Abstract.
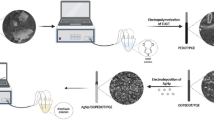
A composite film containing a heteropolyanion was prepared on a 2-aminoethanethiol (AT) self-assembled monolayer film-modified gold electrode by attaching the Keggin-type phosphomolybdic anion. The surface structures and electrochemical properties of the composite films were characterized by using ATR-FTIR, AC impedance, cyclic voltammetry and chronocoulometry. FTIR studies indicated that there was some electrostatic interaction between Pmo12O427− and surface NH3+. Three reversible redox couples were observed in 1.0 mol L−1 H2SO4 in the potential range of 0–0.7 V for the composite film modified electrode, which were attributed to two-electron and two-proton electrochemical processes of PMo12O427−. The diffusion coefficient is determined to be 2.04×10−7 cm2 s−1. The composite film shows good catalytic activities for the reduction of nitrite (NO2−) in acidic solution and the catalytic mechanisms are described. The modified electrode provides a linear response for NO2− in the concentration range of 1.0×10−4 to 1.0×10−7 mol L−1 by differential pulse adsorptive stripping voltammetry with a correlation coefficient of 0.9965. The detection limit (three times the signal blank/slope) was 2.0×10−8 mol L−1. The modified electrode was used for the determination of NO2− in wastewater.
Similar content being viewed by others

Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Wang, S., Sun, Y., Wang, X. et al. Electrochemical Properties and Analytical Applications of Keggin-type Phosphomolybdic Anions in Electrostatically Linked 2-Aminoethanethiol Self-Assembled Monolayers. Microchim Acta 149, 185–191 (2005). https://doi.org/10.1007/s00604-005-0321-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00604-005-0321-z