Abstract
Purpose
To investigate whether the volume doubling time is a preoperative predictor of lymph node metastasis of clinical stage IA non-small cell lung cancer (NSCLC).
Methods
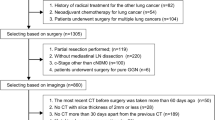
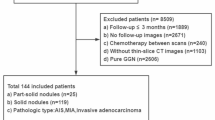
The subjects of this retrospective study were 204 patients who underwent lobectomy and mediastinal lymph node dissection for clinical stage IA NSCLC. We analyzed the relationship between lymph node metastasis and clinicopathological factors, including the volume doubling time.
Results
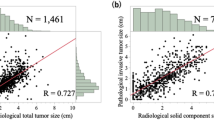
Lymph node metastasis developed in 24 (11.8%) patients. Multivariable analysis identified consolidation diameter (Odds ratio = 1.407; 95% confidence interval, 1.007–1.966, p = 0.046) and the solid-part tumor volume doubling time (Odds ratio = 0.982; 95% confidence interval, 0.973–0.991, p < 0.001) as independent predictors of lymph node metastasis. The combination of a larger consolidation diameter (> 1.9 cm) and a shorter solid-part tumor volume doubling time (< 132 days) had sensitivity, specificity, and accuracy of 79.2%, 94.4%, and 92.6%, respectively.
Conclusions
The consolidation diameter and solid-part tumor volume doubling time can be useful preoperative predictors of lymph node metastasis of clinical stage IA NSCLC.






Similar content being viewed by others
References
Nasim F, Sabath BF, Eapen GA. Lung Cancer Med Clin N Am. 2019;103:463–73.
National Comprehensive Cancer Network. NCCN Guidelines in: Non-Small Cell Lung Cancer; Version 4.2021. https://www.nccn.org/professionals/physician_gls/pdf/nscl.pdf. Accessed 24 Aug 2021.
Asamura H, Nakayama H, Kondo H, Tsuchiya R, Shimosato Y, Naruke T. Lymph node involvement, recurrence, and prognosis in resected small, peripheral, non-small-cell lung carcinomas: are these carcinomas candidates for video-assisted lobectomy? J Thorac Cardiovasc Surg. 1996;111:1125–34.
Darling GE, Allen MS, Decker PA, Ballman K, Malthaner RA, Inculet RI, et al. Randomized trial of mediastinal lymph node sampling versus complete lymphadenectomy during pulmonary resection in the patient with N0 or N1 (less than hilar) non-small cell carcinoma: results of the American College of Surgery Oncology Group Z0030 Trial. J Thorac Cardiovasc Surg. 2011;141:662–70.
Tsutani Y, Miyata Y, Nakayama H, Okumura S, Adachi S, Yoshimura M, et al. Prediction of pathologic node-negative clinical stage IA lung adenocarcinoma for optimal candidates undergoing sublobar resection. J Thorac Cardiocvasc. 2012;144:1365–71.
Takamochi K, Nagai K, Suzuki K, Yoshida J, Ohde Y, Nishiwaki Y. Clinical predictors of N2 disease in non-small cell lung cancer. Chest. 2000;117:1577–82.
Ye B, Cheng M, Li W, Ge XX, Geng JF, Fen J, et al. Predictive factors for lymph node metastasis in clinical stage IA lung adenocarcinoma. Ann Thorac Surg. 2014;98:217–23.
Collins VP, Loeffler RK, Tivey H. Observations on growth rates of human tumors. Am J Roentgenol Radium Ther Nucl Med. 1956;76:988–1000.
Setojima Y, Shimada Y, Tanaka T, Shigefuku S, Makino Y, Maehara S, et al. Prognostic impact of solid-part tumour volume doubling time in patients with radiological part-solid or solid lung cancer. Eur J Cardiothorac Surg. 2020;57:763–70.
Veronesi G, Maisonneuve P, Bellomi M, Rampinelli C, Durli I, Bertolotti R, et al. Estimating overdiagnosis in low-dose computed tomography screening for lung cancer: a cohort study. Ann Intern Med. 2012;157:776–84.
Goldstraw P, Chansky K, Crowley J, Rami-Porta R, Asamura H, Eberhardt WEE, et al. The IASLC lung cancer staging project: proposals for revision of the TNM stage groupings in the forthcoming (eighth) edition of the TNM classification for lung cancer. J Thorac Oncol. 2016;11:39–51.
Suzuki K, Koike T, Asakawa T, Kusumoto M, Asamura H, Nagai K, et al. A prospective radiological study of thin-section computed tomography to predict pathological noninvasiveness in peripheral clinical IA lung cancer (Japan Clinical Oncology Group 0201). J Thorac Oncol. 2011;6:751–6.
Wang YH, Chen CF, Lin YK, Chian C, Tzao C, Yen Y. Predicting malignancy: subsolid nodules detected on LDCT in a surgical cohort of East Asian patients. J Thorac Dis. 2020;12:4315–26.
Schwartz M. A biomathematical approach to clinical tumor growth. Cancer. 1961;14:1272–94.
Okami J, Shintani Y, Okumura M, Ito H, Ohtsuka T, Toyooka S, et al. Demographics, safety and quality, and prognostic information in both the seventh and eighth editions of the TNM classification in 18,973 surgical cases of the Japanese joint committee of lung cancer registry database in 2010. J Thorac Oncol. 2019;14:212–22.
Nakamura K, Saji H, Nakajima R, Okada M, Asamura H, Shibata T, et al. A phase III randomized trial of lobectomy versus limited resection for small-sized peripheral non-small cell lung cancer (JCOG0802/WJOG4607L). Jpn J Clin Oncol. 2010;40:271–4.
Hishida T, Saji H, Watanabe S, Asamura H, Aokage K, Mizutani T, et al. A randomized phase III trial of lobe-specific vs. systematic nodal dissection for clinical Stage I-II non-small cell lung cancer (JCOG1413). Jpn J Clin Oncol. 2018;48:190–4.
Ginsberg RH, Rubinstein LV. Randomized trial of lobectomy versus limited resection for T1N0 non–small cell lung cancer. Lung Cancer Study Group. Ann Thorac Surg. 1995;60:615–23.
Tsutani Y, Miyata Y, Nakayama H, Okumura S, Adachi S, Yoshimura M, et al. Oncologic outcomes of segmentectomy compared with lobectomy or clinical stage IA lung adenocarcinoma: Propensity score-matched analysis in a multicenter study. J Thorac Cardiocvasc. 2013;146:358–64.
Meng D, Zhou Z, Wang Y, Wang L, Lv W, Hu J. Lymphadenectomy for clinical early-stage non-small-cell lung cancer: a systematic review and meta-analysis. Eur J Cardiothorac Surg. 2016;50:597–604.
Nakahashi K, Tsunooka N, Hirayama K, Matsuno M, Endo M, Akahira J, et al. Preoperative predictors of lymph node metastasis in clinical T1 adenocarcinoma. J Thorac Dis. 2020;12:2352–60.
Hattori A, Suzuki K, Maeyashiki T, Fukui M, Kitamura Y, Matsunaga T, et al. The presence of air bronchogram is a novel predictor of negative nodal involvement in radiologically pure-solid lung cancer. Eur J Cardiothorac Surg. 2014;45:699–702.
Miyasaka Y, Suzuki K, Takamochi K, Matsunaga T, Oh S. The maximum standardized uptake value of fluorodeoxyglucose positron emission tomography of the primary tumour is a good predictor of pathological nodal involvement in clinical N0 non-small-cell lung cancer. Eur J Cardiothorac Surg. 2013;44:83–7.
Cho S, Song I, Yang H, Kim K, Jheon S. Predictive factors for node metastasis in patients with clinical stage I non-small cell lung cancer. Ann Thorac Surg. 2013;96:239–46.
Cerfolio RJ, Ojha B, Bryant AS, Bass CS, Bartalucci AA, Mountz JM. The role of FDG-PET scan in staging patients with nonsmall cell carcinoma. Ann Thorac Surg. 2003;76:861–6.
Gupta NC, Tamim WJ, Graeber GG, Bishop HA, Hobbs GR. Mediastinal lymph node sampling following positron emission tomography with fluorodeoxyglucose imaging in lung cancer staging. Chest. 2001;120:521–7.
Li J, Xia T, Yang X. Malignant solitary pulmonary nodules: assessment of mass growth rate and doubling time at follow-up CT. J Thorac Dis. 2018;10:797–806.
Casiraghi M, Travaini LL, Maisonneuve P, Tessitore A, Brambilla D, Agoglia B, et al. Lymph node involvement in T1 non-small-cell lung cancer: could glucose uptake and maximal diameter be predictive criteria? Eur J Cardiothorac Surg. 2011;39:e38-43.
Acknowledgements
We thank Mr. Masataka Taguri, Ph.D., of the Department of Data Science, Yokohama City University, School of Data Science, 22-2 Seto, Kanazawa-ku, Yokohama, Kanagawa, Japan, for assistance with the statistical analyses in this study.
Funding
We received no funding for this study.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
We have no conflicts of interest to declare in association with the present study.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
595_2022_2450_MOESM3_ESM.docx
Fig. S1 Distribution of time from the date when computed tomography detected lung nodules to the date of preoperative computed tomography and (a) the whole-tumor volume doubling time or (b) the solid-part tumor volume doubling time
Rights and permissions
About this article
Cite this article
Nakahashi, K., Shiono, S., Nakatsuka, M. et al. Prediction of lymph node metastasis of clinical stage IA non-small cell lung cancer based on the tumor volume doubling time. Surg Today 52, 1063–1071 (2022). https://doi.org/10.1007/s00595-022-02450-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00595-022-02450-6