Abstract
Purpose
Postoperative acute kidney injury (AKI) remains a serious complication of liver resection with restrictive fluid therapy. However, unlike open hepatectomy, laparoscopic liver resection (LLR) does not have established anesthesia management strategies. We compared our goal-directed therapy (GDT) protocol for LLR with/without carperitide and the conventional restrictive method regarding AKI prevention.
Methods
The GDT thresholds in this retrospective observational cohort study were as follows: stroke volume variation, ≤ 15%; pulse pressure variation, ≤ 13%; oxygen delivery index, ≥ 600 mL/min/m2; and mean arterial pressure (MAP), ≥ 55 mmHg. If the thresholds were not achieved, a 250 mL infusion fluid bolus was administered. The MAP target was changed to > 65 mmHg if the urine output was < 0.3 mL/kg/h. Postoperative AKI within 48 h and perioperative outcomes within 90 days were analyzed.
Results
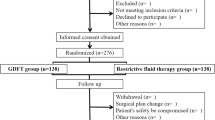
Forty-seven propensity score-matched pairs from 127 patients were investigated. We adjusted for AKI risk factors and surgical difficulty; 46.8% of the GDT group received carperitide. The GDT group had a lower postoperative AKI rate (10.6% vs. 27.7%, P = 0.04) and shorter overall (P = 0.04) and postoperative (P < 0.01) hospital stays than the conventional group. Furthermore, the GDT group received more intraoperative fluid (P = 0.001) and phenylephrine (P = 0.02), without significant increases in blood loss and transfusion volume, than the conventional group.
Conclusions
GDT reduced the AKI rates post-LLR.


Similar content being viewed by others
Data availability
The raw data used in this study are available from the corresponding author upon reasonable request.
References
Katz SC, Shia J, Liau KH, Gonen M, Ruo L, Jarnagin WR, et al. Operative blood loss independently predicts recurrence and survival after resection of hepatocellular carcinoma. Ann Surg. 2009;249:617–23.
Tranchart H, O’Rourke N, Van Dam R, Gaillard M, Lainas P, Sugioka A, et al. Bleeding control during laparoscopic liver resection: a review of literature. J Hepatobiliary Pancreat Sci. 2015;22:371–8.
Bressan AK, James MT, Dixon E, Bathe OF, Sutherland FR, Ball CG. Acute kidney injury following resection of hepatocellular carcinoma: prognostic value of the acute kidney injury network criteria. Can J Surg. 2018;61:E11–6.
Chertow GM, Burdick E, Honour M, Bonventre JV, Bates DW. Acute kidney injury, mortality, length of stay, and costs in hospitalized patients. J Am Soc Nephrol. 2005;16:3365–70.
Ratti F, Cipriani F, Reineke R, Catena M, Paganelli M, Comotti L, et al. Intraoperative monitoring of stroke volume variation versus central venous pressure in laparoscopic liver surgery: a randomized prospective comparative trial. HPB (Oxford). 2016;18:136–44.
Soubrane O, Schwarz L, Cauchy F, Perotto LO, Brustia R, Bernard D, et al. A conceptual technique for laparoscopic right hepatectomy based on facts and oncologic principles: the caudal approach. Ann Surg. 2015;261:1226–31.
Brienza N, Giglio MT, Marucci M, Fiore T. Does perioperative hemodynamic optimization protect renal function in surgical patients? A meta-analytic study Crit Care Med. 2009;37:2079–90.
Walsh M, Devereaux PJ, Garg AX, Kurz A, Turan A, Rodseth RN, et al. Relationship between intraoperative mean arterial pressure and clinical outcomes after noncardiac surgery: toward an empirical definition of hypotension. Anesthesiology. 2013;119:507–15.
Sun LY, Wijeysundera DN, Tait GA, Beattie WS. Association of intraoperative hypotension with acute kidney injury after elective noncardiac surgery. Anesthesiology. 2015;123:515–23.
Salmasi V, Maheshwari K, Yang D, Mascha EJ, Singh A, Sessler DI, et al. Relationship between intraoperative hypotension, defined by either reduction from baseline or absolute thresholds, and acute kidney and myocardial injury after noncardiac surgery: a retrospective cohort analysis. Anesthesiology. 2017;126:47–65.
Giglio M, Dalfino L, Puntillo F, Brienza N. Hemodynamic goal-directed therapy and postoperative kidney injury: an updated meta-analysis with trial sequential analysis. Crit Care. 2019;23:232.
Mizuguchi T, Kawamoto M, Meguro M, Hui TT, Hirata K. Preoperative liver function assessments to estimate the prognosis and safety of liver resections. Surg Today. 2014;44:1–10.
Wakabayashi G. What has changed after the Morioka consensus conference 2014 on laparoscopic liver resection? Hepatobiliary Surg Nutr. 2016;5:281–9.
Khwaja A. KDIGO clinical practice guidelines for acute kidney injury. Nephron Clin Pract. 2012;120:c179–84.
Kheterpal S, Tremper KK, Heung M, Rosenberg AL, Englesbe M, Shanks AM, et al. Development and validation of an acute kidney injury risk index for patients undergoing general surgery: results from a national data set. Anesthesiology. 2009;110:505–15.
Kheterpal S, Tremper KK, Englesbe MJ, O’Reilly M, Shanks AM, Fetterman DM, et al. Predictors of postoperative acute renal failure after noncardiac surgery in patients with previously normal renal function. Anesthesiology. 2007;107:892–902.
Ricksten SE, Bragadottir G, Redfors B. Renal oxygenation in clinical acute kidney injury. Crit Care. 2013;17:221.
Kunst G, Ostermann M. Intraoperative permissive oliguria - how much is too much? Br J Anaesth. 2017;119:1075–7.
Turan A, Cohen B, Adegboye J, Makarova N, Liu L, Mascha EJ, et al. Mild acute kidney injury after noncardiac surgery is associated with long-term renal dysfunction: a retrospective cohort study. Anesthesiology. 2020;132:1053–61.
Guest JF, Boyd O, Hart WM, Grounds RM, Bennett ED. A cost analysis of a treatment policy of a deliberate perioperative increase in oxygen delivery in high risk surgical patients. Intensive Care Med. 1997;23:85–90.
Fenwick E, Wilson J, Sculpher M, Claxton K. Pre-operative optimisation employing dopexamine or adrenaline for patients undergoing major elective surgery: a cost-effectiveness analysis. Intensive Care Med. 2002;28:599–608.
Chong MA, Wang Y, Berbenetz NM, McConachie I. Does goal-directed haemodynamic and fluid therapy improve peri-operative outcomes?: A systematic review and meta-analysis. Eur J Anaesthesiol. 2018;35:469–83.
Saner F. Kidney failure following liver resection. Transplant Proc. 2008;40:1221–4.
Sear JW. Kidney dysfunction in the postoperative period. Br J Anaesth. 2005;95:20–32.
Saito K, Uchino S, Fujii T, Saito S, Takinami M, Uezono S. Effect of low-dose atrial natriuretic peptide in critically ill patients with acute kidney injury: a retrospective, single-center study with propensity-score matching. BMC Nephrol. 2020;21:31.
Yamada H, Doi K, Tsukamoto T, Kiyomoto H, Yamashita K, Yanagita M, et al. Low-dose atrial natriuretic peptide for prevention or treatment of acute kidney injury: a systematic review and meta-analysis. Crit Care. 2019;23:41.
Yamada T, Kotake Y, Nagata H, Takeda J. Atrial natriuretic peptide reduces hepatic ischemia-reperfusion injury in rabbits. J Anesth. 2013;27:901–8.
Gregory A, Stapelfeldt WH, Khanna AK, Smischney NJ, Boero IJ, Chen Q, et al. Intraoperative hypotension is associated with adverse clinical outcomes after noncardiac surgery. Anesth Analg. 2021;132:1654–65.
Acknowledgements
The authors wish to thank Mr. Kenta Sato for providing invaluable help with the statistical analyses.
Funding
This study was not funded by any external sources.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
All authors have no conflicts of interest to declare.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Imai, E., Morohashi, Y., Mishima, K. et al. A goal-directed therapy protocol for preventing acute kidney injury after laparoscopic liver resection: a retrospective observational cohort study. Surg Today 52, 1262–1274 (2022). https://doi.org/10.1007/s00595-022-02453-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00595-022-02453-3