Riu J, Pingarrón JM, Rius FX (2010) Electrochemical sensing based on carbon nanotubes. Trends Anal Chem 29:939–953
Sun D, Wan C, Li G, Wu K (2007) Electrochemical determination of lead (II) using a montmorillonite calcium-modified carbon paste electrode. Microchim Acta 158:255–260
Majid S, Rhazi EM, Amine A, Curulli A, Palleschi G (2003) carbon paste electrode bulk-modified with the conducting oolymer poly(1,8-diaminonaphthalene): Application to lead determination. Microchim Acta 143:195–204
Tesarova E, Baldrianova L, Hocevar SB, Svancara I, Vytras K, Ogorevc B (2009) Anodic stripping voltammetric measurement of trace heavy metals at antimony film carbon paste electrode. Electrochim Acta 54:1506–1510
Aroua MK, Yin CY, Lim FN, Kan WL, Daud WMAW (2009) Adsorption of copper from the sulphate solution of low copper contents using the cationic resin Amberlite IR 120. J Hazard Mater 166:1526–1529
Jha MK, Nguyen NV, Lee J, Jeong J, Yoo J (2009) Effect of impregnation of activated carbon with chelating polymer on adsorption kinetics of Pb2+. J Hazard Mater 164:948–953
Rajesh N, Manikandan S (2008) Spectrophotometric determination of lead after preconcentration of its diphenylthiocarbazone complex on an Amberlite XAD-1180 column. Spectrochim Acta part A 70:754–757
Demirbas A, Pehlivan E, Gode F, Altun T, Arslan G (2005) Adsorption of Cu(II), Zn(II), Ni(II), Pb(II), and Cd(II) from aqueous solution on Amberlite IR-120 synthetic resin. J Colloid Interface Sci 282:20–25
Yang G, Qu X, Shen M, Wang C, Qu Q, Hu X (2008) Electrochemical behavior of lead at poly(phenol red) modified glass carbon electrode, and its trace determination by differential pulse anodic stripping voltammetry. Microchim Acta 160:275–281
Mandil A, Idrissi L, Amine A (2010) Stripping voltammetric determination of mercury(II) and lead(II) using screen-printed electrodes modified with gold films, and metal ion preconcentration with thiol-modified magnetic particles. Microchim Acta 170:299–305
Guo J, Luo Y, Ge F, Ding Y (2010) Voltammetric determination of cadmium (II) based on composite film of thiol-functionalized mesoporous molecular sieve and ionic liquid. Microchim Acta. doi:10.1007/s00604-010-0506-y
Mousavi MF, Rahmani A, Golabi SM, Shamsipur M, Sharghi H (2001) Differential pulse anodic stripping voltammetric determination of lead(II) with a 1,4-bis(prop-2-enyloxy)-9,10-anthraquinone modified carbon paste electrode. Talanta 55:305–312
Oswald S, Havel M, Gogotsi Y (2007) Monitoring oxidation of multiwalled carbon nanotubes by Raman spectroscopy. J Raman Spectrosc 38:728–736
Nuget JM, Santhanam KSV, Rubio A, Ajayan PM (2001) Fast electron transfer kinetics on multiwalled carbon nanotube microbundle electrodes. Nano Lett 1:87–91
Wei B, Zhang L, Chen G (2009) A multi-walled carbon nanotube/poly(urea-formaldehyde) composite prepared by in situ polycondensation for enhanced electrochemical sensing. New J Chem 34:453–457
Kurusu F, Tsunod H, Saito A, Tomita A, Kadota A, Kayahara N, Karube I, Gotoh M (2006) The advantage of using carbon nanotubes compared with edge plane pyrolitc graphite as an electrode material for oxidase-based biosensors. Analyst 131:1292–1298
Analytical Methods Committee of Royal Society of Chemistry (1987) Recommendations for the definition, estimation and use of the detection limit. Analyst. doi:10.1039/AN9871200199
Tsai Y-C, Chen J-M, Marken F (2005) Simple cast-deposited multi-walled carbon nanotube/Nafion thin film electrodes for electrochemical stripping analysis. Microchim Acta 150:269–276
Injang U, Noyrod P, Siangproh W, Dungchai W, Motomizu S, Chailapakul O (2008) Electrochemical determination of Pb2+ using carbon nanotube/Nafion composite film-modified electrode. J Appl Electrochem 668:54–60
Tarley CRT, Santos VS, Baeta BEL, Pereira AC, Kubota LT (2009) Simultaneous determination of zinc, cadmium and lead in environmental water samples by potentiometric stripping analysis (PSA) using multiwalled carbon nanotube electrode. J Hazard Mater 169:256–262
Hwang GH, Han WK, Park JS, Kang SG (2008) Determination of trace metals by anodic stripping voltammetry using a bismuth-modified carbon nanotube electrode. Talanta 76:301–308
Injang U, Noyrod P, Siangproh W, Dungchai W, Motomizu S, Chailapakul O (2010) Determination of trace heavy metals in herbs by sequential injection analysis-anodic stripping voltammetry using screen-printed carbon nanotubes electrodes. Anal Chim Acta 668:54–60
Wu K, Hu S, Fei J, Bai W (2003) Mercury-free simultaneous determination of cadmium and lead at a glassy carbon electrode modified with multi-wall carbon nanotubes. Anal Chim Acta 489:215–221
Mohadesi A, Motallebi Z, Salmanipour A (2010) Multiwalled carbon nanotube modified with 1-(2-pyridylazo)-2-naphthol for stripping voltammetric determination of Pb (II). Analyst 135:1686–1690
Acknowledgements
This research was supported by the Brazilian Agencies FAPEMIG, FINEP, CNPq, CAPES and by the Brazilian Institute of Nanotechnology (INCT).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
do Nascimento, M.E., Martelli, P.B., Furtado, C.A. et al. Determination of lead(II) in aqueous solution using carbon nanotubes paste electrodes modified with Amberlite IR-120. Microchim Acta 173, 485–493 (2011). https://doi.org/10.1007/s00604-011-0583-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00604-011-0583-6