Abstract
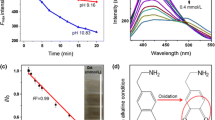
A novel method has been developed for methimazole analysis based on the quenching of fluorescence emission from CdSe quantum dots by methimazole. Under optimum conditions, the calibration graph was linear over the range of 50 nM to 5 μM (r 2 = 0.990). The limit of detection (S/N = 3) was 30 nM. The R.S.D. for ten determinations of 500 nM methimazole was 3.4%. The method was applied to determine methimazole in tablets and rat urine, and the results were satisfactory, i.e. consistent with those of gas chromatography–mass spectrometry. The possible cause of fluorescence quenching is due to the exchange of surface capping organic molecules of quantum dots induced by methimazole.





Similar content being viewed by others
References
Eshghi H, Tayyari SF, Rezvani-Amin Z, Roohi H (2008) Methimazole-disulfide as an anti-thyroid drug metabolite catalyzed the highly regioselective conversion of epoxides to halohydrins with elemental halogens. Bull Korean Chem Soc 29:51
Sułkowska A (2002) Interaction of drugs with bovine and human serum albumin. J Mol Struct 614:227
Kasraee B, Hugin A, Tran C, Sorg O, Saurat JH (2004) Methimazole is an inhibitor of melanin synthesis in cultured B16 melanocytes. J Invest Dermatol 122:1338
Kuśmierek K, Bald E (2007) Determination of methimazole in urine by liquid chromatography. Talanta 71:2121
Wasch KD, Brabander HFB, Impens S, Vandewiele M, Courtheyn D (2001) Determination of mercaptobenzimidazol and other thyreostat residues in thyroid tissue and meat using high-performance liquid chromatography–mass spectrometry. J Chromatogr A 912:311
Zakrzewski R (2008) Determination of methimazole in urine with the iodine-azide detection system following its separation by reversed-phase high-performance liquid chromatography. J Chromatogr B Anal Technol Biomed Life Sci 869:67
Batjoens P, Brabander HFD, Wasch KD (1996) Rapid and high-performance analysis of thyreostatic drug residues in urine using gas chromatography–mass spectrometry. J Chromatogr A 750:127
Aslanoglu M, Peker N (2003) Potentiometric and voltammetric determination of methimazole. J Pharm Biomed Anal 33:1143
Shahrokhian S, Ghalkhani M (2008) Voltammetric determination of methimazole using a carbon paste electrode modified with a Schiff base complex of cobalt. Electroanalysis 20:1061
Economou A, Tzanavaras PD, Notou M, Themelis DG (2004) Determination of methimazole and carbimazole by flow-injection with chemiluminescence detection based on the inhibition of the Cu(II)-catalysed luminol–hydrogen peroxide reaction. Anal Chim Acta 505:129
Liu XL, Yuan H, Pang DW, Cai RX (2004) Resonance light scattering spectroscopy study of interaction between gold colloid and thiamazole and its analytical application. Spectrochim Acta Part A 60:385
Spangler C, Schaeferling M, Wolfbeis OS (2008) Fluorescent probes for microdetermination of inorganic phosphates and biophosphates. Microchim Acta 161:1
Sutherland AJ (2002) Quantum dots as luminescent probes in biological systems. Curr Opin Solid State Mater Sci 6:365
Wu HM, Liang JG, Han HY (2008) A novel method for the determination of Pb2+ based on the quenching of the fluorescence of CdTe quantum dots. Microchim Acta 161:81
Liang JG, Huang S, Zeng DY, He ZK, Ji XH, Ai XP, Yang HX (2006) CdSe quantum dots as luminescent probes for spironolactone determination. Talanta 69:126
Wang YQ, Ye C, Zhu ZH, Hu YZ (2008) Cadmium telluride quantum dots as pH-sensitive probes for tiopronin determination. Anal Chim Acta 610:50
Qu LH, Peng XG (2002) Control of photoluminescence of CdSe nanocrystals in growth. J Am Chem Soc 124:2049
Skellern GG, Steer S (1981) The metabolism of [2–14C]methimazole in the rat. Xenobiotica. 11(9):627–34
Liang JG, Zhang SS, Ai XP, Ji XH, He ZK (2005) The interaction between some diamines and CdSe quantum dots. Spectrochim Acta Part A 61:2974
Yu WW, Qu LH, Guo WZ, Peng XG (2003) Experimental determination of the extinction coefficient of CdTe, CdSe, and CdS nanocrystals. Chem Mater 15:2854
Cao QE, Zhao YK, Yao XJ, Hu ZD, Xu QH (2000) The synthesis and application of 1-(o-nitrophenyl)-3-(2-thiazolyl)triazene for the determination of palladium(II) by the resonance enhanced Rayleigh light-scattering technique. Spectrochim Acta Part A 56:1319
Landi BJ, Evans CM, Worman JJ, Castro SL, Bailey SG, Raffaelle RP (2006) Noncovalent attachment of CdSe quantum dots to single wall carbon nanotubes. Mater Lett 60:3502
Dong F, Han HY, Liang JG, Lu DL (2008) Study on the interaction between 2-mercaptoethanol, dimercaprol and CdSe quantum dots. Luminescence 23:321
Smith E, Dent G (2005) Modern Raman spectroscopy—a practical approach. Wiley, New York
Loo BH, Tse Y, Parsons K, Adelman C, El-Hage A, Lee YG (2006) Surface-enhanced Raman spectroscopy of imidazole adsorbed on electrode and colloidal surfaces of Cu, Ag, and Au. J Raman Spectrosc 37:299
Cao PG, Gu RA, Tian ZQ (2003) Surface-enhanced Raman spectroscopy studies on the interaction of imidazole with a silver electrode in acetonitrile solution. J Phys Chem B 107:769
Krishnakumar V, Xavier RJ (2004) FT Raman and FT-IR spectral studies of 3-mercapto-1, 2, 4-triazole. Spectrochim Acta Part A 60:709
Sharmaa SN, Sharmaa H, Singh G, Shivaprasad SM (2008) Studies of interaction of amines with TOPO/TOP capped CdSe quantum dots: Role of crystallite size and oxidation potential. Mater Chem Phys 110:471
Acknowledgements
The authors gratefully acknowledge the support for this research provided by New Century Excellent Talents in Chinese Ministry of Education (NCET-05-0668), the National Natural Science Foundation of China (20675034), Program for academic pacesetter of Wuhan (200851430484).
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
ESM 1
(DOC 215 KB)
Rights and permissions
About this article
Cite this article
Dong, F., Hu, K., Han, H. et al. A novel method for methimazole determination using CdSe quantum dots as fluorescence probes. Microchim Acta 165, 195–201 (2009). https://doi.org/10.1007/s00604-008-0120-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00604-008-0120-4