Abstract.
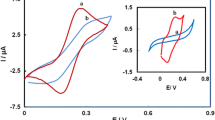
The fabrication and electrochemical characteristics of a penicillamine (PCA) self-assembled monolayer modified gold electrode were investigated. The self-assembled electrode shows obvious electrocatalytic activity for the oxidation of epinephrine (EP). In phosphate buffer (pH 7.73), a sensitive oxidation peak was observed at 0.190 V with the PCA modified Au electrode. The peak current is proportional to the concentration of EP in the range of 2.0 × 10−5 ∼6.0 × 10−4 mol L−1 and 5.0 × 10−6 ∼ 2.0 × 10−4 mol L−1 for cyclic voltammetry (CV) and differential pulse voltammetry (DPV) with the detection limits of 1.8 × 10−7 and 1.3 × 10−7 mol L−1, respectively. The possible reaction mechanism is also discussed. The PCA self-assembled monolayer modified gold electrode is highly stable and can be applied to the determination of EP in practical injection samples. Application is simple, rapid and produces accurate results.
Similar content being viewed by others
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Niu, L., Luo, H. & Li, N. Electrochemical Behavior of Epinephrine at a Penicillamine Self-Assembled Gold Electrode, and its Analytical Application. Microchim Acta 150, 87–93 (2005). https://doi.org/10.1007/s00604-005-0331-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00604-005-0331-x