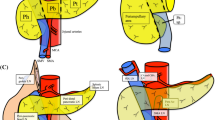
Abstract
Purpose
The optimal range of lymph-node dissection for pancreatic tail cancer remains unclear. We investigated the location and frequency of lymph-node metastases to identify the correct range of lymph-node dissection for pancreatic tail cancer.
Methods
We analyzed clinical data retrospectively, on patients who underwent distal pancreatectomy for resectable left-sided pancreatic cancer, between February, 2006 and March, 2021. Eligible patients were divided into two groups according to the tumor location: those with pancreatic tail cancer (Pt group) and those with pancreatic body or body and tail cancer (non-Pt group).
Results
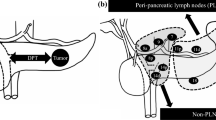
Of the 96 patients analyzed, 61 (64%) were assigned to the Pt group and 35 (36%) were assigned to the non-Pt group. Metastases to stations 7, 8, 9, 10, 11, 14, and 18 were found in 0 (0%), 0 (0%), 0 (0%), 4 (7%), 18 (30%), 2 (4%), and 10 (17%) patients in the Pt group, and in 1 (3%), 4 (12%), 2 (6%), 1 (3%), 18 (51%), 3 (9%), and 6 (17%) patients in the non-Pt group, respectively.
Conclusion
Lymph-node dissection at stations 7, 8, and 9 might not be necessary in patients with resectable pancreatic cancer confined to the pancreatic tail.


Similar content being viewed by others
References
Statistics graph database. Center for Cancer Control and Information Services. https://ganjoho.jp/reg_stat/statistics/stat/summary.html.
Siegel RL, Miller KD, Fuchs HE, Jemal A. Cancer statistics, 2021. CA Cancer J Clin. 2021;71:7–33.
Carioli G, Bertuccio P, Boffetta P, Levi F, La Vecchia C, Negri E, et al. European cancer mortality predictions for the year 2020 with a focus on prostate cancer. Ann Oncol. 2020;31:650–8.
Yamaguchi K, Okusaka T, Shimizu K, Furuse J, Ito Y, Hanada K, et al. Clinical practice guidelines for pancreatic cancer 2016 from the Japan Pancreas Society: a synopsis. Pancreas. 2017;46:595–604.
Takaori K, Bassi C, Biankin A, Brunner TB, Cataldo I, Campbell F, et al. International Association of Pancreatology (IAP)/European Pancreatic Club (EPC) consensus review of guidelines for the treatment of pancreatic cancer. Pancreatology. 2016;16:14–27.
Yamamoto T, Satoi S, Kawai M, Motoi F, Sho M, Uemura K, et al. Is distal pancreatectomy with en-bloc celiac axis resection effective for patients with locally advanced pancreatic ductal adenocarcinoma? Multicenter surgical group study. Pancreatology. 2018;18:106–13.
Wei K, Hackert T. Surgical treatment of pancreatic ductal adenocarcinoma. Cancers (Basel). 2021;13:1971.
Morales-Oyarvide V, Rubinson DA, Dunne RF, Kozak MM, Bui JL, Yuan C, et al. Lymph node metastases in resected pancreatic ductal adenocarcinoma: predictors of disease recurrence and survival. Br J Cancer. 2017;117:1874–82.
Fujita T, Nakagohri T, Gotohda N, Takahashi S, Konishi M, Kojima M, et al. Evaluation of the prognostic factors and significance of lymph node status in invasive ductal carcinoma of the body or tail of the pancreas. Pancreas. 2010;39:e48-54.
Murakami Y, Uemura K, Sudo T, Hayashidani Y, Hashimoto Y, Nakashima A, et al. Number of metastatic lymph nodes, but not lymph node ratio, is an independent prognostic factor after resection of pancreatic carcinoma. J Am Coll Surg. 2010;211:196–204.
Okada K, Murakami Y, Kondo N, Uemura K, Nakagawa N, Seo S, et al. Prognostic significance of lymph node metastasis and micrometastasis along the left side of superior mesenteric artery in pancreatic head cancer. J Gastrointest Surg. 2019;23:2100–9.
Japan Pancreas Society. Description of findings. In: Classifcation of pancreatic carcinoma. 4th English. Tokyo Kanehara & Co Ltd; 2017. p. 12–58.
Kayker S, Pawlik TM, Allen PJ, Vauthey JN. TNM classification of malignant tumors. 8th ed. New York: Wiley; 2018.
Tol JA, Gouma DJ, Bassi C, et al. Definition of a standard lymphadenectomy in surgery for pancreatic ductal adenocarcinoma: a consensus statement by the International Study Group on Pancreatic Surgery (ISGPS). Surgery. 2014;156:591–600.
Zhou Y, Lin J, Wang W, Chen H, Deng X, Peng C, Cheng D, Shen B. Should a standard lymphadenectomy include the No. 9 lymph nodes for body and tail pancreatic ductal adenocarcinoma? Pancreatology. 2019;19:414–8.
Imamura T, Yamamoto Y, Sugiura T, Okamura Y, Ito T, Ashida R, et al. Reconsidering the optimal regional lymph node station according to tumor location for pancreatic cancer. Ann Surg Oncol. 2021;28:1602–11.
Hirashita T, Iwashita Y, Fujinaga A, Nakanuma H, Masuda T, Endo Y, et al. Relationship between the tumor location and clinicopathological features in left-sided pancreatic ductal adenocarcinoma. Surg Today. 2021;51:814–20.
Network NC. NCCN clinical Practice Guidelines in Oncology. Pancreatic Adenocarcinoma. Version 1. 2021 ed 2021. http://www.vccn.org/professionals/physiciangls/pdf/pancreatic.pdf. 2021.
Unno M, Motoi F, Matsuyama Y, Satoi S, Matsumoto I, Aosasa S, et al. Randomized phase II/III trial of neoadjuvant chemotherapy with gemcitabine and S-1 versus upfront surgery for respectable pancreatic cancer (Prep-02/JSAP-05). J Clin Oncol. 2019;37(4 suppl):189.
Strasberg SM, Drebin JA, Linehan D. Radical antegrade modular pancreatosplenectomy. Surgery. 2003;133:521–7.
Kanda M, Fujii T, Nagai S, Kodera Y, Kanzaki A, Sahin TT, et al. Pattern of lymph node metastasis spread in pancreatic cancer. Pancreas. 2011;40:951–5.
Murakami Y, Uemura K, Sudo T, Hashimoto Y, Yuasa Y, Sueda T. Prognostic impact of para-aortic lymph node metastasis in pancreatic ductal adenocarcinoma. World J Surg. 2010;34:1900–7.
Cesmebasi A, Malefant J, Patel SD, Du Plessis M, Renna S, Tubbs RS, et al. The surgical anatomy of the lymphatic system of the pancreas. Clin Anat. 2015;28:527–37.
Nagakawa Y, Sahara Y, Hosokawa Y, Murakami Y, Yamaue H, Satoi S, et al. Clinical impact of neoadjuvant chemotherapy and chemoradiotherapy in borderline resectable pancreatic cancer: analysis of 884 patients at facilities specializing in pancreatic surgery. Ann Surg Oncol. 2019;26:1629–36.
Ocuin LM, Hardacre JM, Ammori JB, Rothermel LD, Mohamed A, Selfridge JE, et al. Neoadjuvant chemotherapy is associated with improved survival in patients with left-sided pancreatic adenocarcinoma. J Surg Oncol. 2020;122:1595–603.
Mokdad AA, Minter RM, Zhu H, Augustine MM, Porembka MR, Wang SC, et al. Neoadjuvant therapy followed by resection versus upfront resection for resectable pancreatic cancer: a propensity score matched analysis. J Clin Oncol. 2017;35:515–22.
de Rooij T, van Hilst J, van Santvoort H, Boerma D, van den Boezem P, Daams F, Dutch Pancreatic Cancer Group, et al. Minimally invasive versus open distal pancreatectomy (LEOPARD): a multicenter patient-blinded randomized controlled trial. Ann Surg. 2019;269:2–9.
Ohtsuka T, Ban D, Nakamura Y, Nagakawa Y, Tanabe M, Gotoh Y, et al. Difficulty scoring system in laparoscopic distal pancreatectomy. J Hepatobiliary Pancreat Sci. 2018;25:489–97.
Wada Y, Aoki T, Murakami M, Fujimori A, Koizumi T, Kusano T, et al. Individualized procedures for splenic artery dissection during laparoscopic distal pancreatectomy. BMC Surg. 2020;20:32.
Deiro G, De Pastena M, Paiella S, Balduzzi A, Montagnini G, Andreotti E, et al. Assessment of difficulty in laparoscopic distal pancreatectomy: a modification of the Japanese difficulty scoring system—a single-center high-volume experience. J Hepatobiliary Pancreat Sci. 2021. https://doi.org/10.1002/jhbp.1010.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
We have no conflicts of interest to declare.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Seo, S., Uemura, K., Sumiyoshi, T. et al. Optimal lymph-node dissection for pancreatic tail cancer. Surg Today 52, 1307–1312 (2022). https://doi.org/10.1007/s00595-022-02463-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00595-022-02463-1